Advanced Synthetic Route for Encetavir Intermediates Enabling Commercial Scale-Up
Advanced Synthetic Route for Encetavir Intermediates Enabling Commercial Scale-Up
The global pharmaceutical landscape has witnessed a paradigm shift with the emergence of potent 3CL protease inhibitors as critical therapeutics for managing viral outbreaks. Among these, Encetavir (S-217622) stands out as a promising oral antiviral agent, necessitating robust and scalable manufacturing processes to meet potential market demands. Patent CN115109041A discloses a groundbreaking synthetic methodology that addresses the inherent bottlenecks of previous generation routes, specifically focusing on the construction of the hexahydrotriazine-2,4-dione core scaffold. This technical insight report analyzes the proprietary chemistry detailed in the patent, highlighting its potential to redefine the supply chain dynamics for high-purity antiviral intermediates. By leveraging a direct cyclization strategy mediated by carbonyldiimidazole (CDI), the disclosed method circumvents the need for cumbersome protecting group manipulations, thereby streamlining the path from raw materials to the final active pharmaceutical ingredient (API). For industry stakeholders, understanding these mechanistic nuances is paramount for evaluating the feasibility of integrating this technology into existing production lines.
The Limitations of Conventional Methods vs. The Novel Approach
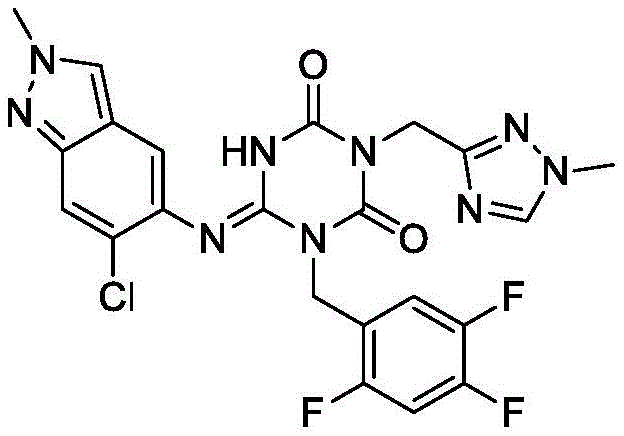
The Limitations of Conventional Methods
Prior to the innovations described in CN115109041A, the synthesis of Encetavir relied heavily on methodologies reported in literature, such as those originating from Shionogi & Co., Ltd. These conventional routes typically involve a multi-step sequence commencing with the alkylation of a tert-butyl protected triazine derivative, followed by a harsh acid hydrolysis to remove the tert-butyl group. This approach presents significant drawbacks for industrial application, primarily due to the generation of hazardous waste streams and the instability of intermediates under acidic conditions. Furthermore, the introduction of the methylthio group in early stages leads to the formation of toxic and odorous byproducts during the final condensation, complicating the refining process and increasing environmental compliance costs. The cumulative effect of these inefficiencies results in lower overall yields and extended production cycles, creating vulnerabilities in the supply chain for pharmaceutical intermediates.
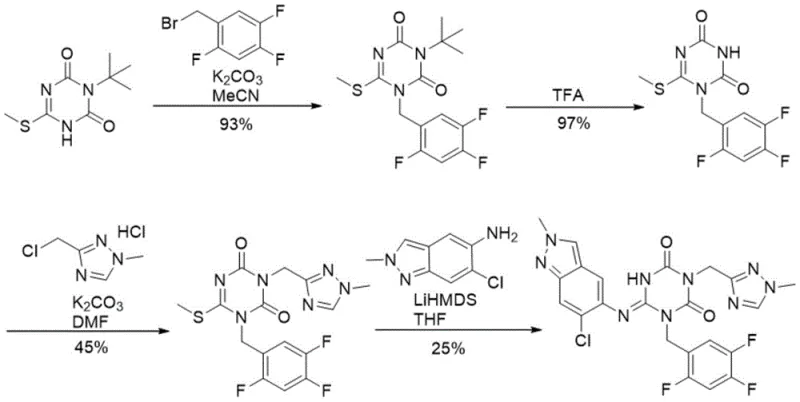
The Novel Approach
In stark contrast to the legacy methods, the novel approach outlined in the patent utilizes a convergent strategy that constructs the triazine core directly from unprotected precursors. By reacting 1H-pyrazole-1-formamidine hydrochloride with (1-methyl-1H-1,2,4-triazol-3-yl)methanamine hydrochloride in the presence of CDI, the process achieves rapid cyclization without the need for temporary protecting groups. This fundamental shift not only reduces the total number of unit operations but also significantly enhances the atom economy of the synthesis. The subsequent alkylation with 2,4,5-trifluorobenzyl halides proceeds under mild basic conditions, preserving the integrity of the sensitive heterocyclic rings. This streamlined pathway exemplifies how modern process chemistry can achieve cost reduction in API manufacturing by eliminating unnecessary synthetic steps and minimizing waste generation, ultimately delivering a more sustainable and economically viable production model.
