Advanced Synthesis of Pyrazolylcarboxanilides: A Cost-Effective Route for Commercial Fungicide Production
Advanced Synthesis of Pyrazolylcarboxanilides: A Cost-Effective Route for Commercial Fungicide Production
The global demand for high-performance fungicides, particularly the class of pyrazolylcarboxanilides including prominent active ingredients like Bixafen and Fluxapyroxad, necessitates robust and economically viable manufacturing processes. Patent CN103619822A, published on March 5, 2014, introduces a transformative methodology for producing these compounds by reacting pyrazolyl carboxylic acid esters directly with anilines in the presence of a base. This innovation addresses long-standing inefficiencies in the synthesis of antifungal agents by bypassing the traditional, multi-step activation of the carboxylic acid moiety. For R&D directors and procurement specialists in the agrochemical sector, this patent represents a pivotal shift towards greener chemistry and reduced operational expenditures. The core breakthrough lies in the ability to perform direct aminolysis on unactivated esters under relatively mild conditions, a feat previously considered difficult due to the low reactivity of esters compared to acid chlorides. By leveraging specific solvent systems and equilibrium-shifting techniques, this method delivers high-purity intermediates suitable for commercial-scale production.
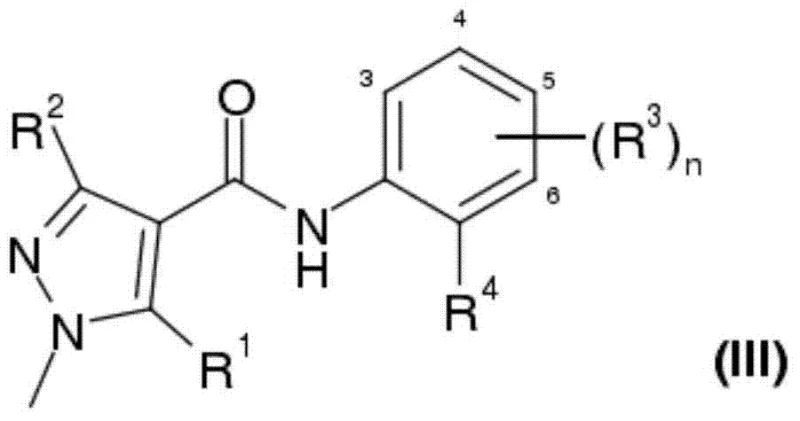
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pyrazolylcarboxanilides has relied heavily on the coupling of pyrazolyl carbonyl halides, specifically acid chlorides, with various aniline derivatives. As detailed in the background art of the patent, this conventional pathway suffers from significant economic and ecological drawbacks. The primary bottleneck is that the pyrazolyl carbonyl chloride coupling partner must be prepared from the corresponding pyrazolyl carboxylic acid ester through a two-step sequence involving hydrolysis to the acid followed by chlorination. Each additional synthetic step introduces cumulative yield losses, increases solvent consumption, and generates substantial quantities of hazardous waste, such as thionyl chloride byproducts. Furthermore, acid chlorides are inherently unstable and corrosive, requiring stringent safety protocols and specialized equipment for handling, which inflates capital expenditure. The necessity for stoichiometric bases to neutralize the hydrogen chloride generated during the coupling further complicates the workup procedure, often leading to difficult purification challenges and lower overall atom economy. For a reliable agrochemical intermediate supplier, these factors translate into higher production costs and longer lead times, making the traditional acid chloride route increasingly unsustainable in a competitive market.
