Advanced Synthesis of N-Acetylcysteine from Cysteine Mother Liquor for Commercial Scale-up
Advanced Synthesis of N-Acetylcysteine from Cysteine Mother Liquor for Commercial Scale-up
The pharmaceutical industry is constantly seeking sustainable pathways to transform waste streams into high-value active pharmaceutical ingredients (APIs) and intermediates. Patent CN109096161B introduces a groundbreaking preparation method for N-acetylcysteine, a critical mucolytic agent and antioxidant, by utilizing cysteine mother liquor as the primary raw material. This innovation represents a paradigm shift from traditional linear manufacturing to a circular economy model within fine chemical synthesis. Instead of discarding the mother liquor generated during cysteine production or subjecting it to energy-intensive recycling processes, this method directly converts the residual cysteine into N-acetylcysteine through a controlled acylation reaction. The technical breakthrough lies in the precise manipulation of pH levels and temperature profiles to maximize yield while suppressing the formation of complex impurities such as N,S-diacetylcysteine. For global procurement teams and R&D directors, this patent offers a compelling value proposition: a robust, scalable, and environmentally friendly route to a high-demand pharmaceutical intermediate that significantly lowers the barrier to entry for cost-effective manufacturing.
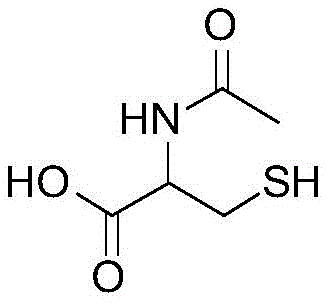
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of cysteine and its derivatives has been plagued by inefficient waste management strategies that inflate operational expenditures and environmental liabilities. In the conventional process, cysteine hydrochloride is typically produced by electrolyzing cystine, and the subsequent neutralization to obtain free cysteine generates a mother liquor rich in unreacted cysteine, often containing concentrations between 100 g/L and 120 g/L. Traditionally, this mother liquor was either discarded, creating a substantial wastewater treatment burden, or recycled back into the electrolysis loop to regenerate cysteine hydrochloride. This recycling approach is fundamentally flawed because the cysteine hydrochloride is susceptible to oxidation into cystine during the process, leading to a prolonged and convoluted process route. Furthermore, the electrolysis step requires significant electrical energy and specialized equipment, driving up capital and operational costs. The accumulation of chloride ions and other byproducts in the recycling loop can also compromise the quality of the final product, making it difficult to meet stringent pharmacopoeial standards without extensive purification steps. Consequently, enterprises face increased investment in manpower, material resources, and energy, alongside heightened environmental protection pressures due to the complex composition of the resulting wastewater.
The Novel Approach
The novel approach detailed in patent CN109096161B elegantly circumvents these bottlenecks by repurposing the cysteine mother liquor directly for the synthesis of N-acetylcysteine. Rather than attempting to recover pure cysteine first, the process adjusts the pH of the mother liquor to an alkaline range of 9 to 12 and introduces acetic anhydride at a controlled temperature of 40°C to 60°C. This direct acylation strategy eliminates the need for the energy-intensive electrolysis step entirely, thereby shortening the production cycle and reducing the overall carbon footprint of the manufacturing facility. By integrating the acylation reaction directly into the downstream processing of the cysteine mother liquor, the method transforms a waste liability into a revenue-generating asset. The process includes a sophisticated multi-stage crystallization and recovery system where the acylation mother liquor is treated to recover additional crude product, ensuring that the utilization rate of the raw material is maximized. This holistic approach not only improves the profit margin by reducing raw material costs but also alleviates the environmental pressure associated with wastewater discharge, thereby enhancing the overall competitiveness of the enterprise in the global market for pharmaceutical intermediates.
