Scalable Production of 4,4-Difluoro-3-Oxobutanoates for Advanced Fungicide Manufacturing
Scalable Production of 4,4-Difluoro-3-Oxobutanoates for Advanced Fungicide Manufacturing
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for complex fluorinated compounds essential to modern crop protection. Patent CN100532345C introduces a transformative methodology for the preparation of 4,4-difluoro-3-oxobutanoic acid esters, which serve as critical building blocks for pyrazole anilide fungicides. These specific esters are pivotal in constructing the heterocyclic cores found in next-generation agricultural active ingredients, addressing the industry's relentless pursuit of efficacy and environmental safety. The disclosed process represents a significant departure from legacy synthetic routes, offering a robust pathway that balances high chemical efficiency with operational safety. By leveraging a novel condensation strategy between specific difluoroacetamides and acetate esters, this technology resolves long-standing bottlenecks related to yield optimization and hazard management in fluorine chemistry. For R&D directors and process engineers, understanding this mechanistic shift is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The ability to produce these high-value intermediates with improved purity profiles and reduced waste generation positions this patent as a cornerstone for sustainable agrochemical development.
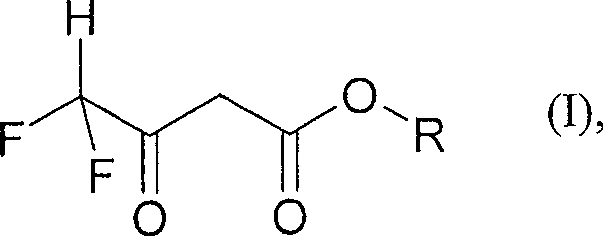
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,4-difluoro-3-oxobutanoates has been plagued by significant technical and economic inefficiencies that hinder large-scale adoption. Early literature, such as JACS 69, 1819 (1947), described the condensation of fluoroesters with acetates using weak bases like sodium ethoxide, but this approach suffered from unacceptably low yields, often stagnating around 35%. While subsequent research indicated that stronger bases like sodium hydride could elevate yields to the 75-85% range, the industrial application of sodium hydride is fraught with peril due to the evolution of explosive hydrogen gas and the stringent safety protocols required for handling pyrophoric materials. Furthermore, alternative pathways involving polyfluorocarboxylic acid chlorides, as detailed in EP-A-694526, have proven unreliable for difluoro derivatives; the inherent instability of difluoroacetyl chloride under reaction conditions leads to decomposition and side reactions, resulting in theoretical yields as low as 25%. These conventional methods impose heavy burdens on procurement teams through the need for specialized, hazardous reagents and on supply chain managers through inconsistent batch quality and extended processing times required for rigorous safety checks and purification.
The Novel Approach
The methodology outlined in CN100532345C circumvents these historical pitfalls by utilizing N,N-disubstituted-2,2-difluoroacetamides as the primary starting materials instead of unstable acid chlorides or difficult-to-handle fluoroesters. This strategic substitution allows the reaction to proceed smoothly in the presence of standard alkali metal alkoxides, such as sodium methoxide or sodium ethoxide, which are inexpensive, commercially abundant, and far safer to handle on a multi-ton scale. The process operates under mild thermal conditions, typically between 50°C and 70°C, eliminating the need for cryogenic cooling or extreme heating that characterizes many fluorination reactions. By employing the acetate ester both as a reactant and potentially as a solvent in large excess, the reaction equilibrium is driven favorably towards the product, achieving combined yields that significantly outperform the legacy acid chloride routes. This novel approach not only simplifies the operational workflow by removing dangerous unit operations but also enhances the overall atom economy of the synthesis, making it an ideal candidate for cost reduction in agrochemical intermediate manufacturing where margin pressure is intense.
Mechanistic Insights into Base-Catalyzed Condensation
The core of this synthetic breakthrough lies in the unique reactivity of the alpha-protons in the 2,2-difluoroacetamide structure, which are rendered highly acidic by the strong electron-withdrawing effect of the two fluorine atoms. In the presence of an alkoxide base, the amide readily forms a stabilized enolate species that acts as a potent nucleophile. This enolate subsequently attacks the carbonyl carbon of the acetate ester in a crossed Claisen-type condensation mechanism. Unlike traditional ester-enolate chemistry where self-condensation can be a major competing pathway, the specific electronic properties of the difluoroamide ensure high regioselectivity for the cross-coupling reaction. The tetrahedral intermediate formed during this nucleophilic acyl substitution collapses to expel the amine leaving group, thereby generating the desired beta-keto ester framework. This mechanistic pathway is exceptionally clean because the amine byproduct is easily removed during the aqueous workup, preventing contamination of the final product with difficult-to-separate organic impurities. For quality control teams, this translates to a much simpler impurity profile, reducing the burden on analytical resources and ensuring that the final intermediate meets the stringent purity specifications required for downstream pharmaceutical or agrochemical synthesis.
