Advanced One-Pot Synthesis of Mefenpyr-Diethyl for Scalable Agrochemical Production
Advanced One-Pot Synthesis of Mefenpyr-Diethyl for Scalable Agrochemical Production
The global demand for high-efficiency herbicide safeners continues to surge as agricultural sectors seek to maximize crop yields while minimizing phytotoxicity. In this context, the preparation method disclosed in patent CN111592493A represents a pivotal technological breakthrough for the synthesis of mefenpyr-diethyl, a critical pyrazole-type antidote used to protect wheat and barley. This patent introduces a novel one-pot synthetic strategy that fundamentally alters the traditional manufacturing landscape by stabilizing reactive intermediates and eliminating cumbersome isolation steps. For R&D directors and process engineers, the significance lies in the transformation of a hazardous, multi-step batch process into a streamlined, continuous-flow-compatible operation. By integrating diazotization, substitution, hydrolysis, and cyclization into a cohesive sequence, the technology not only enhances operational safety but also drastically improves the overall atom economy of the production line. This report analyzes the technical merits of this innovation, providing a comprehensive roadmap for procurement and supply chain leaders seeking reliable agrochemical intermediate suppliers who can deliver high-purity materials with consistent quality and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of mefenpyr-diethyl has been plagued by significant safety hazards and inefficiencies inherent to the handling of unstable diazonium salts. As illustrated in the prior art referenced within the patent documentation, traditional routes such as those disclosed in EP0635996 require the separate preparation and isolation of 2,4-dichlorophenylhydrazine or its diazonium salt precursors. These intermediates are notoriously thermally unstable; specifically, the diazonium salt generated from 2,4-dichloroaniline decomposes rapidly at neutral temperatures above 10°C, posing severe explosion risks if cooling systems fail during large-scale production. Furthermore, conventional coupling reactions often rely on excessive amounts of sodium acetate—typically 6 to 10 molar equivalents relative to the aniline substrate—which drives up raw material costs and generates substantial solid waste. The necessity to isolate intermediates after each reaction stage not only extends the production cycle time but also leads to cumulative yield losses, making the process economically unsustainable for high-volume commercial scale-up of complex agrochemical intermediates.
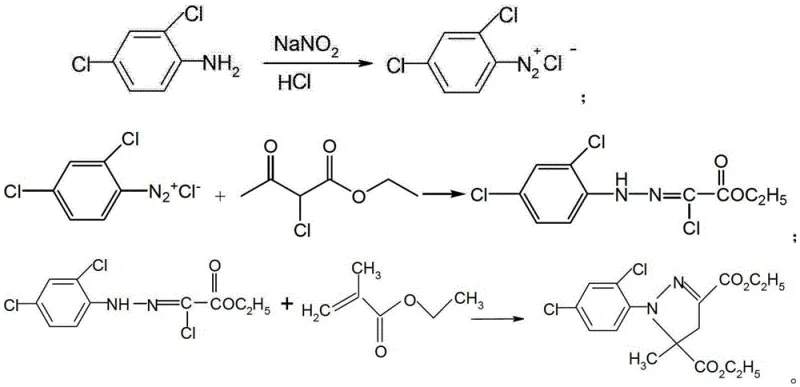
The Novel Approach
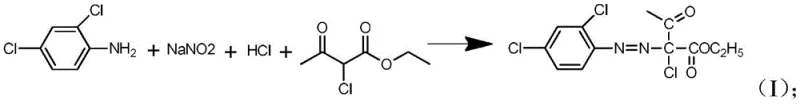
In stark contrast to these legacy methods, the technology described in CN111592493A employs a sophisticated one-pot methodology that mitigates risk while enhancing efficiency. The core innovation involves the immediate consumption of the transient diazonium species by introducing ethyl 2-chloroacetoacetate directly into the diazotization mixture. This ensures that the highly reactive diazonium salt never accumulates in the reactor; instead, it instantly undergoes a substitution reaction to form a remarkably stable intermediate, 2-chloro-2-(2,4-dichlorobenzenediazo) ethyl acetoacetate. This stable species can withstand temperatures up to 80°C without decomposition, effectively removing the thermal runaway risks associated with traditional diazotization. Moreover, the process eliminates the need for intermediate filtration and drying, allowing the reaction mixture to proceed directly to hydrolysis and cyclization. By replacing the expensive and wasteful sodium acetate catalyst with inexpensive sodium or potassium bicarbonate, the method achieves a dual benefit of cost reduction in herbicide safener manufacturing and a significant decrease in the volume of chemical waste requiring treatment, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into One-Pot Diazotization and Cyclization
The mechanistic elegance of this synthesis lies in the precise control of reaction kinetics and pH levels to favor the formation of the desired pyrazole ring system. The process initiates with the diazotization of 2,4-dichloroaniline in an acidic aqueous medium at low temperatures, typically between -10°C and 15°C. Unlike standard protocols where the diazonium salt is isolated, here it reacts in situ with the enol form of ethyl 2-chloroacetoacetate. This rapid coupling prevents the accumulation of hazardous nitrogen-rich species and locks the molecular structure into a stable azo-configured intermediate. Following this, the reaction environment is carefully neutralized to a pH of 5-7 using an inorganic base, creating the optimal conditions for the subsequent hydrolysis step. The addition of a bicarbonate aqueous solution facilitates the conversion of the acetoacetate moiety into the corresponding hydrazone derivative, 2-chloro-2-(2,4-dichlorophenylhydrazone) ethyl acetate, which serves as the direct precursor for the final ring closure.
