Advanced Chiral Thioether-Phosphine Ligands for Scalable Asymmetric Allylic Alkylation
Advanced Chiral Thioether-Phosphine Ligands for Scalable Asymmetric Allylic Alkylation
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN113980053A, which introduces a novel class of chiral thioether-phosphine compounds designed to serve as highly effective ligands for palladium-catalyzed reactions. This technology addresses a critical bottleneck in modern organic synthesis: the need for versatile, tunable catalytic systems that can deliver consistent enantioselectivity across a broad range of substrates. By constructing a ligand library based on a modular thioether-phosphine scaffold, the inventors have created a platform that allows for precise steric and electronic tuning. This capability is paramount for R&D directors seeking to optimize reaction pathways for complex drug candidates, as it provides a systematic approach to solving stereoselectivity challenges that often plague late-stage functionalization processes.
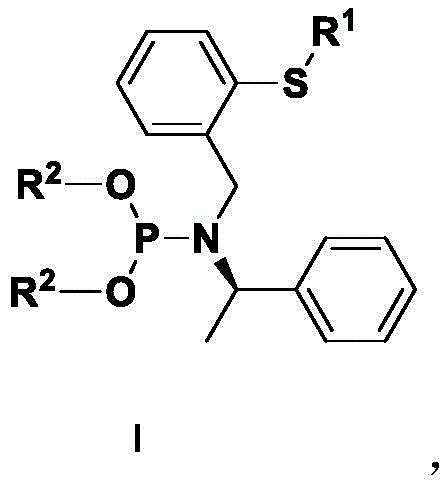
The core innovation lies in the structural flexibility of the general Formula I, where the R1 and R2 groups can be independently varied to modulate the catalyst's performance. For instance, the patent demonstrates that varying the thioether substituent (R1) from a simple methyl group to bulky naphthyl or halogenated phenyl groups significantly impacts the outcome of the asymmetric allylic alkylation. This modularity ensures that the technology is not a one-size-fits-all solution but rather a customizable toolkit for process chemists. Furthermore, the integration of a chiral amine backbone derived from (R)-(+)-alpha-methylbenzylamine provides a rigid chiral environment essential for inducing high optical purity in the final products. The ability to rapidly synthesize and screen these variants makes this patent a valuable asset for any organization focused on the efficient development of chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral ligands for transition metal catalysis has been fraught with challenges related to cost, complexity, and accessibility. Traditional methods often rely on naturally occurring chiral pools, such as amino acids or terpenes, which can be expensive and limited in structural diversity. Moreover, the synthesis of phosphine ligands frequently involves hazardous reagents, sensitive intermediates, and multi-step sequences that suffer from poor overall yields. For procurement managers, this translates into volatile supply chains and inflated raw material costs. Additionally, many conventional catalytic systems require stringent reaction conditions, such as cryogenic temperatures or inert atmospheres with ultra-low moisture content, which impose significant energy burdens and engineering constraints on manufacturing facilities. These factors collectively hinder the commercial viability of many promising asymmetric transformations, forcing companies to settle for less efficient racemic syntheses followed by resolution, a process that inherently wastes at least half of the material.
The Novel Approach
In stark contrast, the methodology outlined in CN113980053A offers a streamlined, three-step synthetic route that utilizes commercially available starting materials. The process begins with a nucleophilic aromatic substitution between 2-fluorobenzaldehyde and various thiophenols, a reaction that proceeds efficiently in polar aprotic solvents like DMSO at elevated temperatures. This is followed by a reductive amination step using sodium borohydride, a mild and cost-effective reducing agent, to install the chiral amine moiety. The final phosphitylation step connects the scaffold to the phosphorus center using phenolic compounds. This approach drastically simplifies the supply chain by relying on commodity chemicals rather than exotic precursors. From a process chemistry perspective, the robustness of these reactions allows for easier scale-up, as they do not demand specialized equipment for handling pyrophoric reagents or extreme cooling. The result is a ligand synthesis protocol that is not only chemically elegant but also economically superior, aligning perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Allylic Alkylation
The efficacy of the chiral thioether-phosphine ligands described in this patent stems from their unique ability to coordinate with palladium centers to form stable, chiral pi-allyl complexes. In the catalytic cycle, the ligand binds to the palladium atom, creating a chiral pocket that dictates the trajectory of the incoming nucleophile. The presence of both sulfur and phosphorus donor atoms (a soft-soft coordination environment) enhances the stability of the metal complex, preventing premature decomposition or aggregation of the catalyst. This dual-donor characteristic is crucial for maintaining catalytic activity over extended reaction times, which is a common failure point in industrial batch processes. The steric bulk introduced by the R1 and R2 substituents further shields specific faces of the allyl intermediate, ensuring that the nucleophilic attack occurs with high facial selectivity. This mechanistic precision is what enables the achievement of significant enantiomeric excess (ee) values, as evidenced by the experimental data where specific ligand variants achieved substantial stereocontrol.
