Industrial Scale-Up of Sertindole Intermediates Using Green Aqueous Copper Catalysis
Industrial Scale-Up of Sertindole Intermediates Using Green Aqueous Copper Catalysis
The pharmaceutical industry constantly seeks robust manufacturing pathways that balance high purity with environmental sustainability and cost efficiency. Patent CN1301963C presents a transformative approach to synthesizing N-(4-fluorophenyl)-N-(2-carboxy-4-chlorophenyl)glycine, a critical intermediate in the production of the antipsychotic agent Sertindole. This invention fundamentally shifts the paradigm from traditional organic solvent-based systems to an aqueous alkaline environment, leveraging copper catalysis to achieve superior results. By replacing hazardous solvents like N-methylpyrrolidone (NMP) with water, the process not only mitigates significant environmental risks but also streamlines the isolation of the final product. For R&D directors and procurement managers, this patent represents a viable route to enhance supply chain resilience while adhering to increasingly stringent green chemistry regulations. The technical depth of this methodology offers a compelling case for adopting water-mediated copper-catalyzed arylation in large-scale API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in Dansk Kemi, relied heavily on N-methylpyrrolidone (NMP) as the reaction solvent for the copper-catalyzed coupling of 2,5-dichlorobenzoic acid and N-(4-fluorophenyl)glycine. While chemically feasible, this approach suffers from severe industrial drawbacks that hinder commercial viability. The reaction typically requires elevated temperatures between 120°C and 130°C, which increases energy consumption and poses thermal safety risks on a multi-ton scale. Furthermore, NMP is known for its poor water solubility and high boiling point, making its removal and recovery from the reaction mixture an energy-intensive and time-consuming distillation process. The conventional method also tends to generate substantial amounts of tarry by-products, which complicate purification and significantly lower the overall yield of the desired glycine derivative. These factors collectively inflate the cost of goods sold (COGS) and create bottlenecks in production throughput, making the NMP-based route unattractive for modern, high-volume pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel process disclosed in CN1301963C utilizes an aqueous alkaline environment, effectively replacing the problematic NMP solvent with water or water mixed with minor co-solvents like ethylene glycol. This strategic substitution allows the reaction to proceed at significantly lower temperatures, typically around the reflux temperature of the aqueous medium (approximately 80°C to 100°C), thereby reducing energy demands. The use of water facilitates a much cleaner reaction profile, resulting in negligible amounts of tarry by-products and markedly higher yields. Workup is dramatically simplified; the product can be precipitated directly by acidifying the aqueous filtrate, bypassing complex extraction and distillation steps. This innovation not only enhances the purity of the intermediate but also aligns with green chemistry principles by minimizing volatile organic compound (VOC) emissions. For a reliable pharmaceutical intermediate supplier, adopting this aqueous protocol translates to a more robust and scalable manufacturing process.
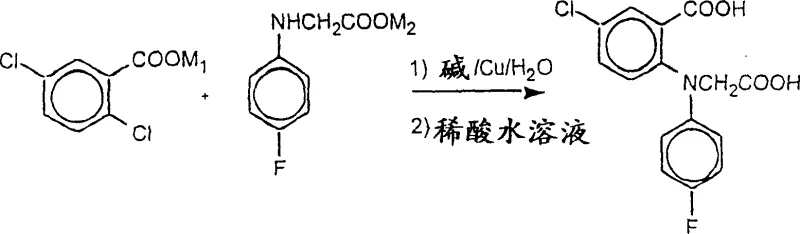
Mechanistic Insights into Copper-Catalyzed Arylation in Aqueous Media
The core of this technological advancement lies in the efficient execution of copper-catalyzed arylation within a heterogeneous aqueous system. The reaction involves the interaction of alkali metal salts, specifically potassium salts, of both 2,5-dichlorobenzoic acid and N-(4-fluorophenyl)glycine in the presence of a Cu(0) catalyst, preferably copper-bronze. Mechanistically, the alkaline environment, maintained by bases such as potassium carbonate, plays a dual role: it ensures the solubility of the carboxylic acid starting materials as their salts and neutralizes the hydrochloric acid generated during the substitution reaction. Maintaining the correct stoichiometry is crucial; the patent suggests a molar ratio of N-(4-fluorophenyl)glycine salt to 2,5-dichlorobenzoic acid salt of approximately 2.0:2.3 to drive the reaction to completion while allowing for the regeneration of excess starting material. The copper catalyst facilitates the nucleophilic aromatic substitution, likely through a mechanism involving oxidative addition and reductive elimination cycles, although the exact nature of the active copper species in water remains a subject of detailed kinetic study. The ability to perform this transformation in water without sacrificing catalytic activity is a testament to the optimized reaction conditions defined in the patent.