The Novel Approach
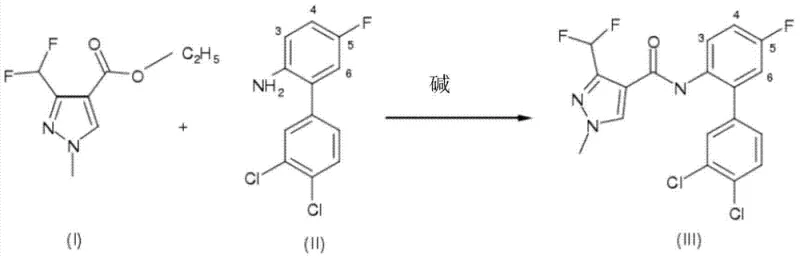
In stark contrast, the novel approach described in CN103619822A streamlines the synthesis by enabling the direct reaction of pyrazolyl carboxylic acid esters with anilines. This method effectively collapses the multi-step sequence into a single pot operation, drastically simplifying the process flow. The reaction proceeds in the presence of a base, such as sodium methoxide or sodium ethoxide, within an inert organic solvent system. A critical component of this success is the continuous removal of at least one reaction product, specifically the alcohol formed during the aminolysis (e.g., methanol or ethanol). By employing techniques such as azeotropic distillation under reduced pressure, the reaction equilibrium is forcibly shifted towards the formation of the desired pyrazolylcarboxanilide product, overcoming the thermodynamic limitations typically associated with ester aminolysis. This strategy not only enhances conversion rates but also mitigates the risk of reverse reactions. The result is a highly efficient process that operates at moderate temperatures, typically between 50°C and 100°C, ensuring energy efficiency while maintaining high selectivity for the target amide bond formation.
Mechanistic Insights into Base-Catalyzed Direct Aminolysis
The mechanistic foundation of this synthesis relies on the nucleophilic attack of the aniline nitrogen on the carbonyl carbon of the pyrazolyl ester. Under standard conditions, this reaction is sluggish because the alkoxide leaving group is a stronger base than the incoming aniline nucleophile, making the equilibrium unfavorable. However, the introduction of a strong alkoxide base serves a dual purpose: it increases the nucleophilicity of the aniline and facilitates the departure of the alkoxy group. Crucially, the patent highlights a surprising chemical stability: the acidic difluoromethyl group located at the 3-position of the pyrazole ring, which is a key pharmacophore for antifungal activity, remains intact despite the strongly basic environment. Typically, such fluorinated groups are susceptible to decomposition or elimination under alkaline conditions, but the specific reaction parameters defined in this invention preserve the structural integrity of the molecule. This stability is paramount for maintaining the biological efficacy of the final fungicide. The mechanism is further driven by Le Chatelier's principle; as the alcohol byproduct is physically removed from the reaction matrix via distillation, the system compensates by producing more product, effectively pushing the reaction to near-completion without the need for excessive reagent loading.
Impurity control is another vital aspect of this mechanistic design. In conventional acid chloride routes, side reactions often lead to the formation of urea derivatives or over-chlorinated byproducts that are difficult to separate. The direct aminolysis route minimizes these risks by avoiding highly reactive acylating agents. The use of a mixed solvent system, preferably comprising toluene and N-methylpyrrolidone (NMP), optimizes the solubility of both the polar transition states and the non-polar organic substrates. This solvation effect ensures a homogeneous reaction mixture, which promotes uniform heat transfer and reduces the likelihood of localized hot spots that could trigger thermal degradation. Furthermore, the removal of volatile alcohols prevents transesterification side reactions that could occur if the alcohol were allowed to accumulate. The resulting crude product typically exhibits high purity, reducing the burden on downstream crystallization and filtration steps. For quality assurance teams, this means a more consistent impurity profile and a simpler path to meeting stringent regulatory specifications for active pharmaceutical or agrochemical ingredients.
How to Synthesize Pyrazolylcarboxanilides Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize cycle time. The process begins with the dissolution of the specific pyrazolyl carboxylic acid ester and the chosen aniline derivative in a solvent mixture, with a toluene to NMP ratio of approximately 5:1 being particularly effective. The base, typically supplied as a solution in the corresponding alcohol (e.g., 30% sodium methoxide in methanol), is added gradually to manage the exotherm and ensure complete mixing. Once the base is introduced, the system is heated, and vacuum is applied to initiate the removal of the alcohol byproduct. The pressure is carefully modulated, often starting around 500 mbar and decreasing to 200 mbar, to facilitate the azeotropic removal of volatiles without bumping or losing valuable solvent. Monitoring the reaction progress via GC or HPLC allows operators to determine the optimal endpoint, usually achieved within a few hours depending on the specific substrate sterics. Upon completion, the product is isolated through controlled crystallization, often induced by the addition of water and cooling, followed by filtration and drying.
- Dissolve the pyrazolyl carboxylic acid ester and the substituted aniline compound in an inert organic solvent mixture, preferably toluene and N-methylpyrrolidone (NMP).
- Add an alkali metal alkoxide base, such as sodium methoxide or sodium ethoxide, to the reaction mixture under stirring at elevated temperatures between 50°C and 100°C.