Mechanistic Insights into CDI-Mediated Cyclization and N-Alkylation
The cornerstone of this improved synthetic route is the CDI-mediated cyclization reaction, which serves as the gateway to forming the 1,3,5-triazine-2,4(1H,3H)-dione skeleton. Mechanistically, carbonyldiimidazole acts as a dehydrating coupling agent, activating the amidine functionality of the pyrazole precursor to facilitate nucleophilic attack by the amine group of the triazole derivative. This reaction is conducted in polar aprotic solvents such as DMF or THF, utilizing organic bases like DIPEA or N-methylmorpholine to scavenge the generated imidazole byproduct. The choice of base and solvent is critical; for instance, the patent data indicates that switching from DMF to THF can improve the isolated yield of the intermediate compound 3 from 74.7% to 79.5%. This level of optimization demonstrates a deep understanding of solvation effects on reaction kinetics, ensuring that the formation of the desired cyclic urea structure is favored over potential oligomerization or hydrolysis side reactions.
Following the core construction, the N-alkylation step introduces the crucial 2,4,5-trifluorobenzyl moiety, which is essential for the biological activity of the final inhibitor. This transformation involves the displacement of a halide leaving group (chlorine or bromine) from the benzyl halide by the nitrogen atom of the triazine ring. The patent specifies the use of inorganic bases like potassium carbonate or cesium carbonate, often supplemented with catalytic amounts of potassium iodide to enhance the leaving group ability via the Finkelstein reaction mechanism. This step is remarkably robust, tolerating a wide range of reaction temperatures from 50°C to 90°C without significant degradation. The ability to use simple inorganic salts instead of expensive organometallic bases further underscores the economic advantages of this route, making it an attractive option for a reliable pharmaceutical intermediates supplier aiming to optimize production costs while maintaining rigorous quality standards.
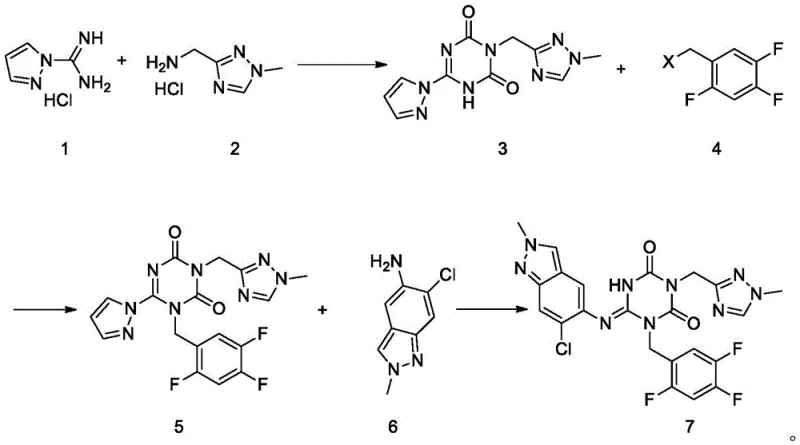
How to Synthesize Encetavir Intermediate Efficiently
The synthesis of the key intermediate Compound 5 represents a pivotal stage in the manufacturing of Encetavir, bridging the gap between simple heterocyclic building blocks and the complex final drug substance. The process described in the patent offers a reproducible protocol that balances reaction efficiency with ease of workup, utilizing crystallization as the primary purification method rather than resource-intensive chromatography. This operational simplicity is a key factor in determining the commercial viability of any chemical process, particularly when scaling from laboratory benchtop to pilot plant reactors. The following guide outlines the critical operational parameters derived from the patent examples, providing a roadmap for technical teams to replicate these high-yield results in their own facilities.
