Advanced Visible-Light Mediated Synthesis of Polysubstituted Naphthols for Pharmaceutical Intermediates
Advanced Visible-Light Mediated Synthesis of Polysubstituted Naphthols for Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more sustainable methodologies, a transition vividly exemplified by the innovations detailed in Chinese Patent CN103467282A. This pivotal intellectual property introduces a groundbreaking synthesis method for polysubstituted naphthols and their derivatives, leveraging the power of visible light photoredox catalysis. Unlike conventional thermal processes that often demand harsh conditions and stoichiometric oxidants, this novel approach utilizes a photosensitive metal iridium complex, specifically Ir(ppy)3, to drive an intermolecular single electron transfer (SET) radical cascade reaction. The significance of this technology extends far beyond academic curiosity; it addresses critical pain points in the production of high-value pharmaceutical intermediates and electronic materials. By enabling the coupling of alkynes and 2-bromo-1,3-dicarbonyl compounds under mild, room-temperature conditions, this patent offers a robust pathway for constructing complex naphthalene scaffolds found in bioactive lignans like Neojusticin and Justicidin B. For industry leaders seeking a reliable polysubstituted naphthols supplier, understanding the mechanistic elegance and operational simplicity of this patent is essential for securing a competitive edge in the global supply chain.
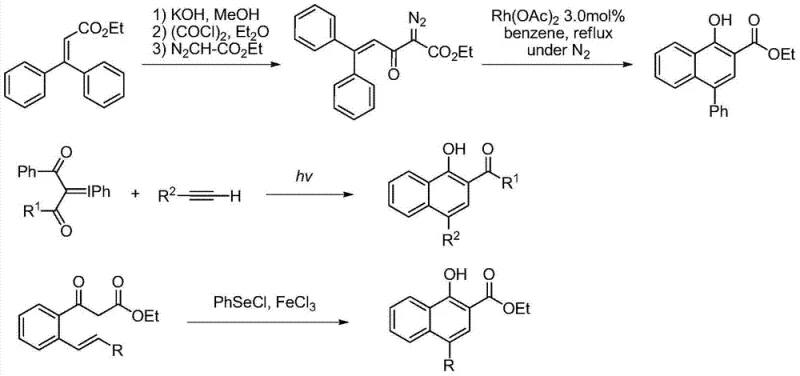
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted naphthol cores has been fraught with significant technical and safety challenges that hinder large-scale commercial application. Traditional synthetic routes often rely on hazardous reagents and extreme reaction conditions that pose substantial risks to both personnel and equipment. For instance, earlier methodologies frequently employed rhodium-catalyzed carbon-hydrogen insertion reactions involving azo-compounds, which necessitate the handling of unstable and potentially explosive diazo precursors. Furthermore, other established pathways, such as the photochemical reaction of 1,3-dicarbapentaborane iodobenzene ylides with terminal alkynes, require high-energy ultraviolet (UV) irradiation sources. The use of UV light not only increases energy consumption but also introduces safety hazards related to radiation exposure and the potential for uncontrolled side reactions. Additionally, methods utilizing selenide and iron trichloride for intramolecular rearrangement, while effective, suffer from poor atom economy due to the requirement for stoichiometric amounts of toxic additives. These legacy processes generate substantial chemical waste, complicating downstream purification and increasing the environmental burden, thereby reducing the overall prospects for commercial viability in a regulated manufacturing environment.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN103467282A represents a leap forward in process efficiency and sustainability. This novel approach harnesses the unique redox properties of excited-state iridium complexes to facilitate a redox-neutral radical cascade reaction under visible light irradiation. By utilizing common white LED lamps as the energy source, the reaction proceeds smoothly at room temperature, eliminating the need for external heating or dangerous UV sources. The core innovation lies in the ability of the photosensitizer to undergo single electron transfer with the substrates, generating reactive radical intermediates that spontaneously cyclize to form the desired naphthol structure. This strategy completely avoids the use of equivalent oxidants, which are typically required in traditional oxidative cyclizations and contribute heavily to waste generation. The result is a process with high atom economy and exceptional environmental friendliness, making it ideally suited for the cost reduction in pharmaceutical intermediates manufacturing. The mild conditions also ensure better functional group tolerance, allowing for the synthesis of diverse derivatives without the degradation issues often seen in harsher thermal protocols.
