Advanced Synthesis Of Pyrido Thioazepines For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
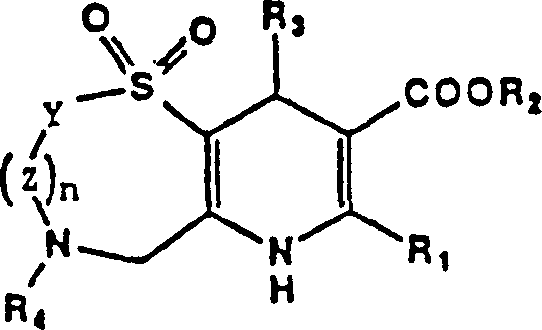
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve with the discovery of novel calcium channel antagonists, as detailed in the seminal patent CN1034077C. This intellectual property discloses a sophisticated class of pyrido[2,3-f][1,4]thioazepines and pyrido[3,2-b][1,5]benzothiazepines that demonstrate potent smooth muscle relaxation properties. These compounds are specifically engineered to inhibit calcium ion influx, offering significant therapeutic potential for treating hypertension, angina, congestive heart failure, and asthma. The technical breakthrough lies not only in the biological efficacy but also in the robust synthetic methodologies provided, which enable the production of these complex heterocyclic systems with high structural fidelity. For R&D directors and procurement specialists, understanding the nuances of this synthesis is critical for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity calcium channel antagonists. The patent outlines specific reaction pathways that avoid the pitfalls of earlier dihydropyridine syntheses, introducing a sulfone-containing scaffold that enhances pharmacological stability and activity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of calcium antagonists often relied on dihydropyridine scaffolds with acyclic sulfone substituents, as seen in prior art such as US4,285,955. However, these conventional methods frequently suffered from limitations regarding chemical stability and the complexity of introducing the sulfone moiety at later stages of the synthesis. Traditional routes often required harsh conditions that could compromise the integrity of sensitive functional groups, leading to lower overall yields and difficult purification processes. Furthermore, the lack of a fused ring system in some earlier analogs resulted in reduced conformational rigidity, which negatively impacted binding affinity to calcium channels. The reliance on multiple protection and deprotection steps in older methodologies also increased the operational cost and extended the manufacturing timeline, creating bottlenecks for supply chain heads looking for cost reduction in cardiovascular drug manufacturing. These inefficiencies underscore the need for a more streamlined approach that integrates the sulfone functionality early in the synthetic sequence.
The Novel Approach
The methodology presented in CN1034077C introduces a paradigm shift by constructing the sulfo-azepine ring system prior to the final condensation steps. This novel approach utilizes a Boc-protected cystamine derivative as a starting material, which undergoes a controlled cyclization with epichlorohydrin to form the core seven-membered ring. By establishing the sulfone-containing scaffold early, the process ensures that the oxidation state is correctly managed before the introduction of the dihydropyridine fusion. This strategy significantly simplifies the downstream processing, as the key intermediates are more stable and easier to purify compared to their acyclic counterparts. The use of specific oxidants like MCPBA and Jones reagent allows for precise control over the oxidation of the sulfur atom to the 1,1-dioxo state, which is crucial for the biological activity. This refined synthetic logic not only enhances the purity of the final API intermediate but also facilitates the commercial scale-up of complex pharmaceutical intermediates by reducing the number of unit operations required.
Mechanistic Insights into Sulfo-Azepine Ring Formation and Oxidation
The core of this synthetic innovation lies in the formation of the sulfo-azepine alcohol intermediate, which serves as the precursor to the active ketone. The mechanism begins with the nucleophilic attack of the protected amino thiol on epichlorohydrin, followed by an intramolecular cyclization mediated by a strong base such as sodium hydride. This step is critical as it forms the seven-membered heterocyclic ring with high regioselectivity, avoiding the formation of unwanted polymeric byproducts. The subsequent oxidation sequence is equally vital; the initial treatment with meta-chloroperbenzoic acid (MCPBA) converts the sulfide to the sulfoxide, while the follow-up oxidation with Jones reagent completes the transformation to the sulfone. This two-step oxidation protocol is superior to single-step methods as it prevents over-oxidation of other sensitive sites on the molecule, such as the ester or amine functionalities. The resulting 1,1-dioxo-sulfo-azepine ketone is a highly electrophilic species, perfectly poised for the Hantzsch-like condensation with aldehydes and amino acrylates that follows.
