Optimizing Nateglinide Production: A Technical Breakthrough in Catalytic Synthesis and Crystallization
Optimizing Nateglinide Production: A Technical Breakthrough in Catalytic Synthesis and Crystallization
The global demand for effective antidiabetic medications continues to surge, driving the need for robust and scalable manufacturing processes for key active pharmaceutical ingredients like Nateglinide. Patent CN103159640A introduces a refined synthetic methodology that addresses critical bottlenecks in traditional production lines, specifically focusing on the efficiency of catalytic oxidation and stereochemical control. This technical insight report analyzes the proprietary route starting from 4-isopropyltoluene, highlighting its potential to serve as a benchmark for reliable nateglinide supplier operations seeking to enhance process integrity. By leveraging molecular oxygen and transition metal catalysis, the described method offers a pathway to high-purity intermediates while mitigating the environmental burden associated with stoichiometric oxidants. For R&D directors and procurement strategists, understanding the nuances of this catalytic cycle is essential for evaluating long-term supply chain viability and cost structures in the competitive diabetes care market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Nateglinide often suffer from excessive step counts, reliance on expensive chiral starting materials, or the use of hazardous reagents that complicate waste management and increase operational expenditures. Many legacy processes involve early-stage resolution of racemates, which inherently caps the maximum theoretical yield at fifty percent unless dynamic kinetic resolution is employed, a technique that adds significant complexity and cost. Furthermore, conventional oxidation methods frequently utilize strong mineral acids or heavy metal oxidants like permanganates, which generate substantial quantities of saline wastewater and require extensive downstream purification to meet stringent heavy metal limits for pharmaceutical products. These inefficiencies not only inflate the cost reduction in API manufacturing but also introduce variability in batch-to-batch consistency, posing risks to supply chain continuity for large-scale commercial partners.
The Novel Approach
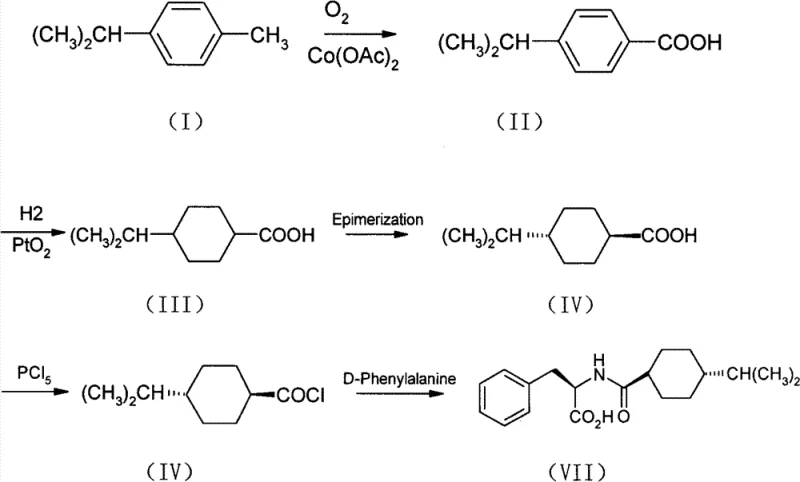
The innovative approach detailed in the patent data circumvents these issues by utilizing 4-isopropyltoluene, a readily available and cost-effective petrochemical derivative, as the primary feedstock. This strategy employs a cobalt-catalyzed aerobic oxidation to convert the methyl group directly into a carboxylic acid, a transformation that is both atom-economical and environmentally benign compared to stoichiometric alternatives. Subsequent steps involve a highly selective catalytic hydrogenation using platinum oxide, followed by a thermodynamic epimerization that ensures the formation of the crucial trans-isomer with high fidelity. This streamlined sequence minimizes unit operations and eliminates the need for early-stage chiral resolution, thereby significantly improving overall process mass intensity. The final crystallization step is engineered to reliably produce the bioactive H-type polymorph, ensuring that the final product meets all pharmacopeial standards without requiring complex post-synthetic modifications.
Mechanistic Insights into Cobalt-Catalyzed Oxidation and Platinum Hydrogenation
The cornerstone of this synthesis is the initial oxidation of 4-isopropyltoluene to 4-isopropylbenzoic acid, which proceeds via a free-radical chain mechanism facilitated by the cobalt acetate catalyst. In this cycle, the cobalt species acts as an initiator and propagator, abstracting hydrogen atoms from the benzylic position to form stable radical intermediates that react with molecular oxygen. The reaction conditions, typically maintained between 60°C and 100°C with a controlled oxygen flow rate, are critical for balancing reaction kinetics against over-oxidation or ring degradation. This catalytic system allows for the continuous regeneration of the active cobalt species, ensuring sustained turnover numbers and minimizing catalyst loading requirements. The selectivity of this oxidation is paramount, as it sets the stage for the subsequent hydrogenation by preserving the integrity of the isopropyl group while functionalizing the aromatic ring.
