Advanced Synthesis of Bioactive Alkaloid Intermediates for Pharmaceutical Development
Advanced Synthesis of Bioactive Alkaloid Intermediates for Pharmaceutical Development
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds that serve as critical building blocks for new drug candidates. Patent CN101220035A introduces a significant advancement in this domain by detailing the preparation of a novel class of alkaloids derived from Mansonone analogs. These compounds, characterized by their intricate fused ring systems, display potent antitumor and antibacterial properties, making them highly valuable as pharmaceutical intermediates. The core innovation lies in the direct condensation of Mansonone-like quinones with various amines, including primary amines, hydroxylamine, and diamines, to construct complex nitrogen-containing heterocycles efficiently. This approach bypasses many of the tedious multi-step sequences traditionally required to assemble such dense molecular architectures.
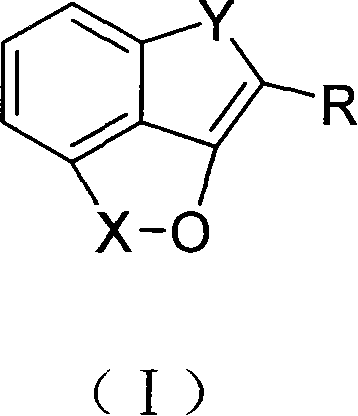
From a structural perspective, the versatility of this synthetic platform is evident in the diverse substituents accommodated within the general formula. The ability to introduce various functional groups at the R, X, and Y positions allows medicinal chemists to fine-tune the physicochemical and biological properties of the final active pharmaceutical ingredients (APIs). For instance, the Y moiety can be modified to include phenazine, quinoxaline, or oxazole rings, each imparting distinct electronic and steric characteristics to the molecule. This modularity is crucial for lead optimization campaigns where structure-activity relationship (SAR) studies demand rapid access to a wide array of analogs. Consequently, this technology offers a reliable alkaloid intermediate supplier solution for companies developing next-generation antimicrobial and anticancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex fused heterocyclic systems like isochromeno-phenazines or isochromeno-quinoxalines has posed significant challenges in organic synthesis. Traditional routes often rely on harsh reaction conditions, expensive transition metal catalysts, or lengthy protection-deprotection strategies that drastically reduce overall throughput. In many prior art methods, forming the critical carbon-nitrogen bonds within a crowded steric environment requires elevated temperatures and prolonged reaction times, which can lead to decomposition of sensitive quinone precursors. Furthermore, achieving regioselectivity in the functionalization of the quinone core is notoriously difficult, often resulting in complex mixtures of isomers that are arduous to separate. These inefficiencies translate directly into higher production costs and extended timelines for cost reduction in pharmaceutical manufacturing, creating bottlenecks for supply chain managers who require consistent, high-volume delivery of key intermediates.
The Novel Approach
The methodology described in the patent represents a paradigm shift by leveraging the inherent reactivity of the o-quinone system towards nucleophilic attack by amines. Instead of forcing bond formation through external energy inputs, this process utilizes the thermodynamic drive towards forming stable, conjugated aromatic systems. By reacting Mansonone analogs directly with diamines or primary amines, the synthesis achieves cyclization and aromatization in a single operational step under mild conditions. This streamlining eliminates the need for intermediate isolation and purification steps that typically plague conventional syntheses. The result is a process that not only simplifies the workflow but also enhances the overall atom economy. For procurement teams, this translates to a more predictable supply of high-purity alkaloid intermediates with reduced risk of batch-to-batch variability caused by complex processing parameters.
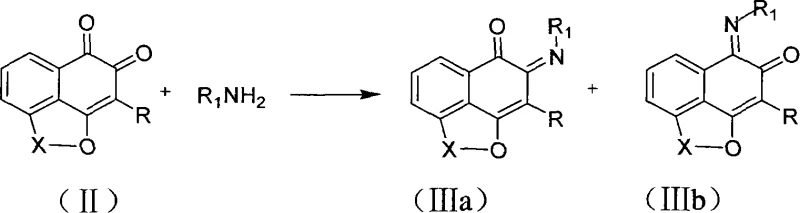
Mechanistic Insights into Amine-Quinone Condensation and Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors aiming to scale the process effectively. The reaction is not merely a simple dehydration condensation; rather, it involves a sophisticated sequence of nucleophilic addition, double bond rearrangement, and oxidative aromatization. When amines possessing an alpha-methylene group react with the quinone, the initial adduct undergoes a rearrangement that facilitates ring closure. This is followed by an oxidation step, often spontaneous in the presence of air or facilitated by the quinone itself, to establish the fully conjugated heterocyclic system. The driving force behind this transformation is the formation of a large, stable conjugated pi-system, which thermodynamically favors the bis-imine or fused ring products over mono-imine intermediates.
