Advanced Synthesis of Fluoroalkyl Isoxazole Derivatives for Commercial Drug Discovery
Advanced Synthesis of Fluoroalkyl Isoxazole Derivatives for Commercial Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking novel scaffolds that offer enhanced metabolic stability and bioactivity, and the introduction of fluorine atoms into organic molecules has become a cornerstone strategy in modern medicinal chemistry. Patent CN110845432B introduces a groundbreaking methodology for synthesizing fluoroalkyl-substituted isoxazole derivatives, which serve as critical difluoroalkyl building blocks for advanced drug discovery. These compounds uniquely combine a bisfluoromethylene group with an isoxazole ring, a structural motif known to improve lipid regulation properties similar to HMG-CoA reductase inhibitors and enhance fungicidal activity akin to hymexazol. 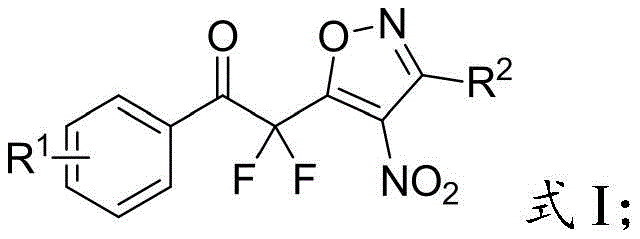 . For R&D directors and procurement specialists, understanding the synthetic accessibility of such complex fluorinated heterocycles is paramount for securing reliable supply chains for next-generation active pharmaceutical ingredients (APIs) and agrochemical intermediates.
. For R&D directors and procurement specialists, understanding the synthetic accessibility of such complex fluorinated heterocycles is paramount for securing reliable supply chains for next-generation active pharmaceutical ingredients (APIs) and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of molecules containing both a difluoromethylene group and a heterocyclic ring like isoxazole has been fraught with significant challenges, often requiring multi-step sequences with poor atom economy and harsh reaction conditions. Conventional fluorination techniques frequently rely on expensive transition metal catalysts or hazardous reagents that complicate purification and increase the environmental footprint of the manufacturing process. Furthermore, achieving regioselective introduction of fluorine atoms adjacent to carbonyl groups without affecting other sensitive functional groups on the aromatic ring often necessitates tedious protection and deprotection strategies. These inefficiencies lead to extended lead times, higher production costs, and inconsistent batch-to-batch quality, which are critical pain points for supply chain managers aiming to maintain continuity in the production of high-value fine chemicals and pharmaceutical intermediates.
The Novel Approach
The innovative process disclosed in the patent offers a streamlined, three-step synthetic route that elegantly overcomes these historical bottlenecks by utilizing readily available starting materials and mild reaction conditions. The method begins with a nucleophilic addition between a nitro-isoxazole and an aromatic aldehyde, followed by a selective oxidation and a final fluorination step using a specific fluorinating agent. 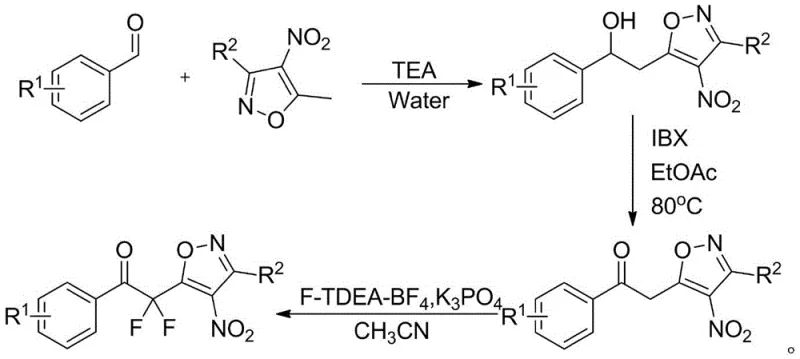 . This approach eliminates the need for complex protecting groups and allows for the direct construction of the carbon-fluorine bond with high selectivity. By employing common solvents like ethyl acetate and acetonitrile, and avoiding cryogenic temperatures, this novel approach significantly simplifies the operational complexity, making it an ideal candidate for cost reduction in fine chemical manufacturing and facilitating easier technology transfer from laboratory to pilot plant scales.
. This approach eliminates the need for complex protecting groups and allows for the direct construction of the carbon-fluorine bond with high selectivity. By employing common solvents like ethyl acetate and acetonitrile, and avoiding cryogenic temperatures, this novel approach significantly simplifies the operational complexity, making it an ideal candidate for cost reduction in fine chemical manufacturing and facilitating easier technology transfer from laboratory to pilot plant scales.
Mechanistic Insights into Selective Fluorination and Oxidation
The core of this synthetic strategy lies in the precise control of oxidation states and the subsequent electrophilic fluorination of the alpha-position of the ketone. The process initiates with a base-catalyzed Henry-like reaction where the acidic methyl proton of the 3-substituted-4-nitro-5-methylisoxazole attacks the carbonyl carbon of the aromatic aldehyde, forming a beta-hydroxy ketone precursor after tautomerization. This intermediate is then subjected to oxidation using 2-iodoxybenzoic acid (IBX), a hypervalent iodine reagent known for its ability to oxidize alcohols to ketones under mild thermal conditions, typically around 80°C in ethyl acetate. This step is crucial as it generates the necessary enolizable ketone substrate required for the subsequent fluorination, ensuring that the reaction proceeds without over-oxidation or degradation of the sensitive nitro-isoxazole moiety.