Following oxidation, the aromatic ring is saturated using a platinum oxide catalyst under hydrogen pressure, a step that requires precise control to avoid over-reduction or hydrogenolysis of the carboxylic acid functionality. The resulting cis-isomer is then subjected to base-catalyzed epimerization, where the thermodynamic stability of the trans-diequatorial conformation drives the equilibrium toward the desired stereoisomer. This stereochemical precision is vital because the biological activity of Nateglinide is strictly dependent on the spatial arrangement of the cyclohexyl ring relative to the amide linkage. The final coupling with D-phenylalanine preserves this chirality, and the subsequent crystallization from acetone-water mixtures exploits solubility differences to isolate the H-type crystal lattice. This rigorous control over solid-state chemistry ensures that the final API possesses the necessary dissolution profile and stability for tablet formulation.
How to Synthesize Nateglinide Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to maximize yield and purity while ensuring safety. The process begins with the aerobic oxidation in acetic acid, followed by high-pressure hydrogenation and a thermal epimerization step that demands careful temperature monitoring. The conversion to the acid chloride must be performed under anhydrous conditions to prevent hydrolysis, and the final amide coupling requires pH control to optimize nucleophilic attack by the amino acid. For a detailed breakdown of the standardized operating procedures, including specific molar ratios, solvent volumes, and workup protocols, please refer to the technical guide below.
- Oxidize 4-isopropyltoluene to 4-isopropylbenzoic acid using oxygen and cobalt acetate catalyst at controlled temperatures.
- Perform catalytic hydrogenation using PtO2 to form 4-isopropylcyclohexylcarboxylic acid, followed by epimerization to the trans-isomer.
- Convert the trans-acid to acid chloride using phosphorus pentachloride, then couple with D-phenylalanine to form crude Nateglinide.
- Recrystallize the crude product in acetone-water mixture to transform the crystal lattice into the physiologically active H-type polymorph.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical industry. By shifting the starting material to 4-isopropyltoluene, manufacturers can leverage a commodity chemical with a stable global supply, reducing exposure to price volatility associated with specialized chiral building blocks. The elimination of stoichiometric oxidants and the use of recoverable transition metal catalysts contribute to a greener process profile, which simplifies regulatory filings and reduces waste disposal costs. Furthermore, the robustness of the crystallization step ensures high recovery rates of the active polymorph, minimizing material loss and enhancing overall process economics. These factors combine to create a manufacturing framework that is both cost-effective and resilient against market fluctuations.
- Cost Reduction in Manufacturing: The substitution of expensive chiral precursors with achiral 4-isopropyltoluene fundamentally alters the cost structure of the synthesis, removing the premium associated with enantiopure starting materials. Additionally, the catalytic nature of the oxidation and hydrogenation steps means that reagent consumption is minimized, leading to substantial savings in raw material expenditures over large production campaigns. The simplified workup procedures, which avoid complex extractions or chromatographic purifications, further reduce labor and solvent costs, making the process highly attractive for commercial scale-up of complex antidiabetic agents.
- Enhanced Supply Chain Reliability: Utilizing widely available petrochemical derivatives as feedstocks ensures that production is not bottlenecked by the limited capacity of niche intermediate suppliers. The robustness of the catalytic systems described allows for flexible manufacturing schedules, as the reactions are tolerant to minor variations in input quality without compromising final product specifications. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond swiftly to changes in market demand and secure long-term contracts with major pharmaceutical companies.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations for oxidation and hydrogenation that are common in fine chemical plants. The avoidance of hazardous reagents and the generation of less toxic byproducts align with increasingly stringent environmental regulations, reducing the risk of production shutdowns due to compliance issues. This environmental stewardship not only protects the corporate reputation but also lowers the total cost of ownership by minimizing the need for specialized waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Nateglinide synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic benefits for potential partners. Understanding these details is key to making informed decisions about technology transfer and supply agreements.
Q: What is the key advantage of the catalytic oxidation step in this Nateglinide synthesis?
A: The use of molecular oxygen with a cobalt acetate catalyst allows for a direct and atom-economical oxidation of the methyl group on 4-isopropyltoluene, avoiding harsh stoichiometric oxidants and reducing waste generation significantly.
Q: How is the stereochemistry controlled to ensure the correct trans-isomer formation?
A: Stereocontrol is achieved through a specific epimerization step following hydrogenation. The process utilizes thermodynamic control under basic conditions (KOH) at elevated temperatures to favor the more stable trans-4-isopropylcyclohexylcarboxylic acid configuration.
Q: Why is the crystal transformation to H-type Nateglinide critical?
A: The H-type polymorph possesses the requisite physiological activity and stability for pharmaceutical formulation. The patented recrystallization method using acetone-water ensures high purity and consistent bioavailability, which is essential for regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nateglinide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of process efficiency and product quality in the production of life-saving medications like Nateglinide. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of API or intermediate we deliver meets the highest international standards. Our expertise in catalytic chemistry and crystallization engineering allows us to optimize yields and minimize impurities, providing a distinct competitive advantage in the marketplace.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the diabetes therapeutic sector.
