Advanced Catalytic Synthesis of Cycloaliphatic Diol Ethers for Commercial Scale Manufacturing
Advanced Catalytic Synthesis of Cycloaliphatic Diol Ethers for Commercial Scale Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust synthetic routes that balance high purity with economic viability, particularly for complex intermediates like cycloaliphatic diol ethers. Patent CN111479795A introduces a transformative methodology for preparing dialkyl or dienyl ethers of cycloaliphatic or araliphatic diols, addressing long-standing challenges in selectivity and reagent costs. This innovation utilizes a catalytic amount of specific monoether-monoalcohols in conjunction with metallic sodium to drive the deprotonation of diol substrates to completion. By shifting away from stoichiometric expensive bases like sodium hydride, this process offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates. The technical breakthrough lies in the ability to achieve near-quantitative conversion while minimizing the formation of stubborn mono-ether impurities that typically plague direct alkylation reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of diethers from diols often relies on harsh conditions or prohibitively expensive reagents that hinder commercial scalability. Conventional protocols frequently employ sodium hydride (NaH) or strong Lewis acids, which not only inflate raw material costs but also introduce significant safety hazards due to hydrogen gas evolution and pyrophoric nature. Furthermore, direct alkylation of diols is notoriously difficult to drive to completion; reaction kinetics often stall at the mono-ether stage, resulting in product mixtures contaminated with substantial amounts of unreacted diol and mono-alkylated species. To force the reaction forward, practitioners historically applied extreme temperatures or prolonged reaction times, which unfortunately promoted elimination side reactions and degradation of the sensitive cycloaliphatic backbone. These inefficiencies necessitate tedious downstream purification steps, such as multiple distillations or chromatographic separations, which drastically reduce overall process mass intensity and increase the environmental footprint of the manufacturing operation.
The Novel Approach
The novel approach detailed in the patent data circumvents these bottlenecks by introducing a catalytic promoter system that fundamentally alters the reaction interface between the metal and the substrate. By employing specific monoether-monoalcohols of formula (I) alongside metallic sodium, the process ensures that the sodium surface remains active and accessible throughout the deprotonation phase. This strategy allows for the efficient generation of the disodium glycol intermediate under much milder conditions than previously possible. The versatility of this method is demonstrated by its applicability to a wide range of structurally diverse diols, including various substituted cyclohexane and benzene derivatives. 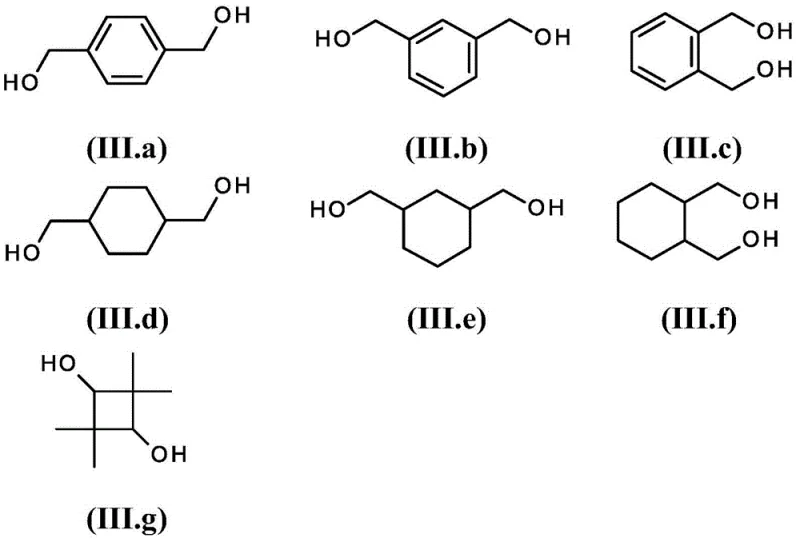 This broad substrate scope means that a single optimized protocol can be adapted for multiple high-value intermediates, streamlining technology transfer and reducing the need for bespoke process development for each new compound. The result is a cleaner reaction profile with significantly higher selectivity for the desired diether product.
This broad substrate scope means that a single optimized protocol can be adapted for multiple high-value intermediates, streamlining technology transfer and reducing the need for bespoke process development for each new compound. The result is a cleaner reaction profile with significantly higher selectivity for the desired diether product.
Mechanistic Insights into Monoether-Monoalcohol Catalyzed Deprotonation
The core mechanistic advantage of this invention stems from the interaction between the catalytic monoether-monoalcohol and the metallic sodium surface during the initial deprotonation step. In the absence of the catalyst, the freshly formed disodium salt of the diol tends to coat the surface of the sodium metal, effectively passivating it and preventing further reaction with the remaining diol molecules. This phenomenon, known as surface encapsulation, is the primary cause of incomplete conversion in standard metallic sodium reactions. The presence of the formula (I) additive, as depicted in the structural diagram below, disrupts this passivation layer. 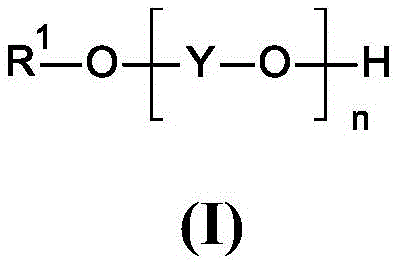 It is hypothesized that the ether oxygen atoms coordinate with the sodium cations, increasing the solubility or dispersibility of the alkoxide species, thereby keeping the metal surface exposed and reactive. This ensures that the deprotonation proceeds essentially to completion, generating the full equivalent of the disodium glycolate required for the subsequent alkylation step without the need for a large excess of base.