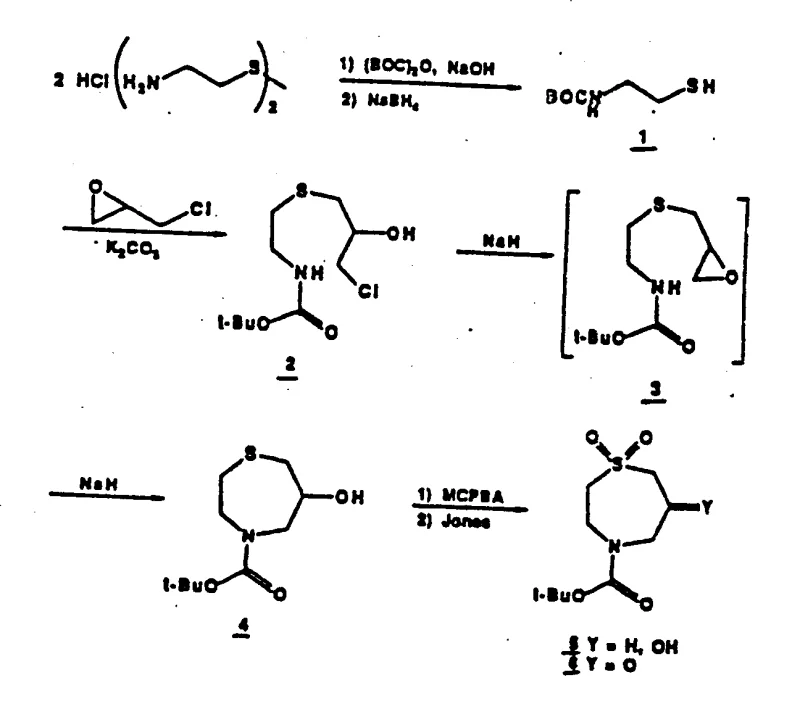
Impurity control is inherently built into this mechanistic pathway through the use of crystalline intermediates and specific reagent stoichiometry. The Boc protecting group plays a dual role: it prevents self-polymerization of the amine during the cyclization step and provides a handle for easy removal under acidic conditions later in the synthesis. By isolating the chloroethanol intermediate or driving the reaction directly to the cyclic alcohol depending on the base equivalents used, chemists can tune the process to minimize side reactions. The final condensation step to form the dihydropyridine ring is conducted under reflux conditions, which drives the equilibrium towards the desired fused product while allowing volatile byproducts to be removed. This rigorous control over reaction parameters ensures that the impurity profile remains within stringent purity specifications, a key requirement for regulatory approval in the pharmaceutical sector. The ability to produce optically active enantiomers further adds value, as specific isomers often exhibit superior pharmacokinetic properties.
How to Synthesize Pyrido Thioazepine Efficiently
The synthesis of these high-value cardiovascular intermediates requires precise adherence to the reaction conditions outlined in the patent to ensure reproducibility and yield. The process begins with the preparation of the Boc-protected cystamine, followed by reduction to the mercaptan, which must be handled under inert atmosphere to prevent disulfide formation. The cyclization step requires careful temperature control, typically starting at 0°C and warming to room temperature to manage the exotherm associated with the base addition. Subsequent oxidation steps must be monitored closely, typically via TLC or HPLC, to ensure complete conversion to the ketone without degrading the sensitive ester groups. The final condensation with the appropriate aldehyde and amino crotonate is the culmination of the sequence, yielding the target pyrido thioazepine scaffold. For detailed operational parameters, safety data, and specific workup procedures, the standardized synthesis steps are provided below.