Mechanistic Insights into Acylation and Impurity Control
The core of this synthesis lies in the nucleophilic acyl substitution reaction where the amino group of cysteine attacks the carbonyl carbon of acetic anhydride. To ensure high selectivity and yield, the reaction conditions must be meticulously optimized. The patent specifies adjusting the pH of the cysteine mother liquor to between 9.5 and 11.8 prior to the addition of acetic anhydride. This alkaline environment is crucial for deprotonating the ammonium group of cysteine, thereby increasing its nucleophilicity and facilitating the attack on the acetic anhydride. However, the temperature must be strictly maintained between 40°C and 60°C; temperatures below 40°C result in sluggish reaction kinetics, while temperatures exceeding 60°C promote undesirable side reactions, such as the acetylation of the thiol group or the formation of N,S-diacetylcysteine. The dropwise addition of acetic anhydride over a period of 20 to 60 minutes allows for precise thermal management, preventing localized exotherms that could degrade the product or trigger runaway side reactions. Following the addition, the mixture is held at 60°C to 70°C for 20 to 30 minutes to ensure complete conversion, with the reaction endpoint monitored via ninhydrin testing to confirm the absence of free primary amines.
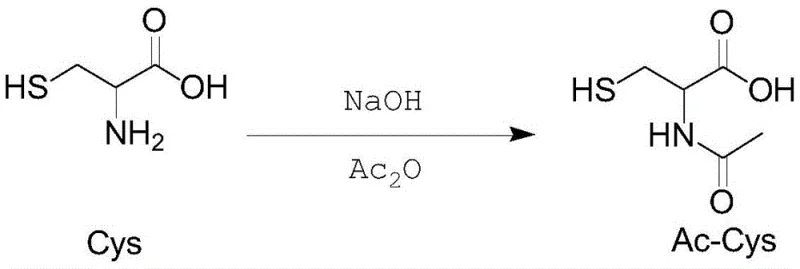
Impurity control is another critical aspect of this mechanistic design, particularly concerning the recovery of the mother liquor. During the recovery of the acylation mother liquor, the patent highlights the importance of pH adjustment before concentration. Specifically, adjusting the pH to 4.0-5.0 using sodium hydroxide prior to vacuum concentration effectively inhibits the formation of Impurity D (N,S-diacetylcysteine). If the pH is not adjusted, the acidic conditions during concentration can catalyze the migration of the acetyl group or further acylation, leading to higher levels of diacetylated byproducts which are difficult to remove in subsequent recrystallization steps. The data indicates that maintaining the correct pH profile throughout the recovery stages keeps the levels of impurities such as cystine, N,N-diacetylcystine, and N,S-diacetylcysteine well below the 0.5% threshold required by the Japanese AJI97 standard. This rigorous control over the chemical environment ensures that the final recrystallized product achieves a purity of over 99.3% with a specific rotation consistent with the L-enantiomer, demonstrating the robustness of the process for producing high-quality pharmaceutical grade material.
How to Synthesize N-Acetylcysteine Efficiently
The synthesis of N-acetylcysteine via this patented route involves a sequence of unit operations designed for maximum efficiency and yield. The process begins with the concentration of the cysteine mother liquor under reduced pressure to increase the substrate density, followed by pH adjustment and the controlled addition of acetic anhydride. The subsequent neutralization and crystallization steps are critical for isolating the crude product, which is then subjected to a multi-cycle recovery process to extract remaining value from the mother liquors. Finally, a recrystallization step using activated carbon decolorization ensures the removal of trace organic impurities and color bodies, yielding a white crystalline powder that meets international quality standards. The detailed standardized synthesis steps, including specific reagent quantities, timing, and equipment specifications, are outlined in the guide below to assist process engineers in replicating this high-efficiency protocol.
- Adjust the pH of cysteine mother liquor to 9-12 and concentrate under reduced pressure.
- Add acetic anhydride dropwise at 40-60°C to perform the acylation reaction.
