Advanced Synthetic Route For S-3-Morpholinecarboxylic Acid Ensuring Commercial Scalability
Introduction to Novel Synthetic Methodology
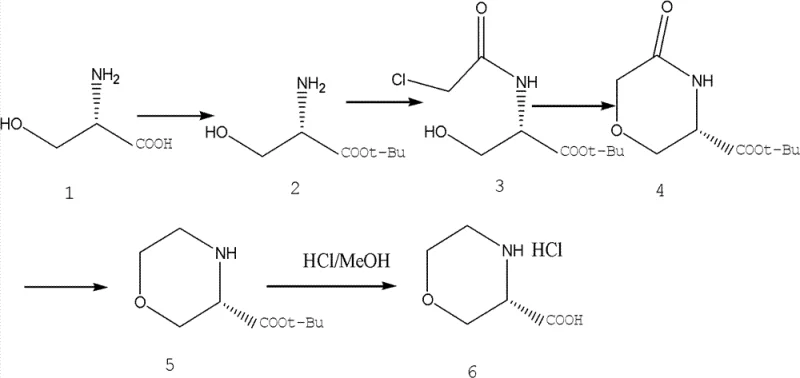
The pharmaceutical industry continuously demands more efficient, cost-effective, and environmentally benign pathways for the production of chiral building blocks. A pivotal advancement in this domain is documented in patent CN102617503B, which discloses a novel synthetic method for (S)-3-morpholinyl carboxylic acid. This compound serves as a critical intermediate in the development of various bioactive molecules, including potential therapeutic agents targeting metabolic and neurological disorders. The disclosed technology represents a paradigm shift from traditional methodologies that often rely on expensive, hard-to-source starting materials and harsh reaction conditions. By leveraging L-Serine as the foundational chiral pool material, this innovation addresses key pain points in modern process chemistry, specifically focusing on atom economy, stereochemical integrity, and operational simplicity. The methodology outlined in the patent provides a robust framework for manufacturers seeking to optimize their supply chains for high-value morpholine derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of morpholine-3-carboxylic acid derivatives has been plagued by significant technical and economic hurdles. Conventional routes frequently employ hydroxyl-containing raw materials reacting with unsaturated ketones under catalytic conditions. These legacy processes suffer from severe drawbacks, including the necessity for rigorous temperature control and the difficulty in selecting appropriate catalysts that do not promote side reactions. The reliance on unsaturated ketones often introduces impurities that are difficult to separate, leading to lower overall yields and compromised optical purity. Furthermore, the acylation and guanidine ring formation steps in traditional methods typically require elevated temperatures and extended reaction times, which not only increase energy consumption but also elevate the risk of racemization. These factors collectively render conventional methods unsuitable for large-scale industrial production, creating bottlenecks in the supply of high-purity intermediates for downstream drug synthesis.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes L-Serine, a readily available and inexpensive amino acid, as the primary feedstock. This strategic choice fundamentally alters the economic and technical landscape of the synthesis. The process initiates with the protection of the serine amino group, followed by a highly efficient acylation with chloroacetyl chloride. The subsequent cyclization step, mediated by sodium ethoxide in toluene, proceeds with exceptional selectivity to form the morpholinone core. Perhaps most notably, the reduction of the carbonyl group is achieved using a gentle aluminum chloride and sodium borohydride system, avoiding the need for high-pressure hydrogenation equipment. This sequence of reactions operates under mild conditions, often requiring only ice-water or warm water baths, thereby drastically simplifying the engineering requirements for production facilities. The result is a streamlined process that minimizes waste generation and maximizes throughput, making it an ideal candidate for commercial scale-up.
Mechanistic Insights into AlCl3-Catalyzed Reduction and Cyclization
The core of this synthetic innovation lies in the precise control of stereochemistry and functional group transformations. The cyclization step involves an intramolecular nucleophilic substitution where the hydroxyl group of the serine derivative attacks the chloroacetyl moiety. The use of sodium ethoxide in toluene facilitates this ring closure by generating the alkoxide nucleophile in situ, driving the equilibrium towards the formation of the six-membered morpholinone ring. This step is crucial as it establishes the rigid cyclic structure while preserving the chiral center derived from the natural L-Serine. The reaction conditions are optimized to prevent epimerization, ensuring that the final product retains the desired (S)-configuration with high optical purity. The high yields observed in this step, often exceeding 95%, underscore the efficiency of the base-mediated cyclization in a non-polar solvent environment.
Furthermore, the reduction mechanism employing aluminum chloride and sodium borohydride warrants detailed attention. In this system, aluminum chloride acts as a Lewis acid, coordinating with the carbonyl oxygen of the morpholinone intermediate to enhance its electrophilicity. Simultaneously, sodium borohydride serves as the hydride source. The interaction between these reagents generates a reactive alkoxy-aluminum-hydride species that delivers hydride to the carbonyl carbon with high chemoselectivity. This mild reduction protocol is superior to traditional catalytic hydrogenation as it avoids the potential for ring opening or over-reduction of other sensitive functional groups. The reaction proceeds smoothly at temperatures ranging from -10°C to 40°C, demonstrating remarkable tolerance and control. This mechanistic elegance ensures that the final deprotection step yields the target carboxylic acid with minimal impurity profiles, satisfying the stringent quality requirements of pharmaceutical applications.