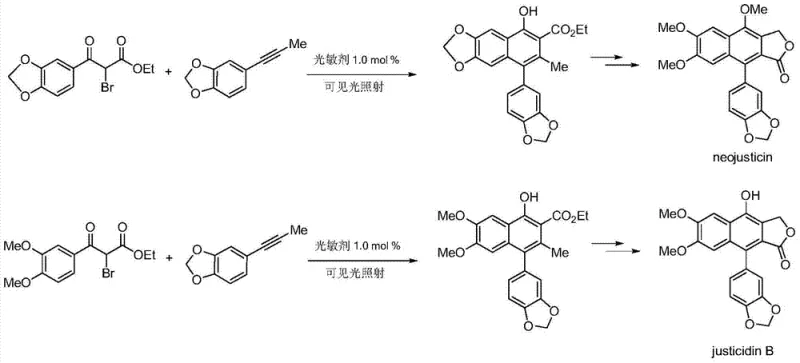
Mechanistic Insights into Ir-Catalyzed Visible Light Radical Cascade
To fully appreciate the technical superiority of this synthesis route, one must delve into the intricate mechanistic details of the iridium-catalyzed photoredox cycle. The reaction initiates when the ground-state Ir(III) photocatalyst absorbs photons from the visible light source, promoting it to a long-lived excited state, denoted as Ir(III)*. This excited species possesses potent single-electron oxidizing and reducing capabilities, allowing it to interact with the 2-bromo-1,3-dicarbonyl substrate. Through a reductive quenching pathway, the excited catalyst facilitates the homolytic cleavage of the carbon-bromine bond, generating a nucleophilic carbon-centered radical and an Ir(IV) species. This radical intermediate then adds across the triple bond of the alkyne substrate, initiating a cascade of intramolecular cyclization events. The resulting cyclic radical intermediate subsequently undergoes oxidation and deprotonation steps to restore aromaticity, ultimately yielding the polysubstituted naphthol product while regenerating the ground-state Ir(III) catalyst to close the cycle. This elegant mechanism ensures that the catalyst is turned over continuously without being consumed, maintaining high efficiency throughout the reaction duration.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. The specificity of the radical addition and the rapid nature of the cascade cyclization minimize the formation of oligomeric byproducts or regioisomers that often plague traditional electrophilic aromatic substitutions. Furthermore, because the reaction is redox-neutral and does not require external oxidants like peroxides or metal salts, the impurity profile is significantly cleaner, lacking the metal residues or oxidation byproducts associated with older methods. This inherent purity is critical for R&D directors focusing on the synthesis of complex API intermediates where downstream purification can be a bottleneck. The ability to tolerate various substituents on both the alkyne and the dicarbonyl components—ranging from electron-donating methoxy groups to electron-withdrawing trifluoromethyl groups—demonstrates the robustness of this catalytic system. Such versatility ensures that the process can be adapted to produce a wide array of high-purity OLED material precursors or pharmaceutical building blocks with consistent quality.
How to Synthesize Polysubstituted Naphthols Efficiently
The practical implementation of this photoredox methodology is straightforward and amenable to standard laboratory and pilot-plant setups. The general procedure involves combining the alkyne substrate and the 2-bromo-1,3-dicarbonyl compound in a polar aprotic solvent such as DMF, with a catalytic amount of Ir(ppy)3. The reaction vessel is purged with nitrogen to exclude oxygen, which could otherwise quench the radical intermediates or deactivate the catalyst. Upon irradiation with white LEDs, the solution undergoes a visible color change or conversion as monitored by thin-layer chromatography (TLC). Following the reaction period, typically around 12 hours, the workup is simple: the mixture is quenched with water, extracted with an organic solvent like diethyl ether, and the crude product is purified via column chromatography. This operational simplicity underscores the potential for commercial scale-up of complex pharmaceutical intermediates, as it avoids the need for specialized high-pressure or high-temperature reactors.
- Combine alkyne substrate, 2-bromo-1,3-dicarbonyl compound (2.0 equivalents), and Ir(ppy)3 photocatalyst (0.01 equivalent) in DMF solvent under nitrogen atmosphere.
- Irradiate the reaction mixture with 13W white LED lamps while stirring at room temperature for approximately 12 hours until completion monitored by TLC.