- Neutralize the acylation liquid, crystallize the crude product, and recrystallize to obtain refined N-acetylcysteine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative advantages that extend beyond simple unit cost calculations. By valorizing a waste stream that was previously a disposal cost, the process fundamentally alters the cost structure of N-acetylcysteine production. The elimination of the electrolysis step, which is both capital and energy-intensive, results in a drastically simplified manufacturing workflow that requires less specialized equipment and lower utility consumption. This simplification translates directly into reduced operational expenditures and a lower break-even point for production facilities. Furthermore, the reliance on commercially available reagents like acetic anhydride and sodium hydroxide ensures that the supply chain remains resilient and不受制于 specialized precursor shortages. The ability to source raw materials from the existing cysteine production infrastructure creates a synergistic effect, allowing manufacturers to optimize their overall facility utilization and reduce the lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the conversion of a low-value byproduct into a high-value API intermediate. By bypassing the need to recycle mother liquor back to cysteine hydrochloride via electrolysis, manufacturers avoid the substantial electricity costs and electrode maintenance associated with that process. Additionally, the high yield reported in the examples, approaching quantitative conversion in optimized runs, means that less raw material is wasted, further driving down the cost per kilogram of the final product. The process also minimizes the consumption of auxiliary chemicals for pH adjustment compared to the cyclic electrolysis method, contributing to substantial cost savings in reagent procurement. These factors combined create a highly competitive cost position for producers adopting this technology.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex, multi-step processes that have multiple points of failure. This streamlined acylation route reduces the number of processing stages, thereby decreasing the probability of batch failures and production delays. Since the starting material is derived directly from the cysteine production line, the integration of these two processes creates a closed-loop system that is less dependent on external suppliers for intermediate feedstocks. This vertical integration capability allows manufacturers to better predict production schedules and fulfill large-volume orders with greater consistency. Moreover, the robustness of the reaction conditions, which tolerate slight variations in mother liquor concentration without compromising final purity, adds a layer of operational flexibility that is crucial for maintaining steady supply in a fluctuating market.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method significantly reduces the volume and toxicity of wastewater by eliminating the need for electrolysis and minimizing the discharge of untreated cysteine-rich mother liquor. The simplified process flow facilitates easier scale-up from pilot plant to commercial production, as it relies on standard reactor types and crystallization units rather than specialized electrochemical cells. The reduced environmental footprint not only lowers compliance costs related to waste treatment but also aligns with the sustainability goals of major multinational pharmaceutical companies, making the supplier a more attractive partner for long-term contracts. The ability to scale complex pharmaceutical intermediates while adhering to green chemistry principles is a distinct competitive advantage in the modern marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acetylcysteine preparation method. These answers are derived directly from the experimental data and technical disclosures within patent CN109096161B, providing a reliable foundation for decision-making. Understanding the nuances of impurity control, yield optimization, and regulatory compliance is essential for stakeholders evaluating the feasibility of this technology for their own production lines. The insights provided here reflect the proven capabilities of the process to deliver high-quality material consistently.
Q: How does this method improve upon traditional cysteine recycling?
A: Traditional methods involve electrolyzing mother liquor back to cysteine hydrochloride, which is energy-intensive and prone to oxidation. This patent bypasses electrolysis by directly acylating the mother liquor, significantly reducing energy consumption and process steps.
Q: What is the purity level achievable with this synthesis route?
A: The process yields refined N-acetylcysteine that meets the Japanese AJI97 standard, with content exceeding 99.3%, transmittance over 98.7%, and specific rotation between +25.4° and +26.4°.
Q: How are impurities like N,S-diacetylcysteine controlled?
A: Impurity formation is minimized by strictly controlling the pH during the recovery of acylation mother liquor. Adjusting the pH to 4.0-5.0 before concentration effectively inhibits the generation of N,S-diacetylcysteine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetylcysteine Supplier
The technical potential of converting cysteine mother liquor into high-purity N-acetylcysteine is immense, offering a clear pathway to more sustainable and cost-effective pharmaceutical manufacturing. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this one can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, meeting the exacting demands of global pharmacopoeias including USP, EP, and JP. We understand that the transition to a new synthesis route requires a partner who can navigate the complexities of process validation and regulatory filing with precision and speed.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to verify the quality and viability of our N-acetylcysteine offerings firsthand. Let us collaborate to drive efficiency and value in your pharmaceutical intermediate sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
