Advanced Catalytic Synthesis of 1-p-Methylphenyl-4,4,4-trifluoro-1,3-butanedione for Commercial Scale-up
Advanced Catalytic Synthesis of 1-p-Methylphenyl-4,4,4-trifluoro-1,3-butanedione for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable pathways for critical active pharmaceutical ingredient (API) intermediates, particularly for high-value drugs like Celecoxib. A significant technological breakthrough in this domain is detailed in patent CN106892806B, which discloses a highly efficient method for preparing 1-p-methylphenyl-4,4,4-trifluoro-1,3-butanedione. This compound serves as a pivotal building block in the synthesis of selective non-steroidal anti-inflammatory drugs (NSAIDs). Unlike legacy processes that rely on harsh conditions or expensive reagents, this invention utilizes a catalytic acylation strategy between trifluoroacetyl halides and p-methylacetophenone. The result is a process characterized by mild reaction parameters, exceptional yield consistency, and a simplified work-up procedure that drastically reduces environmental burden. For R&D teams and procurement specialists alike, understanding the structural nuances of the target molecule is essential, as it exists in dynamic equilibrium between keto and enol forms.
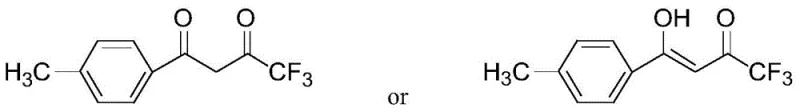
This structural flexibility is managed effectively within the new process, ensuring that the final isolated solid meets stringent purity specifications required for GMP manufacturing. By shifting the synthetic paradigm from traditional condensation to direct acylation, the patent offers a compelling solution for cost reduction in pharmaceutical intermediates manufacturing, addressing both economic and operational inefficiencies inherent in older technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 1-p-methylphenyl-4,4,4-trifluoro-1,3-butanedione has been plagued by significant technical and economic hurdles that hinder large-scale commercial viability. Early literature, such as the work by Penning et al., described a Claisen condensation route using ethyl trifluoroacetate and sodium methoxide, which notoriously suffered from low reaction yields of approximately 71.8%. Furthermore, this legacy method necessitates a cumbersome post-reaction work-up involving acidification with hydrochloric acid, multiple extraction steps with ethyl acetate, and drying procedures that increase solvent consumption and waste generation. Alternative approaches attempted to mitigate yield issues by employing strong bases like lithium bistrimethylsilyl amide; however, these methods introduced severe safety and cost liabilities. Specifically, the requirement for cryogenic reaction temperatures ranging from -60°C to -50°C demands specialized refrigeration infrastructure that is energy-intensive and difficult to maintain on a multi-ton scale. Additionally, the strict necessity for anhydrous and oxygen-free conditions complicates reactor operations, increasing the risk of batch failure and posing safety risks associated with handling reactive organometallic reagents.
The Novel Approach
In stark contrast to these restrictive legacy protocols, the novel approach outlined in the patent data leverages a direct acylation mechanism that operates under remarkably mild and industrially friendly conditions. By reacting p-methylacetophenone directly with trifluoroacetyl halides (such as fluoride, chloride, or bromide) in the presence of accessible catalysts like sodium ethoxide or triethylamine, the process eliminates the need for extreme低温 (low temperatures). The reaction proceeds efficiently at temperatures between 20°C and 50°C, which are easily achievable with standard cooling water systems rather than expensive cryogenic brine. This shift not only simplifies the engineering requirements for the reactor but also significantly enhances the safety profile of the operation by removing hazardous reagents and extreme thermal stresses. Moreover, the work-up procedure is streamlined to a simple filtration and crystallization step, avoiding the complex acid-base extractions of the past. This modernization of the synthetic route represents a paradigm shift towards greener chemistry, aligning with global trends for reducing solvent usage and energy consumption in fine chemical production.
Mechanistic Insights into Catalytic Acylation
The core of this technological advancement lies in the precise manipulation of nucleophilic acyl substitution dynamics. In this catalytic cycle, the base catalyst activates the p-methylacetophenone by abstracting an alpha-proton to generate a reactive enolate species in situ. This nucleophilic enolate then attacks the electrophilic carbonyl carbon of the trifluoroacetyl halide. The presence of the highly electronegative trifluoromethyl group enhances the electrophilicity of the acyl halide, facilitating a rapid and high-yielding coupling reaction even at ambient or slightly elevated temperatures. The choice of catalyst is critical; alkoxides like sodium methoxide or amines like triethylamine provide the optimal basicity to drive enolate formation without promoting undesirable side reactions such as self-condensation of the ketone. The stoichiometry is carefully balanced, typically employing a molar ratio of catalyst to ketone between 0.2:1 and 2:1, ensuring complete conversion while minimizing excess reagent costs.
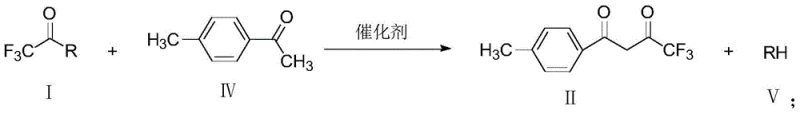
As illustrated in the reaction scheme above, the byproduct is simply a hydrogen halide salt (RH), which is easily removed during the filtration or washing stages, contributing to the high purity of the crude product. Impurity control is inherently superior in this system because the reaction kinetics favor the cross-coupling over homocoupling. The rapid consumption of the acyl halide prevents the accumulation of reactive intermediates that could lead to polymeric byproducts. Furthermore, the ability to isolate the product via crystallization from solvents like acetonitrile or toluene allows for the rejection of trace impurities, resulting in a final solid with content levels consistently exceeding 99.0%. This mechanistic clarity provides R&D directors with confidence in the reproducibility and robustness of the process when transferring from laboratory benchtop to pilot plant scales.
