Advanced Mercury-Free Spongosine Synthesis for Commercial Scale-Up and High Purity Production
Introduction to Next-Generation Spongosine Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for rare nucleosides, and the technology disclosed in patent CN1910194A represents a significant leap forward in the production of spongosine, also known as 9-(β-D-ribofuranosyl)-2-methoxyadenine. This rare natural product, originally isolated from marine sponges, has garnered attention for its unique structural properties as one of the first O-methyl compounds found in animal tissues, yet its historical synthesis has been plagued by inefficiency and toxicity. The patent outlines a comprehensive methodology that replaces hazardous heavy metal catalysts with modern Lewis acid chemistry, fundamentally altering the economic and safety profile of this valuable pharmaceutical intermediate. By shifting from mercury-based coupling agents to trimethylsilyl trifluoromethanesulfonate (TMSOTf), the process not only enhances reaction kinetics at ambient temperatures but also simplifies the downstream purification workflow significantly. For R&D directors and procurement specialists, understanding this transition is critical, as it directly impacts the cost of goods sold and the environmental compliance of the supply chain. This report analyzes the technical nuances of this innovation, demonstrating how it serves as a reliable foundation for securing a stable supply of high-purity spongosine derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spongosine relied heavily on the method described by Bergmann and Stempien, which necessitated the use of chloromercuric-2-methoxyadenine as a key coupling partner. This legacy approach required refluxing the reaction mixture in xylene, a condition that demands significant energy input and poses substantial safety risks due to the volatility of the solvent and the toxicity of the mercury reagent. Furthermore, the workup procedure was notoriously cumbersome, involving chloroform extraction and multiple recrystallization steps from hot water to isolate the product, which ultimately resulted in a disappointing crude yield of merely 31%. The presence of mercury residues created a severe bottleneck for commercialization, as removing these heavy metals to meet pharmaceutical standards requires expensive and specialized scavenging technologies that inflate production costs. Additionally, the harsh thermal conditions often promoted degradation of the sensitive purine base, leading to a complex impurity profile that complicated quality control and regulatory approval processes for any downstream API applications. These factors combined made the conventional route economically unviable for modern, large-scale manufacturing environments where green chemistry principles are increasingly mandated.
The Novel Approach
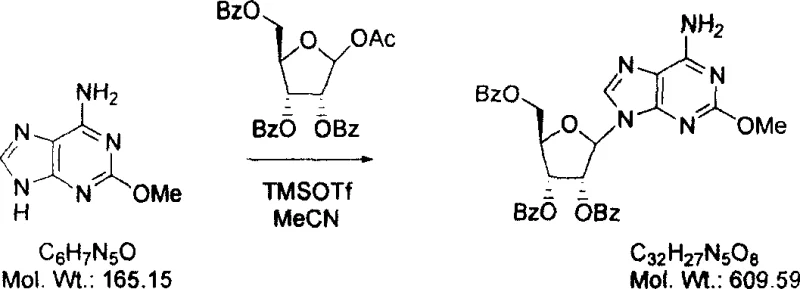
In stark contrast, the novel approach detailed in the patent utilizes 1-O-acetyl-2,3,5-tri-O-benzoyl-beta-D-ribofuranose as the glycosyl donor, reacting it directly with 2-methoxyadenine under mild conditions. This transformation is catalyzed by TMSOTf in anhydrous acetonitrile, allowing the reaction to proceed efficiently at room temperature, thereby eliminating the need for energy-intensive heating and reducing the thermal stress on the reactants. The absence of mercury not only resolves the environmental and safety hazards but also streamlines the isolation process, as the product can be precipitated directly from the organic phase after a simple aqueous wash. This method has been demonstrated to produce the protected intermediate, 9-(2',3',5'-tri-O-benzoyl-beta-D-ribofuranosyl)-2-methoxyadenine, in significantly higher yields compared to the historical benchmark. The subsequent deprotection and crystallization steps are equally optimized, utilizing sodium methoxide and acetic acid to ensure the final spongosine is obtained with exceptional purity. This holistic improvement in the synthetic route positions the technology as a superior choice for any organization seeking a cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into TMSOTf-Catalyzed Glycosylation
The core of this technological advancement lies in the mechanistic efficiency of the Vorbrüggen-type glycosylation facilitated by trimethylsilyl trifluoromethanesulfonate. In this catalytic cycle, TMSOTf acts as a potent Lewis acid that activates the anomeric acetate group of the ribofuranose donor, generating a highly reactive oxocarbenium ion intermediate. This electrophilic species is then attacked by the nucleophilic nitrogen at the N9 position of the 2-methoxyadenine base, forming the crucial N-glycosidic bond with high regioselectivity. The use of benzoyl protecting groups on the sugar moiety plays a vital role in this mechanism, as they provide steric bulk that favors the formation of the beta-anomer while preventing unwanted side reactions at the hydroxyl positions. Unlike the mercury-mediated process which relies on the formation of organometallic complexes, this Lewis acid pathway is purely organic and proceeds through a clean ionic mechanism that is easier to monitor and control. The reaction kinetics are sufficiently fast at room temperature to drive the conversion to completion within hours, minimizing the residence time of the reactants and reducing the opportunity for decomposition. This mechanistic clarity allows process chemists to fine-tune stoichiometry and addition rates to maximize yield, ensuring that the precious purine base is not wasted in non-productive pathways.