Impurity control is another critical aspect where this aqueous method excels over its organic solvent counterparts. In the traditional NMP process, side reactions often lead to the formation of complex, hard-to-remove tars that degrade product quality. The aqueous system suppresses these side pathways, likely due to the specific solvation effects of water on the transition states and intermediates. Furthermore, the subsequent step in the Sertindole synthesis pathway—the condensation of 5-chloro-1-(4-fluorophenyl)indole with 4-piperidone—is also improved by replacing trifluoroacetic acid (TFA) with a mixture of mineral acid (like HCl) and acetic acid. This modification prevents the formation of undesired disubstituted piperidine by-products, which are common when using highly reactive TFA. By minimizing impurity generation at these key synthetic junctures, the overall purification burden is reduced, leading to a final API intermediate that meets stringent purity specifications with less processing.
How to Synthesize N-(4-fluorophenyl)-N-(2-carboxy-4-chlorophenyl)glycine Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize waste. The process begins with the preparation of the alkali metal salts of the starting acids, followed by their suspension in a minimal amount of water to maintain high concentration and reaction rate.
- Prepare alkali metal salts of 2,5-dichlorobenzoic acid and N-(4-fluorophenyl)glycine in an aqueous alkaline environment.
- Add copper-bronze catalyst and heat the suspension to reflux temperature (approx. 80-100°C) under nitrogen atmosphere.
- Precipitate the crude product by pouring the filtered reaction mixture into dilute hydrochloric acid and ice water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from NMP to water represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. The elimination of NMP removes the need for expensive solvent recovery infrastructure and the associated energy costs of high-vacuum distillation. Water is inherently cheaper, non-flammable, and readily available, which stabilizes raw material costs and reduces supply chain volatility. Moreover, the simplified workup procedure—precipitation via acidification rather than extraction—reduces the consumption of auxiliary solvents like toluene or ethyl acetate during purification. This streamlining of the process flow shortens the batch cycle time, allowing for increased production capacity without additional capital investment in reactor volume. The environmental benefits also translate to regulatory advantages, as facilities handling less hazardous waste face lower disposal fees and reduced compliance burdens. Consequently, this technology supports a more agile and cost-effective supply chain capable of meeting fluctuating market demands for antipsychotic medications.
- Cost Reduction in Manufacturing: The replacement of high-boiling organic solvents with water drastically lowers utility costs associated with heating and solvent recovery. By avoiding the use of NMP, manufacturers eliminate the significant expense of purchasing, recycling, or disposing of this regulated solvent. The higher yields achieved through the suppression of tarry by-products mean that less raw material is wasted per kilogram of final product, directly improving the material cost efficiency. Additionally, the ability to regenerate excess N-(4-fluorophenyl)glycine from the filtrate further optimizes material utilization, ensuring that the overall process economics are favorable compared to legacy methods.
- Enhanced Supply Chain Reliability: Relying on water as the primary reaction medium reduces dependency on specialized organic solvents that may be subject to market shortages or price spikes. The robustness of the aqueous process makes it less sensitive to minor variations in raw material quality, ensuring consistent output. Simplified logistics for solvent handling and storage also reduce the risk of supply disruptions caused by transportation regulations for hazardous chemicals. This stability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream API manufacturers, thereby strengthening the reliability of the entire pharmaceutical supply network.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes it inherently safer and easier to scale from pilot plant to commercial production. The lower operating temperatures reduce thermal hazards, and the absence of flammable organic vapors minimizes explosion risks in large reactors. From an environmental perspective, the process generates significantly less hazardous waste, aligning with global sustainability goals and easing the permitting process for facility expansions. This compliance advantage ensures long-term operational continuity and protects the manufacturer's reputation as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this greener synthesis route.
Q: Why is the aqueous method superior to the traditional NMP solvent system?
A: The aqueous method eliminates the need for expensive and toxic N-methylpyrrolidone (NMP), significantly reducing solvent recovery costs and environmental hazards while improving yield and reducing tarry by-products.
Q: What catalyst is preferred for this copper-catalyzed arylation?
A: The patent specifies Cu(0) catalysts, with copper-bronze being the most preferred option due to its effectiveness in the alkaline aqueous medium.
Q: How does this process impact the scalability of Sertindole production?
A: By utilizing water as the primary solvent and avoiding high-boiling organic solvents, the process simplifies workup procedures and allows for safer, more efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(4-fluorophenyl)-N-(2-carboxy-4-chlorophenyl)glycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the aqueous copper-catalyzed arylation described in CN1301963C are executed with precision. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in green chemistry and process optimization, we help our partners reduce time-to-market while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this aqueous protocol. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for your high-value pharmaceutical intermediate needs.