How to Synthesize (S)-3-Morpholinyl Carboxylic Acid Efficiently
The implementation of this synthetic route requires careful attention to stoichiometry and temperature management to replicate the high yields reported in the patent literature. The process is divided into five distinct operational stages, beginning with the esterification of L-Serine and concluding with the acidic removal of the tert-butyl protecting group. Each step has been optimized to balance reaction kinetics with product stability, ensuring that intermediates can be isolated or carried forward with minimal degradation. Operators should note that the exothermic nature of the aluminum chloride addition requires controlled dosing to maintain thermal safety. The following guide outlines the critical parameters for executing this synthesis effectively, serving as a blueprint for process engineers aiming to adopt this technology.
- Protect L-Serine using tert-butyl acetate and perchloric acid catalyst to form L-Serine tert-butyl ester.
- Perform acylation with chloroacetyl chloride in dichloromethane to yield N-chloroacetyl-L-serine tert-butyl ester.
- Execute intramolecular cyclization using sodium ethoxide in toluene to form the morpholinone ring structure.
- Reduce the ketone moiety using aluminum chloride and sodium borohydride in methanol to obtain the morpholine ring.
- Remove the tert-butyl protecting group using hydrogen chloride in methanol to isolate the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Serine-based methodology offers transformative benefits that extend beyond mere technical feasibility. The shift away from specialized, expensive ketone precursors to commodity amino acids fundamentally reshapes the cost structure of the intermediate. L-Serine is produced on a massive global scale for the food and feed industries, ensuring a stable, abundant, and price-resilient supply chain. This abundance mitigates the risks associated with sourcing niche chemicals that are subject to volatile market fluctuations. Moreover, the simplification of the reaction conditions—eliminating the need for high-pressure reactors or cryogenic cooling systems—translates directly into reduced capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturing partners. The ability to run reactions in standard glass-lined or stainless steel reactors using common solvents like toluene and methanol further enhances the accessibility of this process for contract manufacturing organizations (CMOs).
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the drastic reduction in raw material costs. By replacing costly unsaturated ketones with L-Serine, the direct material cost per kilogram of the final product is significantly lowered. Additionally, the high yields achieved in the cyclization and deprotection steps minimize the loss of valuable chiral material, thereby improving the overall mass balance of the process. The elimination of transition metal catalysts, which often require expensive removal and recovery steps to meet regulatory limits, further contributes to cost savings. This streamlined approach allows for a more competitive pricing structure for the final API intermediate, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Unlike specialized catalysts or custom-synthesized starting materials that may have single-source suppliers, the reagents used in this protocol (chloroacetyl chloride, sodium ethoxide, sodium borohydride) are standard industrial chemicals with multiple qualified vendors globally. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to minor variations in utility supplies (such as cooling water temperature), ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for green chemistry initiatives. The process generates fewer by-products and utilizes solvents that are easier to recover and recycle compared to more complex organic media. The mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with corporate sustainability goals. The absence of heavy metals in the reduction step simplifies waste treatment protocols, lowering the cost and complexity of effluent management. These factors combined make the scale-up from pilot plant to multi-ton commercial production a straightforward engineering task, facilitating rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and application of (S)-3-morpholinyl carboxylic acid. These responses are grounded in the specific technical disclosures of the patent and practical experience with similar amino acid-derived transformations. Understanding these nuances is essential for R&D directors assessing the feasibility of integrating this intermediate into their own drug discovery pipelines. The following section addresses critical aspects of purity, scalability, and raw material selection.
Q: Why is L-Serine preferred over traditional ketone raw materials for this synthesis?
A: L-Serine is a naturally abundant, cost-effective amino acid that eliminates the need for expensive, unsaturated ketone precursors. Its use ensures milder reaction conditions, higher stereochemical purity, and significantly reduced raw material costs compared to conventional methods.
Q: How does the AlCl3/NaBH4 reduction system improve process safety?
A: The combination of aluminum chloride and sodium borohydride generates a mild reducing species in situ. This avoids the use of hazardous high-pressure hydrogenation or aggressive hydride reagents, allowing the reduction to proceed safely at near-ambient temperatures with excellent chemoselectivity.
Q: Is this synthetic route suitable for multi-ton commercial production?
A: Yes, the process utilizes common industrial solvents like toluene and methanol and avoids cryogenic conditions or exotic catalysts. The high yields reported in each step (often exceeding 90% in cyclization and deprotection) indicate a robust pathway amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Morpholinyl Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the synthetic route described in CN102617503B and possesses the expertise to execute this chemistry with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with agility and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of the (S)-enantiomer. We are committed to delivering products that not only meet but exceed the expectations of global pharmaceutical partners.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this Serine-based route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your partner in transforming innovative chemistry into commercial success.