The final cyclization stage is a critical determinant of product purity and yield, relying on a condensation reaction with ethyl methacrylate. To prevent the premature polymerization of the methacrylate monomer, a polymerization inhibitor such as hydroquinone is introduced prior to heating. The reaction mixture is then heated to a moderate temperature range of 50-80°C, preferably 60-65°C, for a duration of 8 to 12 hours. During this phase, the bicarbonate acts as a mild base to scavenge the hydrogen chloride byproduct generated during the ring closure, driving the equilibrium towards the formation of the pyrazoline structure. This controlled thermal environment ensures that the cyclization proceeds to completion without degrading the sensitive chloro-substituents on the aromatic ring. The result is a high-purity mefenpyr-diethyl product with a reported yield of up to 92.8%, demonstrating superior impurity control compared to multi-step batch processes where exposure to air and moisture during isolation often leads to oxidative degradation.
How to Synthesize Mefenpyr-Diethyl Efficiently
Implementing this advanced synthesis route requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize the benefits of the one-pot design. The process begins by charging the reactor with ethyl 2-chloroacetoacetate, 2,4-dichloroaniline, and hydrochloric acid, followed by the controlled addition of sodium nitrite to initiate diazotization. Once the stable intermediate is confirmed, the pH is adjusted, and the hydrolysis and cyclization reagents are added sequentially without breaking the vacuum or opening the vessel. This seamless workflow minimizes operator exposure to hazardous chemicals and reduces the potential for contamination. For detailed operational parameters, including specific mixing rates and quenching procedures, please refer to the standardized synthesis guide below which outlines the exact execution protocol derived from the patent examples.
- Mix ethyl 2-chloroacetoacetate, 2,4-dichloroaniline, inorganic acid, and water, then add sodium nitrite solution at -10 to 15°C to form the stable diazo intermediate.
- Adjust pH to 5-7 with inorganic base, add bicarbonate solution for hydrolysis to obtain the hydrazone intermediate without separation.
- Add polymerization inhibitor and ethyl methacrylate, followed by bicarbonate solution, and heat to 50-80°C for cyclization to yield mefenpyr-diethyl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond simple unit price negotiations. The shift from a multi-step isolation process to a telescoped one-pot reaction fundamentally alters the cost structure of mefenpyr-diethyl production. By eliminating the unit operations associated with filtering, washing, and drying intermediate solids, manufacturers can significantly reduce energy consumption and labor hours per batch. Furthermore, the replacement of high-volume sodium acetate with low-cost bicarbonates directly lowers the bill of materials, while the reduction in solid waste simplifies effluent treatment compliance. These factors combine to create a more resilient supply chain capable of sustaining high-volume output without the bottlenecks typically associated with complex intermediate handling.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the workflow and the substitution of expensive reagents. Traditional methods require 6 to 10 equivalents of sodium acetate, a cost factor that is entirely removed in favor of cheap sodium or potassium bicarbonate. Additionally, the elimination of intermediate separation steps means that solvent usage is minimized, and the loss of material during transfer and filtration is virtually eradicated. This streamlined approach allows for a substantial reduction in the overall cost of goods sold (COGS), enabling suppliers to offer more competitive pricing structures while maintaining healthy margins. The ability to recover and recycle unreacted ethyl methacrylate from the oil layer further enhances the economic efficiency of the process, ensuring that raw material utilization is maximized throughout the production cycle.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity, and the instability of diazonium salts in conventional routes presents a persistent risk of plant shutdowns due to thermal incidents. By generating a stable intermediate that does not decompose below 80°C, this new method removes the potential for catastrophic decomposition events that could halt production for extended periods. This inherent stability allows for more flexible scheduling and reduces the need for specialized cryogenic infrastructure, making the manufacturing process robust against utility fluctuations. Consequently, suppliers utilizing this technology can guarantee more consistent lead times for high-purity agrochemical intermediates, as the risk of unplanned downtime caused by safety interlocks or emergency quenching is significantly mitigated.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates waste management challenges, but this one-pot technique inherently limits the generation of three wastes (wastewater, waste gas, and solid residue). The absence of solid intermediate cakes reduces the volume of solid waste requiring disposal, while the use of bicarbonate buffers results in less saline wastewater compared to acetate-based systems. This cleaner profile simplifies the permitting process for capacity expansion and ensures compliance with increasingly stringent environmental regulations. The mild reaction conditions and reduced solvent load make the process highly amenable to scale-up from pilot plants to multi-ton commercial reactors, ensuring that supply can grow in tandem with market demand for herbicide safeners without encountering environmental bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing networks or for procurement specialists verifying the quality claims of potential suppliers.
Q: Why is the new one-pot method for mefenpyr-diethyl safer than conventional routes?
A: The novel process avoids the isolation of unstable diazonium salts by immediately reacting them in-situ with ethyl 2-chloroacetoacetate to form a stable intermediate that does not decompose below 80°C.
Q: How does this patent reduce production costs for herbicide safeners?
A: Costs are reduced by eliminating multi-step separation processes, replacing expensive sodium acetate catalysts with cheap sodium bicarbonate, and significantly lowering three-waste generation.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization reaction typically proceeds at temperatures between 50-80°C, preferably 60-65°C, over a period of 8-12 hours using a bicarbonate aqueous solution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mefenpyr-Diethyl Supplier
As the agricultural sector demands increasingly efficient and safe crop protection solutions, the ability to produce high-quality safeners like mefenpyr-diethyl becomes a critical differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies such as the one described in CN111592493A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global agrochemical companies. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of mefenpyr-diethyl meets the exacting standards required for formulation into final herbicide products. Our commitment to process safety and environmental stewardship aligns perfectly with the sustainable manufacturing goals of our partners.
We invite you to collaborate with us to optimize your supply chain for herbicide safeners. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates exactly how our optimized synthesis routes can lower your total procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs. Let us help you secure a stable, cost-effective, and high-quality supply of mefenpyr-diethyl that supports your long-term growth in the agrochemical market.