- Quench the reaction with water, extract with diethyl ether, concentrate the organic phase, and purify the crude product via column chromatography to obtain the target naphthol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light mediated synthesis offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage lies in the drastic simplification of the supply chain for raw materials and reagents. By eliminating the need for hazardous diazo compounds, stoichiometric selenium reagents, or expensive rhodium catalysts, manufacturers can source cheaper, more stable starting materials like simple alkynes and bromo-dicarbonyls. This shift not only reduces the direct cost of goods sold but also mitigates the regulatory and logistical burdens associated with transporting and storing dangerous chemicals. Furthermore, the mild reaction conditions translate to significant energy savings, as there is no requirement for cryogenic cooling or high-temperature heating, allowing reactions to proceed efficiently at ambient temperatures using low-energy LED arrays.
- Cost Reduction in Manufacturing: The economic implications of switching to this photoredox protocol are profound. Traditional methods often incur high costs due to the use of precious metal catalysts in stoichiometric quantities or the need for extensive waste treatment to handle toxic byproducts like selenium waste. In this new process, the iridium catalyst is used in minute catalytic amounts (0.01 equivalent), and the absence of stoichiometric oxidants means less waste to treat and dispose of. The elimination of expensive purification steps required to remove heavy metal residues further drives down the total manufacturing cost. Additionally, the high atom economy ensures that a greater proportion of the raw material mass ends up in the final product, maximizing yield efficiency and minimizing raw material expenditure per kilogram of output.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents or the volatility of markets for hazardous chemicals. This patented method relies on widely available commodity chemicals—terminal alkynes and alpha-bromo ketones—which are produced globally in large volumes. This abundance ensures a stable supply base and reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions means that production is less susceptible to variations in utility availability; since the process does not rely on extreme temperatures or pressures, it can be run reliably in diverse manufacturing environments. This reliability is crucial for maintaining just-in-time delivery schedules for downstream pharmaceutical clients who depend on consistent intermediate supply.
- Scalability and Environmental Compliance: As the industry moves towards stricter environmental regulations, the green chemistry credentials of this process provide a significant compliance advantage. The avoidance of toxic additives and the use of visible light align perfectly with modern sustainability goals, reducing the facility's environmental footprint. From a scalability standpoint, photochemical reactions have historically been difficult to scale due to light penetration issues (the Beer-Lambert law). However, the advent of flow chemistry and advanced LED reactor designs has largely solved this problem, allowing for the seamless translation of this lab-scale protocol to multi-ton production. The mild conditions also reduce the stress on reactor vessels, extending equipment lifespan and lowering maintenance costs, thereby enhancing the overall long-term viability of the manufacturing asset.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational parameters and strategic benefits for potential partners. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement specialists assessing the quality and consistency of the supply.
Q: What are the advantages of this photoredox method over traditional rhodium or selenium catalyzed routes?
A: Unlike traditional methods requiring hazardous diazo compounds, UV irradiation, or stoichiometric toxic additives like PhSeCl and FeCl3, this patented process utilizes mild visible light and catalytic amounts of iridium. It operates at room temperature, avoids equivalent oxidants, and offers superior atom economy and environmental safety.
Q: Can this synthesis method be applied to complex natural products like Neojusticin?
A: Yes, the patent explicitly demonstrates the utility of this methodology in synthesizing precursors for lignan natural products such as Neojusticin and Justicidin B. The broad substrate scope allows for the introduction of various aryl and alkyl groups essential for building these complex bioactive scaffolds.
Q: What is the typical catalyst loading and reaction time for this transformation?
A: The process is highly efficient, utilizing a low catalyst loading of only 0.01 equivalent (1 mol%) of the Ir(ppy)3 photosensitizer. Under standard conditions with 13W white LED irradiation, the radical cascade reaction typically reaches completion within 12 hours at room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Naphthols Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible-light photoredox synthesis described in CN103467282A and have integrated similar advanced technologies into our manufacturing portfolio. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical companies. We understand that the synthesis of complex scaffolds like polysubstituted naphthols demands precision, and our team of expert chemists is dedicated to optimizing every step of the process to maximize yield and minimize impurities.
We invite you to collaborate with us to leverage these cutting-edge synthetic methods for your next project. Whether you require custom synthesis of lignan precursors or large-scale production of naphthol derivatives, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your supply chain. Let us be your partner in turning advanced patent chemistry into commercial success.
