Advanced Organosilicon Cyclization for High-Purity Quinolone Pharmaceutical Intermediates
Introduction to Patent CN1481365A and Quinolone Innovation
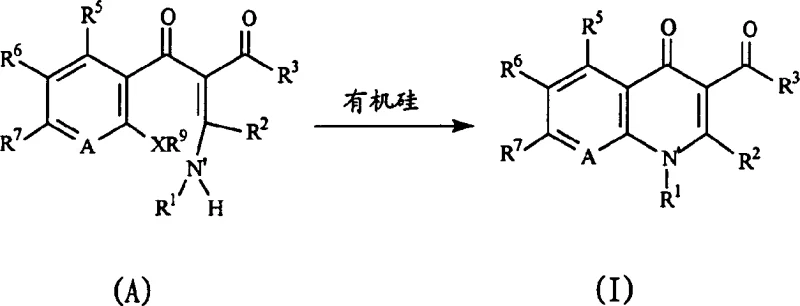
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibacterial and anti-HIV agents, specifically within the quinolone and naphthyridine classes. Patent CN1481365A introduces a transformative cyclization process that leverages organosilicon chemistry to construct these complex heterocyclic cores with unprecedented efficiency. This technology addresses long-standing challenges in medicinal chemistry by enabling the use of diverse starting materials that were previously incompatible with conventional cyclization methods. By shifting away from harsh basic conditions, this innovation opens new avenues for designing next-generation antimicrobial therapeutics with improved safety profiles. For global procurement and R&D teams, understanding this proprietary methodology is essential for securing reliable supply chains of high-value pharmaceutical intermediates. The strategic implementation of this silylation-based approach represents a significant leap forward in process chemistry, offering a pathway to more sustainable and cost-effective manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formation of quinolone rings has relied heavily on cyclization strategies that demand severe reaction conditions, often necessitating the use of strong bases such as sodium hydride. These traditional protocols typically require elevated temperatures ranging from 140°C to 160°C in polar solvents, which imposes significant thermal stress on sensitive molecular architectures. Furthermore, prior art methods frequently mandate the presence of electron-withdrawing groups on the aromatic ring precursor to facilitate the cyclization event, severely limiting the structural diversity of accessible compounds. Such restrictive requirements complicate the synthesis of novel analogs needed to combat emerging microbial resistance. The reliance on aggressive reagents also introduces safety hazards and increases the complexity of waste management, creating substantial operational burdens for manufacturing facilities aiming to scale production efficiently.
The Novel Approach
In stark contrast, the methodology disclosed in CN1481365A utilizes organosilicon reagents to mediate the intramolecular cyclization under markedly milder conditions. This innovative route allows for the successful incorporation of electron-donating leaving groups on the aromatic precursor, vastly expanding the chemical space available for drug discovery efforts. The reaction can proceed effectively at temperatures between 20°C and 140°C, eliminating the need for extreme thermal input and reducing energy consumption significantly. By employing reagents like N,O-bis(trimethylsilyl)acetamide, the process achieves high conversion rates while maintaining the integrity of sensitive functional groups elsewhere in the molecule. This flexibility not only accelerates the development timeline for new candidates but also enhances the overall robustness of the manufacturing process for commercial supply.
Mechanistic Insights into Organosilicon-Mediated Cyclization
The core of this technological advancement lies in the unique activation mechanism provided by the organosilicon species during the ring-closing step. The silicon reagent interacts with the heteroatom systems within the precursor, effectively masking reactive protons and facilitating the nucleophilic attack required for ring closure. This silylation event stabilizes intermediate states that would otherwise be unstable under traditional basic conditions, allowing the reaction to proceed through a lower energy transition state. Consequently, the process tolerates a broader array of substituents, including those that are electronically rich, which are typically problematic in standard cyclizations. This mechanistic nuance ensures that the final quinolone or naphthyridine structure is formed with high regioselectivity and minimal formation of undesired byproducts. Understanding this interaction is crucial for R&D directors aiming to optimize yield and purity parameters for complex API intermediates.
Impurity control is another critical aspect where this mechanism offers distinct advantages over legacy technologies. The milder reaction environment significantly reduces the likelihood of thermal degradation or side reactions such as hydrolysis or polymerization of the starting materials. By avoiding strong bases that can promote elimination or rearrangement pathways, the process yields a cleaner crude product profile. This reduction in impurity burden simplifies the downstream purification steps, such as crystallization or chromatography, leading to higher overall recovery rates. For quality assurance teams, this translates to more consistent batch-to-batch reproducibility and easier compliance with stringent pharmacopoeial standards. The ability to maintain structural fidelity throughout the synthesis is paramount for ensuring the efficacy and safety of the final therapeutic agent.