Impurity control is another critical aspect where the new mechanism offers distinct advantages over the prior art. In the traditional mercury method, the harsh conditions often led to the formation of N7-isomers and other regioisomers that were difficult to separate from the desired N9-product. The TMSOTf-catalyzed reaction, however, exhibits superior selectivity for the N9-position due to the specific electronic activation of the base and the steric environment created by the silylated intermediates. Furthermore, the workup procedure involving washing with aqueous sodium hydroxide effectively removes any unreacted acid catalyst and silyl by-products, preventing them from carrying over into the final isolation step. The subsequent precipitation of the protected intermediate from dichloromethane allows for a solid-state purification before deprotection, effectively filtering out soluble impurities early in the process. This multi-layered approach to impurity management ensures that the final deprotected spongosine meets stringent purity specifications without requiring extensive chromatographic purification, which is often a cost-prohibitive step in large-scale operations. The result is a robust process capable of delivering high-purity pharmaceutical intermediates consistently.
How to Synthesize Spongosine Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to a precise sequence of unit operations that leverage the chemical advantages of the TMSOTf system. The process begins with the preparation of high-quality 2-methoxyadenine, which serves as the nucleobase scaffold, followed by the critical glycosylation step that constructs the nucleoside backbone. Detailed standard operating procedures for each stage, including reagent grades, temperature controls, and mixing parameters, are essential to replicate the high yields reported in the patent data. The following guide outlines the critical path for production, ensuring that the transition from laboratory scale to commercial manufacturing is seamless and reproducible. Operators should pay particular attention to the moisture control during the glycosylation step, as the presence of water can deactivate the TMSOTf catalyst and compromise the reaction yield.
- Convert 2,6-dichloropurine to 2-chloroadenine using saturated methanolic ammonia at 100°C.
- Synthesize 2-methoxyadenine by heating 2-chloroadenine with sodium methoxide in methanol, followed by pH adjustment to 9.5.
- Perform glycosylation of 2-methoxyadenine with 1-O-acetyl-2,3,5-tri-O-benzoyl-beta-D-ribofuranose using TMSOTf in acetonitrile.
- Deprotect the benzoyl groups using sodium methoxide in methanol at room temperature.
- Crystallize the final spongosine product from acetic acid and ethanol to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this mercury-free synthesis route translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of chloromercuric reagents removes a significant regulatory burden and reduces the costs associated with hazardous waste disposal and environmental compliance monitoring. By operating at room temperature, the process also lowers energy consumption compared to the reflux conditions required by the legacy method, contributing to a lower carbon footprint and reduced utility costs per kilogram of product. These operational efficiencies allow for a more competitive pricing structure, making the supply of spongosine derivatives more resilient against market fluctuations in raw material costs. Furthermore, the simplified workup and isolation steps reduce the overall cycle time, enabling faster turnaround for custom orders and improving the responsiveness of the supply chain to customer demand. This agility is crucial for pharmaceutical clients who require reliable partners capable of scaling production rapidly to meet clinical trial timelines or commercial launch schedules without compromising on quality.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of mercury-based reagents, which eliminates the need for expensive heavy metal scavengers and specialized waste treatment protocols. The higher reaction yield means that less starting material is required to produce the same amount of final product, directly lowering the raw material cost per unit. Additionally, the ability to perform the reaction at ambient temperature reduces the energy load on the manufacturing facility, as there is no need for prolonged heating or complex cooling systems to manage exotherms. The simplified isolation procedure, which relies on precipitation and filtration rather than complex extractions or chromatography, further reduces solvent consumption and labor hours. Collectively, these factors create a leaner manufacturing process that delivers substantial cost savings while maintaining high product quality.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of commercially available and stable reagents such as TMSOTf and protected ribose sugars, which are less prone to supply disruptions than specialized organomercury compounds. The robustness of the reaction conditions means that the process is less sensitive to minor variations in operational parameters, reducing the risk of batch failures and ensuring consistent output. This reliability allows suppliers to offer more secure long-term contracts, giving pharmaceutical buyers confidence in their inventory planning. Moreover, the scalability of the process ensures that production volumes can be increased from hundreds of kilograms to multi-ton quantities without the need for fundamental process redesigns. This flexibility is essential for supporting the growth of drug candidates from early-stage development through to full commercialization.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns perfectly with modern green chemistry initiatives by avoiding toxic heavy metals and reducing solvent usage through efficient precipitation techniques. The deprotection and crystallization steps, as illustrated in the reaction scheme below, utilize common solvents like methanol and acetic acid, which are easier to recover and recycle than the chlorinated solvents often used in older methods.