It is hypothesized that the ether oxygen atoms coordinate with the sodium cations, increasing the solubility or dispersibility of the alkoxide species, thereby keeping the metal surface exposed and reactive. This ensures that the deprotonation proceeds essentially to completion, generating the full equivalent of the disodium glycolate required for the subsequent alkylation step without the need for a large excess of base.
Furthermore, this catalytic system plays a critical role in thermal management and impurity control during the subsequent alkylation phase. The formation of the disodium intermediate in a controlled manner allows for a more uniform reaction rate when the alkylating agent is introduced. In comparative examples provided within the patent data, reactions lacking the catalyst exhibited violent exotherms and gas evolution upon addition of the alkylating agent, indicative of uncontrolled side reactions and residual reactivity. In contrast, the catalyzed process showed a mild thermal profile, which directly correlates to the suppression of elimination byproducts. By maintaining a stable reaction temperature, typically between 70°C and 180°C depending on the specific solvent system, the process minimizes the energy input required for purification and ensures that the final impurity profile meets the stringent specifications required for reliable pharmaceutical intermediate supplier standards.
How to Synthesize 1,4-Bis(ethoxymethyl)cyclohexane Efficiently
The synthesis of 1,4-bis(ethoxymethyl)cyclohexane serves as a prime example of the operational simplicity and efficacy of this patented technology. The procedure begins with the emulsification of metallic sodium in an aprotic solvent such as xylene, facilitated by a small molar percentage of a promoter like diethylene glycol monomethyl ether. Once the sodium is finely dispersed, the diol substrate is added gradually to manage hydrogen evolution, forming the reactive disodium salt. Following the complete consumption of the diol, an alkylating agent such as diethyl sulfate is introduced to the mixture. The detailed standardized synthesis steps, including specific stoichiometric ratios, temperature ramps, and workup procedures, are outlined in the guide below for technical reference.
- Emulsify metallic sodium in an aprotic solvent with a catalytic amount of formula (I) monoether-monoalcohol.
- Add the cycloaliphatic diol to form the disodium glycol intermediate under controlled temperature.
- React the intermediate with an alkylating agent such as diethyl sulfate to obtain the final diether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic sodium process represents a strategic opportunity to enhance margin stability and supply security. The most immediate impact is observed in the raw material cost structure, where the substitution of expensive, hazardous sodium hydride with commodity-grade metallic sodium drives down the bill of materials significantly. Since the catalyst is used in sub-stoichiometric amounts, its contribution to the overall cost is negligible, yet its effect on yield is profound. This cost reduction in pharmaceutical intermediate manufacturing is achieved without sacrificing quality; in fact, the improved selectivity reduces the load on purification units, lowering utility consumption and solvent waste disposal costs. The elimination of complex quenching procedures associated with hydride bases further simplifies the operational workflow, reducing labor hours and potential safety incidents.
- Cost Reduction in Manufacturing: The replacement of stoichiometric sodium hydride with catalytic amounts of inexpensive monoether-alcohols and metallic sodium fundamentally alters the cost basis of production. By avoiding the premium pricing associated with specialized hydride reagents and reducing the need for extensive downstream purification to remove mono-ether impurities, the overall cost of goods sold is substantially decreased. This efficiency gain allows for more competitive pricing strategies in the global market while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like metallic sodium and common solvents such as toluene or xylene mitigates the risk of supply disruptions often associated with specialized reagents. The robustness of the process means that production can be scaled up rapidly to meet surging demand without the lead time delays typical of sourcing high-purity specialty bases. This reliability ensures consistent delivery schedules for downstream clients, reinforcing the manufacturer's reputation as a dependable partner in the value chain.
- Scalability and Environmental Compliance: The controlled exothermic profile of this reaction makes it inherently safer for large-scale batch or continuous processing, reducing the engineering controls required for heat dissipation. Additionally, the higher selectivity results in less chemical waste and lower solvent usage per kilogram of product, aligning with increasingly strict environmental regulations and sustainability goals. This green chemistry advantage facilitates smoother regulatory approvals and enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this etherification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities.
Q: What is the primary advantage of using metallic sodium with a monoether-monoalcohol catalyst?
A: This combination prevents the encapsulation of sodium metal by the disodium salt product, ensuring complete deprotonation of the diol and significantly reducing mono-ether byproducts compared to traditional methods.
Q: Can this process replace expensive bases like sodium hydride?
A: Yes, the patent demonstrates that metallic sodium combined with a catalytic promoter effectively replaces costly sodium hydride, leading to substantial raw material cost reductions without compromising yield.
Q: How does this method improve safety during scale-up?
A: The catalytic system moderates the exothermic profile of the alkylation step, preventing dangerous temperature spikes and uncontrolled hydrogen evolution often seen in conventional direct alkylation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bis(ethoxymethyl)cyclohexane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of advanced catalytic processes like CN111479795A are fully realized at scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cycloaliphatic diol ethers meets the exacting standards required for pharmaceutical and high-performance polymer applications. Our commitment to technical excellence ensures that the theoretical yields and selectivity described in the patent are consistently achieved in our commercial reactors.
We invite you to collaborate with us to leverage these process efficiencies for your specific supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term growth and cost optimization strategies.