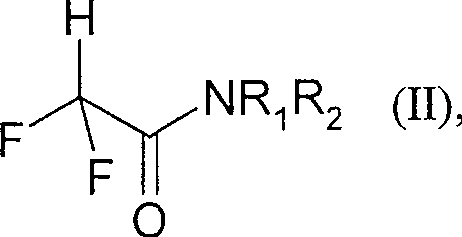
Furthermore, the choice of base and solvent system plays a critical role in optimizing the reaction kinetics and thermodynamic stability of the intermediates. The patent specifies that alkali metal alkoxides are preferred, with sodium ethoxide and sodium methoxide offering the best balance of reactivity and cost. The reaction can be conducted in the neat acetate ester or in mixtures with aromatic solvents like toluene or ethers like THF, providing flexibility for process engineers to tailor the solvent system for optimal heat transfer and mixing in large reactors. The mild temperature range of 15°C to 80°C ensures that thermal degradation of the sensitive fluorinated backbone is minimized, preserving the integrity of the difluoromethyl group which is essential for the biological activity of the final fungicide. Understanding these mechanistic nuances allows R&D directors to confidently scale the process, knowing that the reaction is robust against minor fluctuations in operating parameters and that the risk of runaway exotherms is negligible compared to processes involving strong mineral acids or pyrophoric bases.
How to Synthesize 4,4-Difluoro-3-Oxobutanoates Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the base and the selection of the appropriate acetate ester to match the desired final product specification. The process begins by dissolving the difluoroacetamide precursor in the acetate ester, often using a significant molar excess of the ester to act as both reagent and solvent, which drives the equilibrium forward. Following this, a solution of the alkoxide base is added under controlled conditions to initiate the enolization and subsequent condensation. The reaction mixture is then heated to facilitate the conversion, typically requiring several hours to reach completion as monitored by gas chromatography. Upon cooling, the reaction is quenched with acidified ice water to neutralize the base and protonate the beta-keto ester, followed by extraction into an organic phase. The crude product is then purified via distillation under reduced pressure to isolate the high-purity ester. For detailed operational parameters and specific molar ratios, please refer to the standardized synthesis guide below.
- Dissolve the difluoroacetamide starting material in an excess of the corresponding acetate ester or a suitable solvent mixture.
- Add a solution of an alkali metal alkoxide base, such as sodium ethoxide, under controlled temperature conditions.
- Heat the reaction mixture to 50-70°C until completion, followed by acidic quenching and distillation purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers profound advantages that extend well beyond the laboratory bench, directly impacting the bottom line and supply chain resilience for agrochemical manufacturers. The elimination of hazardous reagents like sodium hydride and unstable acid chlorides drastically reduces the costs associated with safety infrastructure, specialized storage, and waste disposal, leading to substantial cost savings in fungicide manufacturing. Moreover, the reliance on commodity chemicals such as sodium ethoxide and common acetate esters ensures a stable and reliable supply of raw materials, shielding production schedules from the volatility often seen with specialized fluorinating agents. This shift to safer, more abundant inputs enhances supply chain reliability by minimizing the risk of production stoppages due to reagent shortages or regulatory restrictions on hazardous materials. Additionally, the simplified workup and purification procedures reduce the overall cycle time per batch, allowing facilities to increase throughput without capital-intensive expansions. The environmental compliance profile is also markedly improved, as the process generates less hazardous waste and avoids the emission of toxic gases, aligning with increasingly strict global environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The transition from expensive and dangerous reagents to inexpensive alkoxides and stable amides fundamentally alters the cost structure of producing these intermediates. By removing the need for specialized safety measures required for pyrophoric bases and eliminating the low yields associated with acid chloride instability, the overall cost of goods sold is significantly reduced. This economic efficiency allows manufacturers to remain competitive in the global market while maintaining healthy margins, even when facing pressure to lower prices for generic agrochemical products.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key starting materials are widely available from multiple global suppliers, reducing dependency on single-source vendors for exotic reagents. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without the need for highly specialized equipment or expertise, ensuring continuity of supply even in the face of regional disruptions. This reliability is critical for meeting the just-in-time delivery expectations of downstream formulators and protecting the reputation of the supplier as a dependable partner.
- Scalability and Environmental Compliance: The mild operating conditions and absence of explosive byproducts make this process inherently scalable from pilot plant to full commercial production without the engineering challenges typical of hazardous chemistries. The reduced environmental footprint, characterized by lower waste generation and safer effluent streams, facilitates easier permitting and regulatory approval in jurisdictions with strict environmental oversight. This scalability ensures that the technology can meet growing market demand for fluorinated agrochemicals while adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived from the specific advantages and operational details found in the patent documentation. These insights are intended to clarify the practical benefits for stakeholders evaluating this technology for potential licensing or contract manufacturing partnerships. Understanding these details is essential for making informed decisions about process integration and supply chain optimization.
Q: Why is the new amide-based route superior to traditional acid chloride methods?
A: Traditional methods using difluoroacetyl chloride often suffer from instability and low yields (around 25%). The new amide-based route utilizes stable, commercially available starting materials and achieves significantly higher yields without hazardous reagents.
Q: What are the safety advantages of using alkoxide bases over sodium hydride?
A: While sodium hydride can improve yields in older methods, it poses severe explosion risks due to hydrogen gas evolution on an industrial scale. The patented process uses safer alkali metal alkoxides like sodium ethoxide, eliminating these critical safety hazards.
Q: Can this process be adapted for different ester groups?
A: Yes, the process is highly versatile. By selecting different acetate esters (e.g., methyl acetate, ethyl acetate) as solvents or reactants, manufacturers can directly synthesize the corresponding methyl or ethyl esters required for downstream fungicide production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4-Difluoro-3-Oxobutanoates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final agrochemical products. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that the transition to a new synthetic route requires confidence in the partner's capability to deliver both technically and logistically, and our track record in fluorine chemistry demonstrates our ability to handle complex molecular architectures safely and efficiently.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient route. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the compatibility of this technology with your existing manufacturing infrastructure. Let us collaborate to optimize your supply chain and drive innovation in the agrochemical sector together.