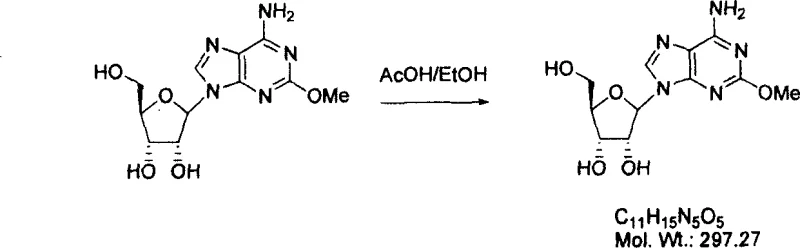 The ease of scaling this chemistry is evidenced by the successful demonstration of the method on multi-hundred gram scales in the patent examples, proving its viability for industrial application. Compliance with strict environmental regulations is simplified, as the effluent streams do not contain persistent mercury contaminants, reducing the liability and administrative overhead for the manufacturing site. This makes the technology not just a chemical improvement, but a sustainable business solution for the future.
The ease of scaling this chemistry is evidenced by the successful demonstration of the method on multi-hundred gram scales in the patent examples, proving its viability for industrial application. Compliance with strict environmental regulations is simplified, as the effluent streams do not contain persistent mercury contaminants, reducing the liability and administrative overhead for the manufacturing site. This makes the technology not just a chemical improvement, but a sustainable business solution for the future.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this spongosine synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the specific advantages over historical methods, providing a clear rationale for the technology's adoption in modern pharmaceutical supply chains.
Q: Why is the TMSOTf method preferred over the traditional Bergmann and Stempien method?
A: The traditional method utilizes toxic chloromercuric-2-methoxyadenine and requires reflux conditions, resulting in a low crude yield of 31%. The TMSOTf method operates at room temperature, eliminates hazardous mercury residues, and significantly improves yield and purity, making it safer and more economically viable for large-scale production.
Q: How does the new process improve impurity control in spongosine synthesis?
A: The novel process employs specific crystallization steps using acetic acid and ethanol, which effectively remove residual solvents and by-products. Additionally, the optimized pH adjustment during the formation of 2-methoxyadenine minimizes side reactions, ensuring the final product achieves purity levels greater than 99% with no single impurity exceeding 0.5%.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the process is explicitly designed for scalability. By removing the need for mercury handling and complex high-temperature reflux setups, the operational complexity is drastically reduced. The use of standard reagents like TMSOTf and sodium methoxide allows for straightforward adaptation from kilogram to multi-ton scales without compromising safety or yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spongosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent supply of high-value pharmaceutical intermediates like spongosine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is executed with precision. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay. Our commitment to quality means that every batch of spongosine produced adheres to the highest industry standards, providing our partners with the confidence needed to advance their drug development programs. By leveraging the mercury-free TMSOTf catalysis technology, we offer a sustainable and cost-effective solution that aligns with the evolving regulatory landscape of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the economic advantages of switching to this greener, more efficient process. Please contact us to request specific COA data and route feasibility assessments tailored to your volume needs. Our goal is to establish a long-term strategic partnership that drives value through innovation, reliability, and unwavering commitment to quality excellence in the supply of complex nucleoside intermediates.
