Scaling High-Purity 2,3,4,6-Tetra-O-Benzyl-D-Glucose Production via Continuous Flow Technology
The pharmaceutical industry is constantly seeking robust manufacturing processes that can deliver high-purity intermediates with consistent quality at scale. A significant breakthrough in this domain is documented in Chinese patent CN108101946B, which outlines a novel continuous synthesis method for 2,3,4,6-tetra-O-benzyl-D-glucose. This compound is a pivotal chiral intermediate used extensively in the synthesis of anti-diabetic drugs like Voglibose and Miglitol. The patent addresses long-standing challenges in the traditional batch production of this molecule, specifically the low yields and reproducibility issues encountered during scale-up. By transitioning from discontinuous batch reactors to a sophisticated continuous flow system, the technology achieves a dramatic enhancement in conversion rates and product isolation efficiency. This report analyzes the technical merits of this innovation and its implications for global supply chains, offering a pathway for reliable pharmaceutical intermediates supplier partnerships that prioritize both quality and economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,4,6-tetra-O-benzyl-D-glucose via the hydrolysis of 2,3,4,6-tetrabenzyl glucoside has been plagued by significant inefficiencies when attempted in traditional batch reactors. The core issue lies in the instability of the substrate and the aggressive nature of the acid-catalyzed reaction environment. In batch processes, maintaining uniform temperature and mixing throughout the reaction vessel becomes increasingly difficult as volume increases, leading to localized hot spots or concentration gradients. These inconsistencies cause the sensitive product to degrade or undergo side reactions before the reaction is complete. Consequently, industrial reports often cite separation yields hovering between 30% and 40%, a stark contrast to the promising 50-60% yields observed in small-scale laboratory trials. This discrepancy, known as the amplification effect, creates a bottleneck for manufacturers, resulting in high production costs due to wasted raw materials and complex downstream purification requirements to remove impurities generated by incomplete conversion.
The Novel Approach
The methodology presented in patent CN108101946B revolutionizes this landscape by implementing a continuous synthesis strategy that decouples reaction efficiency from vessel size. Instead of a large tank, the reaction occurs within a continuous coiled tube reactor where reagents are pumped through a heated zone with precise control over residence time. This setup ensures that every molecule of the substrate experiences identical reaction conditions, eliminating the variability inherent in batch processing. Furthermore, the system integrates immediate continuous purification, where the product stream is directly fed into a crystallization unit. This rapid removal of the product from the acidic reaction mixture prevents the hydrolysis product from sitting in the corrosive environment, thereby halting further degradation. The result is a process that not only replicates laboratory success on an industrial scale but surpasses it, delivering high-purity material with significantly improved throughput and reduced waste generation.
Mechanistic Insights into Acid-Catalyzed Hydrolysis in Flow
The chemical transformation at the heart of this process is the acid-catalyzed cleavage of the glycosidic bond in 2,3,4,6-tetrabenzyl glucoside. As illustrated in the reaction scheme below, the process involves the protonation of the methoxy group at the anomeric center, followed by nucleophilic attack by water to release methanol and form the hemiacetal structure of the target glucose derivative. In a static batch environment, the accumulation of methanol and the prolonged exposure to strong Lewis acids like sulfuric acid can drive equilibrium backward or promote decomposition. However, the continuous flow architecture fundamentally alters the kinetic profile of this reaction.
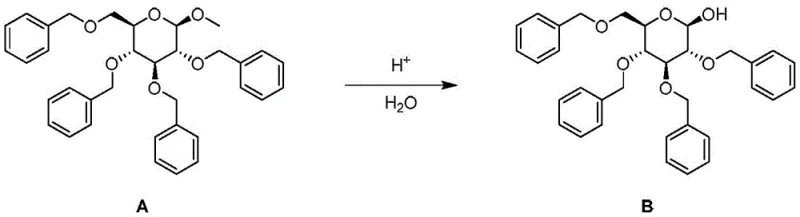
The continuous movement of the reaction mixture ensures that the concentration of reactants remains optimal throughout the reactor length, while the products are swiftly transported away from the reaction zone. This dynamic environment minimizes the contact time between the newly formed glucose derivative and the acid catalyst, effectively 'freezing' the reaction at the point of maximum yield. Additionally, the enhanced heat transfer capabilities of the coiled tube reactor allow for operation at elevated temperatures (70-150°C) without the risk of thermal runaway, accelerating the reaction rate significantly. This precise control over the mechanistic pathway reduces the formation of by-products, simplifying the impurity profile and making the subsequent crystallization step far more effective at isolating the desired stereoisomer.