- Cyclize 1H-pyrazole-1-formamidine and (1-methyl-1H-1,2,4-triazol-3-yl)methanamine using carbonyldiimidazole (CDI) in the presence of a base like DIPEA or N-methylmorpholine to form the triazine-dione core.
- Perform N-alkylation of the intermediate with 2,4,5-trifluorobenzyl halide using potassium carbonate or cesium carbonate in polar aprotic solvents such as DMF or acetonitrile.
- Execute the final condensation reaction between the alkylated intermediate and 6-chloro-2-methyl-2H-indazol-5-amine in an alcoholic solvent like n-butanol at elevated temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond mere chemical elegance. The elimination of the tert-butyl protection and subsequent acid deprotection steps significantly reduces the consumption of raw materials and the volume of hazardous waste requiring disposal. This simplification of the process flow directly correlates to a reduction in the overall cost of goods sold (COGS), allowing for more competitive pricing structures in the global marketplace. Moreover, the avoidance of toxic methylthio-containing intermediates mitigates the risks associated with handling hazardous substances, thereby lowering the barrier for regulatory approval and reducing the complexity of environmental health and safety (EHS) compliance protocols.
- Cost Reduction in Manufacturing: The streamlined synthesis eliminates multiple unit operations, specifically the protection and deprotection sequences that traditionally consume significant time and resources. By removing the need for strong acids and specialized reagents required for tert-butyl removal, the process drastically lowers utility consumption and waste treatment costs. Furthermore, the high yields achieved in each step, particularly the final condensation which exceeds 90%, minimize the loss of valuable starting materials, ensuring that the maximum amount of input is converted into saleable product. This efficiency creates a substantial margin for cost optimization, enabling manufacturers to offer more competitive pricing without compromising on quality or profitability.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as pyrazole formamidines and trifluorobenzyl halides, reduces the risk of supply disruptions caused by the scarcity of exotic reagents. The robustness of the reaction conditions, which tolerate a broad range of temperatures and solvents, ensures consistent production output even when facing minor variations in raw material quality or equipment performance. This resilience is critical for maintaining continuous supply lines to downstream API manufacturers, preventing costly production stoppages and ensuring that patient demand can be met without delay. The simplified purification via crystallization further accelerates the turnover time for each batch, enhancing the overall agility of the supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard chemical engineering unit operations such as stirred tank reactors and filtration units that are ubiquitous in the fine chemical industry. The absence of chromatographic purification steps removes a major bottleneck that often limits batch sizes in traditional pharmaceutical synthesis. Additionally, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, positioning manufacturers who adopt this technology as leaders in sustainable chemistry. This proactive approach to environmental stewardship not only reduces liability but also enhances the corporate reputation of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and claims presented in patent CN115109041A, providing a factual basis for decision-making. Understanding these details is essential for R&D teams evaluating the transferability of the technology and for commercial teams assessing the long-term viability of the supply partnership.
Q: How does the new synthetic route improve yield compared to the prior art?
A: The novel route eliminates the low-yielding deprotection steps and toxic methylthio byproduct formation associated with the conventional Shionogi method, achieving isolated yields of over 90% in the final condensation step.
Q: What are the key advantages for supply chain stability?
A: By utilizing readily available starting materials and avoiding complex protection-deprotection sequences, the process reduces lead times and minimizes the reliance on specialized reagents, ensuring consistent supply continuity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method relies on robust crystallization for purification rather than column chromatography, and operates under moderate temperatures, making it highly scalable for multi-kilogram to ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Encetavir Intermediate Supplier
As the demand for effective antiviral therapies continues to evolve, the ability to rapidly scale the production of high-quality intermediates becomes a decisive competitive advantage. NINGBO INNO PHARMCHEM leverages its extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced synthetic route to fruition. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Encetavir intermediate delivered meets the exacting standards required for clinical and commercial applications. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to process excellence guarantees a reliable flow of materials to support your drug development timelines.
We invite global partners to collaborate with us to fully realize the potential of this innovative manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized synthesis of Encetavir intermediates can enhance your supply chain resilience and drive down your overall manufacturing costs. Together, we can accelerate the availability of life-saving medications to patients worldwide.