Furthermore, the electronic properties of the ligand play a pivotal role in modulating the electrophilicity of the pi-allyl palladium species. Electron-rich or electron-deficient groups on the thioether or phosphine aryl rings can fine-tune the reaction rate and selectivity. For example, the patent highlights that ligand I-4, featuring a methoxy-substituted thioether group, exhibited superior catalytic performance compared to other variants in the library. This suggests that electron-donating groups may facilitate the oxidative addition or reductive elimination steps within the catalytic cycle. Understanding these structure-activity relationships allows chemists to rationally design next-generation ligands tailored for specific difficult substrates. By controlling the impurity profile through precise ligand design, manufacturers can reduce the burden on downstream purification processes, thereby improving the overall mass balance and sustainability of the production line.
How to Synthesize Chiral Thioether-Phosphine Compounds Efficiently
The synthesis of these high-value ligands follows a logical and scalable three-step sequence that minimizes waste and maximizes throughput. The process begins with the formation of the thioether backbone, followed by the introduction of chirality, and concludes with the installation of the phosphorus functionality. Each step has been optimized to use standard laboratory and plant equipment, ensuring that the transition from gram-scale discovery to kilogram-scale production is seamless. The detailed standardized synthetic steps for producing these compounds are provided in the guide below, offering a clear roadmap for technical teams to implement this technology immediately.
- React 2-fluorobenzaldehyde with thiophenol derivatives in DMSO with potassium carbonate at 100°C to form thioether benzaldehyde intermediates.
- Perform reductive amination of the thioether benzaldehyde with (R)-(+)-alpha-methylbenzylamine using sodium borohydride in methanol.
- React the resulting amine intermediate with phosphorus trichloride and a phenolic compound (such as binaphthol) in THF to finalize the chiral phosphine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this ligand technology presents a compelling value proposition centered on reliability and cost efficiency. The reliance on bulk commodity chemicals such as 2-fluorobenzaldehyde, potassium carbonate, and common thiophenols means that raw material sourcing is not subject to the volatility associated with specialized chiral reagents. This stability in the supply base significantly reduces the risk of production delays caused by material shortages. Moreover, the synthetic route avoids the use of precious metal catalysts in the ligand synthesis itself (using iron or base metal catalysis is not required here, just standard organic synthesis), reserving the expensive palladium solely for the final application where its loading can be minimized due to the high activity of the ligand. This strategic allocation of resources leads to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The streamlined three-step synthesis eliminates the need for complex protection-deprotection strategies often seen in traditional ligand synthesis. By utilizing direct nucleophilic substitution and reductive amination, the process reduces the number of unit operations, which directly correlates to lower labor and utility costs. The high yields reported in the patent examples indicate minimal material loss, enhancing the atom economy of the process. Furthermore, the workup procedures involve simple filtration and liquid-liquid extraction, avoiding expensive chromatographic separations on a large scale. These factors combine to create a manufacturing process that is inherently lean, driving down the cost of goods sold (COGS) for the final chiral intermediates produced using these ligands.
- Enhanced Supply Chain Reliability: The modular nature of the ligand library means that if a specific substituent becomes unavailable, alternative variants can be synthesized with minimal process changes, ensuring continuity of supply. The starting materials are widely produced by multiple global suppliers, mitigating the risk of single-source dependency. Additionally, the robustness of the reaction conditions (e.g., tolerance to moderate temperatures and standard solvents) reduces the likelihood of batch failures due to minor process deviations. This reliability is critical for maintaining just-in-time inventory levels and meeting the tight delivery schedules demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process utilizes solvents like DMSO, methanol, and THF, which are well-understood in terms of recovery and recycling, facilitating compliance with environmental regulations. The absence of highly toxic or persistent organic pollutants in the reagent list simplifies waste treatment protocols. From a scalability perspective, the exothermic profiles of the reactions are manageable, allowing for safe scale-up in large reactors without the need for specialized cooling infrastructure. This ease of scale-up ensures that the technology can meet the demands of commercial-scale production, from pilot plant batches to multi-ton annual campaigns, without requiring significant capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this catalytic system into their existing production workflows.
Q: What is the primary advantage of the thioether-phosphine ligand library described in CN113980053A?
A: The primary advantage is the modular construction allowing for rapid diversification of the ligand library using readily available thiophenols and phenols, facilitating the optimization of stereoselectivity for specific substrates without complex de novo synthesis.
Q: How does this catalytic system improve upon traditional asymmetric allylic alkylation methods?
A: This system utilizes a robust palladium complex that operates under relatively mild conditions, avoiding the need for extremely low temperatures or exotic reagents often required by conventional chiral phosphine ligands, thereby simplifying process scalability.
Q: Can these ligands be scaled for industrial pharmaceutical intermediate production?
A: Yes, the synthesis relies on commodity chemicals like 2-fluorobenzaldehyde and standard solvents like DMSO and THF, with straightforward workup procedures involving filtration and extraction, making it highly suitable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Thioether-Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like those described in CN113980053A for the synthesis of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is smooth and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of specific ligand variants or the development of a complete catalytic process for your target molecule, our CDMO capabilities are designed to meet your exacting standards.
We invite you to contact our technical procurement team to discuss how we can support your project with a Customized Cost-Saving Analysis. By leveraging our supply chain expertise and manufacturing prowess, we can help you optimize your production costs while ensuring the highest quality. Please reach out to request specific COA data for our available ligand libraries or to schedule a consultation for route feasibility assessments. Let us be your partner in turning complex chemical challenges into commercial successes.