How to Synthesize 1-p-Methylphenyl-4,4,4-trifluoro-1,3-butanedione Efficiently
Implementing this synthesis requires careful attention to solvent selection and addition rates to maximize thermal control and yield. The patent examples demonstrate that solvents such as diethyl ether, isopropyl ether, and toluene are highly effective, offering good solubility for reactants while facilitating easy removal during the concentration phase. The process generally involves pre-mixing the catalyst and ketone, followed by the controlled introduction of the acyl halide to manage exotherms. Detailed standardized operating procedures regarding specific addition sequences, stirring speeds, and crystallization cooling ramps are critical for maintaining batch-to-batch consistency. For a comprehensive guide on the exact operational parameters validated in the patent examples, please refer to the technical protocol below.
- Prepare the reaction system by dissolving the catalyst (such as sodium ethoxide or triethylamine) and p-methylacetophenone in an organic solvent like toluene or diethyl ether under stirring.
- Introduce the trifluoroacetyl halide (fluoride, chloride, or bromide) into the reaction mixture and maintain the temperature between 20°C and 50°C for several hours.
- Perform filtration to remove salts, distill off the solvent partially, and crystallize the residue at low temperatures (-10°C to 10°C) to obtain the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel acylation process offers tangible strategic benefits that extend beyond simple chemistry. The elimination of cryogenic requirements and hazardous organolithium reagents translates directly into a more resilient and cost-effective supply chain. By relying on commodity chemicals like p-methylacetophenone and trifluoroacetyl chloride, manufacturers can secure raw material sourcing from multiple global vendors, reducing dependency on single-source specialty reagents. This diversification of the supply base is crucial for mitigating risks associated with market volatility and logistical disruptions. Furthermore, the simplified work-up procedure reduces the consumption of auxiliary solvents and processing time, leading to substantial cost savings in utility and waste disposal sectors.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and specialized reagents. Traditional methods requiring lithium bistrimethylsilyl amide involve high material costs and specialized handling protocols that inflate the overall cost of goods sold (COGS). By switching to inexpensive catalysts like sodium ethoxide or triethylamine and avoiding the capital expenditure associated with -60°C reactors, manufacturers can achieve a significantly lower production cost base. Additionally, the high yield (>90%) ensures that raw material utilization is maximized, further driving down the effective cost per kilogram of the API intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher schedule reliability. Processes that operate at mild temperatures (20-50°C) are less susceptible to equipment failures or thermal runaway incidents compared to cryogenic processes. This stability allows for tighter production scheduling and shorter lead times for high-purity pharmaceutical intermediates. The use of common industrial solvents like toluene and methanol also ensures that solvent supply chains remain stable and unaffected by niche market fluctuations, guaranteeing continuous production capability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is vastly superior. The avoidance of strong acidification steps and the reduction in solvent volume for extractions minimize the generation of hazardous wastewater. The byproduct formed is a manageable salt, simplifying effluent treatment. These factors make the commercial scale-up of complex pharmaceutical intermediates much easier to permit and operate within strict regulatory frameworks, ensuring long-term sustainability for the manufacturing site.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is vital for stakeholders evaluating its adoption for commercial production. The following questions address common concerns regarding yield optimization, safety profiles, and product quality, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational advantages and assist in the decision-making process for technology transfer.
Q: How does this new acylation method improve yield compared to traditional Claisen condensation?
A: Traditional Claisen condensation methods often suffer from yields around 71.8% and require complex acidification steps. This novel catalytic acylation approach consistently achieves yields exceeding 90% by utilizing reactive trifluoroacetyl halides under milder conditions, significantly reducing material loss.
Q: What are the safety advantages of using trifluoroacetyl halides over lithium reagents?
A: Previous methods utilizing lithium bistrimethylsilyl amide required dangerous cryogenic conditions (-60°C) and strict anhydrous environments. The new process operates at mild temperatures (20-50°C) using standard industrial solvents, eliminating the need for expensive cryogenic equipment and hazardous reagents.
Q: Is the product obtained as a pure keto form or a mixture?
A: The synthesis produces 1-p-methylphenyl-4,4,4-trifluoro-1,3-butanedione which may exist in equilibrium with its enol-type isomer. The process allows for the isolation of high-purity solids (content >99%) through controlled crystallization, suitable for downstream pharmaceutical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-p-Methylphenyl-4,4,4-trifluoro-1,3-butanedione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. We are equipped with state-of-the-art reactors capable of handling the mild thermal profiles required by this acylation process, alongside stringent purity specifications enforced by our rigorous QC labs. Our commitment to quality ensures that every batch of 1-p-methylphenyl-4,4,4-trifluoro-1,3-butanedione meets the exacting standards necessary for downstream Celecoxib synthesis.
We invite global partners to collaborate with us to leverage this efficient synthetic route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior economics and reliability of our manufacturing capabilities before committing to long-term supply agreements.