- Preparation of Boc-protected cystamine and subsequent reduction to N-(tert-butoxycarbonyl)-2-aminoethyl mercaptan.
- Reaction with epichlorohydrin followed by base-mediated cyclization to form the sulfo-azepine alcohol intermediate.
- Oxidation using MCPBA and Jones reagent to yield the key sulfo-azepine ketone, followed by condensation with aldehydes and amino acrylates.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in CN1034077C offers substantial advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of complex transition metal catalysts, which are often required in alternative cross-coupling strategies, significantly reduces the raw material costs and simplifies the removal of heavy metal residues. This simplification translates directly into lower processing costs and reduced waste disposal liabilities, contributing to substantial cost savings over the lifecycle of the product. Furthermore, the use of commodity chemicals such as epichlorohydrin, sodium hydride, and common oxidants ensures that the supply chain remains robust and less susceptible to fluctuations in the availability of exotic reagents. The high yields reported in the examples, particularly in the cyclization and oxidation steps, indicate a material-efficient process that maximizes output per batch, thereby enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway minimizes the number of isolation and purification steps required, which directly lowers labor and solvent consumption costs. By avoiding the use of expensive chiral catalysts or precious metals, the process achieves a more economical cost of goods sold (COGS) structure. The ability to recycle solvents like ethanol and toluene, as indicated in the workup procedures, further contributes to the overall economic efficiency of the manufacturing process. Additionally, the high purity of the intermediates reduces the need for extensive chromatographic purification, allowing for more cost-effective crystallization-based purification methods to be employed at scale.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions, which tolerate standard industrial equipment and conditions, facilitates technology transfer between manufacturing sites. This flexibility is crucial for maintaining supply continuity, especially in the event of regional disruptions or capacity constraints at a single facility. The well-defined process parameters also reduce the risk of batch failures, ensuring a consistent supply of high-quality intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily adaptable from laboratory to pilot and commercial scale. The waste streams generated are primarily aqueous and organic solvents that can be treated using standard environmental protocols, ensuring compliance with increasingly stringent regulatory requirements. The absence of persistent organic pollutants or highly toxic heavy metals in the waste stream simplifies the environmental permitting process and reduces the long-term environmental liability associated with the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrido thioazepine derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating these intermediates into your existing drug development pipelines. For further technical discussions or custom synthesis requests, please refer to the contact information provided at the end of this report.
Q: What is the primary therapeutic application of the pyrido thioazepine compounds described in CN1034077C?
A: The compounds function as calcium channel antagonists, exhibiting cardiovascular, antiasthmatic, and anti-bronchoconstrictive activity, suitable for treating hypertension and angina.
Q: How does the novel oxidation process improve the synthesis of the key intermediate?
A: The process utilizes a sequential oxidation strategy involving MCPBA and Jones reagent to efficiently convert the sulfo-azepine alcohol into the critical 1,1-dioxo-sulfo-azepine ketone intermediate with high purity.
Q: Are the synthetic routes described scalable for commercial production?
A: Yes, the patent outlines robust reaction conditions using standard reagents like sodium hydride and epichlorohydrin, which are amenable to scale-up for reducing lead time for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrido Thioazepine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN1034077C into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical sector, and our facilities are equipped to meet the highest international standards. By partnering with us, you gain access to a supply chain that is not only reliable but also optimized for cost efficiency and regulatory compliance, allowing you to focus on your core drug development activities.
We invite you to engage with our technical procurement team to discuss your specific requirements for high-purity pharmaceutical intermediates. We are prepared to provide a Customized Cost-Saving Analysis tailored to your project's volume and timeline constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your next breakthrough in cardiovascular therapeutics.