The final and most critical transformation involves the use of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2,2,2]octane bis(tetrafluoroborate) salt, commonly known as Selectfluor or F-TEDA-BF4, in the presence of a phosphate base. This reagent acts as a source of electrophilic fluorine, attacking the enolate formed at the alpha-position of the ketone to install the first fluorine atom, followed by a second fluorination event to yield the gem-difluoro methylene group. 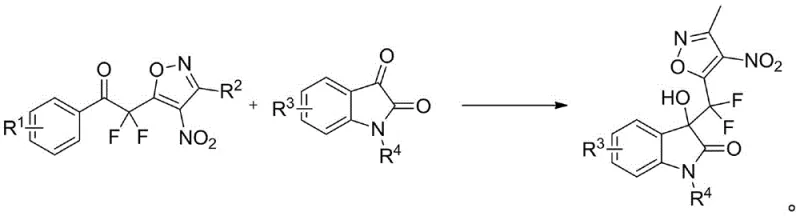 . The choice of potassium phosphate as a catalyst ensures that the pH is maintained at a level that promotes enolate formation without causing hydrolysis of the isoxazole ring. This mechanistic precision results in high purity products with minimal halogenated impurities, a key requirement for reliable pharmaceutical intermediates supplier standards where impurity profiles must be strictly controlled.
. The choice of potassium phosphate as a catalyst ensures that the pH is maintained at a level that promotes enolate formation without causing hydrolysis of the isoxazole ring. This mechanistic precision results in high purity products with minimal halogenated impurities, a key requirement for reliable pharmaceutical intermediates supplier standards where impurity profiles must be strictly controlled.
How to Synthesize Fluoroalkyl Isoxazole Derivatives Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to strict stoichiometric controls and reaction monitoring protocols to ensure maximum yield and purity. The process is designed to be robust, utilizing a molar ratio of approximately 1.00:1.1 for the isoxazole to aldehyde reactants, with catalytic amounts of triethylamine to drive the initial condensation. Detailed standard operating procedures regarding solvent volumes, workup techniques involving ethyl acetate extraction, and purification via column chromatography or recrystallization are essential for reproducibility.
- Perform nucleophilic addition of 3-substituted-4-nitro-5-methylisoxazole with aromatic aldehydes using triethylamine catalyst in water or acetonitrile at room temperature.
- Oxidize the resulting alcohol intermediate to a ketone using 2-iodoxybenzoic acid (IBX) in ethyl acetate under reflux conditions at 80°C.
- Conduct alpha-fluorination of the ketone intermediate using F-TEDA-BF4 and potassium phosphate catalyst in acetonitrile to yield the final difluoro product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty, directly impacting the bottom line through operational efficiency and raw material optimization. The elimination of transition metal catalysts removes the need for expensive metal scavenging steps and reduces the risk of heavy metal contamination in the final product, which is a frequent cause of batch rejection in regulated industries. Furthermore, the use of stable, shelf-stable reagents like IBX and F-TEDA-BF4 ensures that inventory management is simplified, reducing the risks associated with handling hazardous or unstable fluorinating agents that require special storage conditions.
- Cost Reduction in Manufacturing: The streamlined three-step sequence significantly reduces the number of unit operations required compared to traditional multi-step syntheses, leading to substantial cost savings in labor, energy, and solvent consumption. By avoiding the use of precious metal catalysts and utilizing cost-effective oxidants and fluorinating agents, the overall cost of goods sold (COGS) for these complex fluorinated building blocks is drastically lowered. Additionally, the high selectivity of the reaction minimizes the formation of side products, thereby improving the overall yield and reducing the waste disposal costs associated with purifying complex mixtures.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted benzaldehydes and nitro-isoxazoles, are commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of substituents on the aromatic ring (such as chloro, bromo, and nitro groups), allows for the flexible sourcing of raw materials without compromising the integrity of the final product. This flexibility ensures a continuous supply of high-purity pharmaceutical intermediates even in the face of market fluctuations or logistical disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with standard laboratory glassware and easily adaptable to larger reactor vessels without requiring specialized high-pressure or cryogenic equipment. The use of greener solvents like ethyl acetate and water in the initial steps aligns with modern environmental, health, and safety (EHS) guidelines, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing process. This compliance is increasingly important for companies aiming to meet stringent sustainability goals while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluoroalkyl-substituted isoxazole derivatives, providing clarity for stakeholders evaluating this technology for their own pipelines. These insights are derived directly from the experimental data and scope defined within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of this fluorination method over traditional approaches?
A: This method allows for the simultaneous introduction of a difluoromethylene group and an isoxazole ring in a streamlined three-step sequence, avoiding complex protection-deprotection strategies often required in conventional fluorination techniques.
Q: Can this synthesis be scaled for industrial production of API intermediates?
A: Yes, the process utilizes commercially available reagents like IBX and F-TEDA-BF4 under mild conditions (room temperature to 80°C) and standard solvents, making it highly suitable for commercial scale-up without requiring specialized high-pressure equipment.
Q: What is the biological significance of the resulting difluoro-isoxazole scaffold?
A: The scaffold combines the metabolic stability of the difluoromethylene group with the bioactive isoxazole heterocycle, creating a valuable building block for developing HMG-CoA reductase inhibitors and novel fungicides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl Isoxazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorinated building blocks play in the development of next-generation therapeutics and agrochemicals, and we are committed to delivering these complex molecules with the highest standards of quality and reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of fluoroalkyl-substituted isoxazole derivatives meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs, whether you are exploring new chemical entities or optimizing existing supply chains. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements, and to obtain specific COA data and route feasibility assessments for your target molecules. Together, we can accelerate the development of life-saving medicines and sustainable agricultural solutions through superior chemical manufacturing.