How to Synthesize 2,3,4,6-Tetra-O-Benzyl-D-Glucose Efficiently
Implementing this continuous synthesis route requires careful calibration of flow rates and thermal parameters to match the specific kinetics of the hydrolysis reaction. The patent details a robust protocol where two distinct liquid streams are prepared: one containing the dissolved glucoside substrate and another containing the aqueous acid catalyst. These streams are merged and passed through a heated coil, allowing the chemistry to proceed under steady-state conditions. The efficiency of this method relies heavily on the synchronization of the reaction dwell time with the crystallization cooling rate. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Prepare two separate feed streams: one containing the substrate 2,3,4,6-tetrabenzyl glucoside in a suitable organic solvent, and another containing the Lewis acid catalyst mixed with water and solvent.
- Pump both streams continuously into a heated coiled tube reactor maintained at 70-150°C, ensuring a residence time between 20 to 100 minutes for complete acid-catalyzed hydrolysis.
- Direct the effluent immediately into a continuous crystallization unit cooled to 10-20°C to precipitate the product, followed by continuous filtration and drying to obtain high-purity solids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this continuous manufacturing technology represents a strategic opportunity to optimize costs and secure supply continuity. The traditional bottlenecks of low yield and unpredictable scale-up are effectively removed, translating into a more stable and cost-effective sourcing model for this critical pharmaceutical intermediate. The ability to run the process continuously means that production capacity can be scaled linearly by extending run times or numbering up reactors, rather than investing in massive new batch vessels. This flexibility allows suppliers to respond more agilely to market demand fluctuations without compromising on quality or lead times.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic improvement in material efficiency. By elevating yields from the typical 30-40% range to over 80%, the consumption of expensive starting materials like tetrabenzyl glucoside is nearly halved per unit of output. Furthermore, the continuous purification step reduces the volume of solvents and reagents required for workup, as the product precipitates directly from the reaction stream. This elimination of extensive extraction and washing procedures lowers utility costs and reduces the burden on waste treatment facilities, contributing to substantial overall cost savings in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reproducibility of the continuous process. Unlike batch methods where each run can vary slightly, continuous flow systems operate at a steady state, ensuring that every kilogram produced meets the same stringent quality specifications. This consistency reduces the risk of batch failures and the need for re-processing, which often causes delays in delivery. The simplified process flow also shortens the overall production cycle time, enabling faster turnaround from order to shipment and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology offers a cleaner production footprint. The reduced solvent usage and higher atom economy mean less hazardous waste is generated, simplifying compliance with increasingly strict environmental regulations. The compact nature of continuous flow equipment also allows for production to be situated closer to key markets or integrated into existing facilities with limited space. This scalability ensures that as demand for downstream drugs like Voglibose grows, the supply of this key precursor can be expanded seamlessly without the long lead times associated with constructing new large-scale batch plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of 2,3,4,6-tetra-O-benzyl-D-glucose. These answers are derived directly from the experimental data and technical specifications outlined in the relevant patent literature, providing clarity on the feasibility and benefits of adopting this advanced manufacturing approach for your supply chain.
Q: How does continuous flow synthesis improve the yield of 2,3,4,6-tetra-O-benzyl-D-glucose compared to batch processing?
A: Traditional batch methods often suffer from poor heat transfer and mixing at scale, leading to yields around 30-40%. The continuous flow method described in patent CN108101946B optimizes reaction parameters like residence time and temperature control, consistently achieving yields over 80% by preventing product degradation in the acidic medium.
Q: What are the primary applications of this specific glucose intermediate?
A: This compound serves as a critical chiral building block for synthesizing anti-diabetic agents such as Voglibose and Miglitol. It is also utilized in the preparation of antiviral compounds and other glycoside derivatives requiring specific stereochemical configurations.
Q: Is this continuous process scalable for industrial manufacturing?
A: Yes, the patent explicitly demonstrates successful scale-up from laboratory gram-scale to kilogram-scale production without loss of yield or purity. The use of modular continuous reactors eliminates the 'amplification effect' typically seen when scaling batch reactions, ensuring consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4,6-Tetra-O-Benzyl-D-Glucose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. Our team of expert chemists has deeply analyzed emerging technologies like the continuous flow synthesis described in CN108101946B to ensure our manufacturing capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,3,4,6-tetra-O-benzyl-D-glucose we supply supports your downstream synthesis with minimal impurity interference.
We invite you to collaborate with us to leverage these advanced manufacturing efficiencies for your projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage you to reach out today to discuss your requirements,索取 specific COA data, and review our route feasibility assessments to ensure a seamless integration of our materials into your production pipeline.