- Apply vacuum to the system to continuously remove the formed alcohol byproduct (methanol or ethanol) via azeotropic distillation, driving the reaction equilibrium towards the desired amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic advantages that extend beyond simple chemistry. The elimination of the acid chloride intermediate step translates directly into substantial cost savings by removing the need for chlorinating reagents and the associated waste disposal costs. This reduction in process complexity also shortens the manufacturing cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates. Moreover, the reliance on commercially available and stable starting materials—pyrazolyl esters and anilines—enhances supply chain resilience. Unlike acid chlorides, which often have limited shelf lives and require cold storage, esters are robust and easy to transport, reducing logistics risks and storage overheads. The process is inherently safer, operating at atmospheric or slightly reduced pressures without the generation of corrosive hydrogen chloride gas, which lowers insurance premiums and maintenance costs for reactor vessels. These factors collectively contribute to a more sustainable and economically attractive supply model for high-volume fungicide production.
- Cost Reduction in Manufacturing: The streamlined one-pot synthesis eliminates the capital and operational expenses associated with the separate preparation and handling of pyrazolyl carbonyl chlorides. By avoiding the use of hazardous chlorinating agents like thionyl chloride, the process significantly reduces the cost of raw materials and the expense of neutralizing acidic off-gases. Additionally, the high atom economy of the direct aminolysis reaction means less waste is generated per kilogram of product, lowering the environmental compliance costs related to effluent treatment. The ability to recover and recycle the solvent system further amplifies these savings, making the overall cost of goods sold (COGS) markedly lower compared to legacy methods. This economic efficiency allows suppliers to offer more competitive pricing structures to downstream formulators without compromising on margin.
- Enhanced Supply Chain Reliability: The use of stable ester precursors mitigates the supply risks associated with reactive acid chlorides, which can degrade during storage or transport. This stability ensures that raw material inventories remain viable for longer periods, providing a buffer against market fluctuations and supply disruptions. The simplified process flow also reduces the number of unit operations required, decreasing the probability of equipment failure or batch rejection due to procedural errors. Consequently, manufacturers can guarantee more consistent delivery schedules and maintain higher service levels for their clients. The robustness of the reaction conditions means that production can be scaled up with greater confidence, ensuring that large-volume demands for key agrochemical intermediates can be met reliably throughout the growing season.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with modern green chemistry principles by minimizing waste generation and avoiding toxic reagents. The absence of halogenated byproducts simplifies the waste stream, making it easier and cheaper to treat or dispose of in compliance with strict environmental regulations. The process is highly scalable, as the removal of alcohol by distillation is a standard unit operation that can be easily engineered for large-scale reactors ranging from pilot plant to multi-ton production. The mild reaction temperatures reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing facility. These environmental benefits not only satisfy regulatory requirements but also enhance the corporate social responsibility profile of the manufacturer, appealing to eco-conscious partners in the global agricultural value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement officers assessing the quality of intermediates sourced via this method.
Q: Why is the direct aminolysis of esters preferred over the acid chloride route for pyrazolylcarboxanilides?
A: The direct aminolysis route eliminates the need for a separate two-step preparation of pyrazolyl carbonyl chlorides, thereby reducing raw material costs, minimizing hazardous waste generation, and simplifying the overall process flow for industrial scale-up.
Q: How does the process prevent the decomposition of the sensitive difluoromethyl group on the pyrazole ring?
A: Surprisingly, the patent data indicates that the acidic difluoromethyl group remains stable under the specific basic reaction conditions employed (alkali metal alkoxides), allowing for high-yield formation of the amide without degradation of this critical functional group.
Q: What role does vacuum distillation play in this synthesis method?
A: Vacuum distillation is critical for shifting the reaction equilibrium; by continuously removing the alcohol byproduct (methanol or ethanol) as an azeotrope with the solvent, the reaction is driven to completion, significantly improving yield and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolylcarboxanilides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the agrochemical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative methods described in patents like CN103619822A are translated into reality with precision. We are committed to delivering high-purity pyrazolylcarboxanilides that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry allows us to optimize reaction conditions for maximum yield and minimal impurity formation, providing our partners with a distinct competitive advantage in the marketplace.
We invite you to collaborate with us to leverage these advanced synthesis technologies for your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall production costs. Let us be your trusted partner in bringing high-performance fungicides to the global market.