How to Synthesize Quinolone Derivatives Efficiently
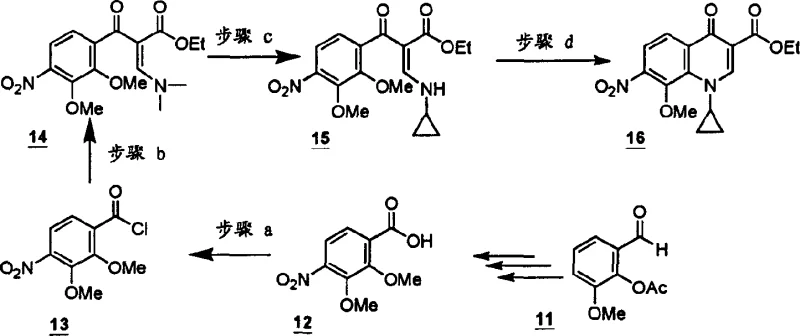
Implementing this synthesis route requires careful selection of the organosilicon reagent and solvent system to match the specific substitution pattern of the target molecule. The process generally involves dissolving the aromatic precursor in an aprotic solvent such as acetonitrile, toluene, or N-methylpyrrolidone before introducing the silylating agent. Detailed standardized synthesis steps see the guide below. Operators must monitor the reaction progress closely, typically utilizing heat to drive the cyclization to completion while ensuring that temperatures remain within the optimal range to prevent decomposition. The workup procedure involves standard aqueous extraction and solvent removal techniques, which are well-established in industrial settings. This straightforward operational protocol makes the technology highly accessible for contract development and manufacturing organizations looking to adopt efficient processes for quinolone production.
- React the aromatic ring precursor (Formula A) with an organosilicon reagent such as N,O-bis(trimethylsilyl)acetamide in an aprotic solvent.
- Maintain the reaction mixture at temperatures ranging from 20°C to 140°C, significantly lower than traditional high-temperature methods.
- Perform standard workup procedures including solvent evaporation and extraction to isolate the high-purity cyclized quinolone product (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this organosilicon-mediated cyclization process offers substantial benefits for procurement and supply chain management strategies. The elimination of harsh reaction conditions directly correlates with reduced operational risks and lower energy costs associated with heating and cooling cycles in large-scale reactors. Furthermore, the ability to utilize a wider range of starting materials mitigates the risk of supply disruptions caused by the scarcity of specialized electron-deficient precursors. This flexibility allows sourcing teams to negotiate better pricing with raw material vendors by accessing a broader market of commodity chemicals. The overall simplification of the synthetic route also reduces the number of processing steps required, leading to shorter manufacturing lead times and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous reagents like sodium hydride, which require special handling and disposal protocols. By replacing these with commercially available organosilicon compounds, the direct material costs are significantly optimized without compromising reaction efficiency. Additionally, the milder temperature requirements result in drastic energy savings, as less fuel or electricity is needed to maintain reactor conditions. The reduced formation of byproducts also lowers the cost associated with waste treatment and solvent recovery, contributing to a leaner and more economical production model. These cumulative savings enhance the overall cost competitiveness of the manufactured intermediates in the global marketplace.
- Enhanced Supply Chain Reliability: Utilizing common aprotic solvents and stable organosilicon reagents ensures that the supply chain is less vulnerable to logistical bottlenecks. Unlike specialized catalysts or sensitive reagents that may have long lead times, the inputs for this process are widely produced and easily sourced from multiple suppliers. This redundancy strengthens supply continuity, ensuring that production schedules can be maintained even during periods of market volatility. The robustness of the chemistry also means that technology transfer between manufacturing sites is smoother, reducing the risk of delays during scale-up activities. Reliable access to high-quality intermediates is thus secured, supporting the uninterrupted production of finished pharmaceutical products.
- Scalability and Environmental Compliance: The inherent safety of operating at lower temperatures and pressures makes this process highly scalable from pilot plants to commercial tonnage production. The reduced hazard profile aligns well with increasingly stringent environmental, health, and safety regulations, minimizing the regulatory burden on manufacturing facilities. Lower energy consumption and reduced waste generation contribute to a smaller carbon footprint, supporting corporate sustainability goals. The process design facilitates efficient containment and treatment of any emissions, ensuring compliance with local and international environmental standards. This alignment with green chemistry principles enhances the corporate reputation and long-term viability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclization technology in pharmaceutical manufacturing. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for stakeholders. Understanding these details helps in making informed decisions about process adoption and supplier qualification. The answers reflect the practical implications of the chemistry on production efficiency and product quality. Stakeholders are encouraged to review these points when evaluating potential partnerships for intermediate supply.
Q: What are the primary advantages of using organosilicon reagents in quinolone synthesis?
A: The use of organosilicon reagents allows for milder reaction conditions compared to traditional methods requiring strong bases like sodium hydride. This approach accommodates a wider range of leaving groups, including electron-donating groups, thereby expanding the scope of synthesizable derivatives while reducing thermal stress on the molecule.
Q: How does this cyclization process impact impurity profiles in pharmaceutical intermediates?
A: By avoiding harsh conditions such as extreme temperatures and aggressive bases, this method minimizes side reactions and degradation pathways. The result is a cleaner crude reaction mixture, which simplifies downstream purification and ensures higher overall purity specifications for the final active pharmaceutical ingredient intermediates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available organosilicon reagents and common aprotic solvents like acetonitrile or toluene. The moderate temperature requirements enhance operational safety and energy efficiency, making it highly scalable for industrial production of complex quinolone and naphthyridine scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Intermediates Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies like the organosilicon cyclization process to deliver superior pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory agencies. Our commitment to process excellence means that we can offer clients a secure and consistent supply of complex quinolone derivatives. By leveraging our expertise in this specific technology, we help partners accelerate their drug development timelines while managing costs effectively.
We invite procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this technology can optimize your supply chain. Contact us today to discuss how our capabilities align with your strategic sourcing goals. Together, we can build a resilient supply partnership that drives innovation and efficiency in the pharmaceutical sector. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