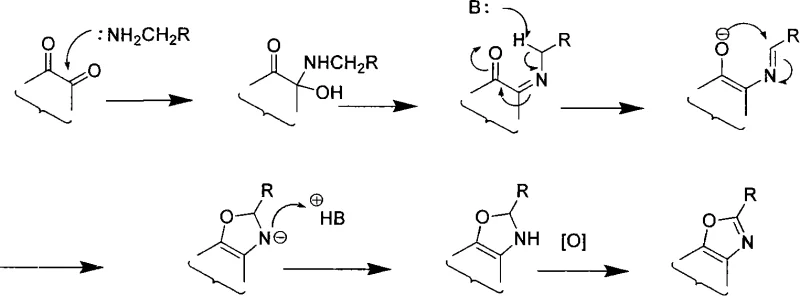
Impurity control is inherently managed by the thermodynamic stability of the final products. Since the reaction pathway leads to highly conjugated, aromatic structures, side reactions that would produce non-conjugated or partially saturated byproducts are disfavored. The patent data indicates that even when yields vary, the crude products are often sufficiently pure to be isolated by simple filtration or basic chromatography. This mechanistic robustness ensures that the commercial scale-up of complex alkaloid intermediates remains feasible without requiring exotic purification technologies. The ability to predict the major product based on thermodynamic stability allows process chemists to optimize reaction times and stoichiometry to maximize the yield of the desired isomer, thereby ensuring consistent quality for downstream applications.
How to Synthesize Isochromeno-Phenazine Alkaloids Efficiently
The practical execution of this synthesis is remarkably straightforward, making it accessible for both laboratory-scale discovery and industrial production. The process typically involves dissolving the Mansonone starting material in a lower alcohol solvent like methanol or ethanol, followed by the addition of the amine component. The reaction proceeds smoothly at ambient temperatures, eliminating the need for specialized heating or cooling equipment. Detailed standard operating procedures for specific derivatives, including stoichiometry and workup protocols, are essential for reproducibility.
- Dissolve Mansonone analog starting material (e.g., WJ-1) in an organic solvent such as methanol or ethanol.
- Add the appropriate amine reagent, such as o-phenylenediamine or ethylenediamine, to the reaction mixture at room temperature.
- Stir the reaction until completion, monitored by TLC, then filter and purify the resulting solid alkaloid product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this synthetic route offers tangible logistical and economic benefits. The reliance on readily available starting materials, such as substituted Mansonone analogs and common diamines like ethylenediamine or o-phenylenediamine, mitigates the risk of raw material shortages. These precursors are commodity chemicals or easily synthesized in bulk, ensuring a stable supply base. Moreover, the use of benign solvents like ethanol and methanol simplifies waste management and reduces the environmental footprint of the manufacturing process, aligning with increasingly stringent global regulatory standards for green chemistry.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the ability to run reactions at room temperature significantly lowers the operational expenditure associated with energy consumption and reagent costs. By avoiding multi-step sequences and complex purification requirements, the overall cost of goods sold (COGS) for these intermediates is substantially reduced. This efficiency allows manufacturers to offer competitive pricing without compromising on the quality or purity of the final product, providing a clear economic advantage in the sourcing of critical pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The simplicity of the reaction protocol reduces the likelihood of process deviations and batch failures. With fewer unit operations and less sensitivity to minor fluctuations in reaction parameters, the manufacturing process becomes more robust and predictable. This reliability is crucial for reducing lead time for high-purity alkaloid intermediates, ensuring that pharmaceutical clients receive their materials on schedule to maintain their own production timelines. The scalability of the process from gram to kilogram quantities further supports uninterrupted supply chains.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous alcohol streams that are easier to treat than heavy metal-contaminated waste from catalytic processes. This environmental compatibility facilitates easier permitting and compliance with local environmental regulations. Additionally, the high atom economy of the condensation reaction means that less raw material is wasted, contributing to a more sustainable manufacturing model that appeals to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these alkaloid derivatives. Understanding these aspects helps stakeholders make informed decisions about integrating these intermediates into their development pipelines.
Q: What are the primary biological activities of these synthesized alkaloids?
A: The synthesized alkaloids exhibit significant antibacterial activity against Gram-positive bacteria including MRSA, and demonstrate cytotoxic activity against tumor cell lines such as K562 and MCF-7.
Q: What solvents are preferred for this condensation reaction?
A: Alcohols are the preferred solvents, with methanol and ethanol providing the best results for dissolving reactants and facilitating the condensation without interfering with the amine-quinone reaction.
Q: Is high temperature required for the cyclization process?
A: No, the reaction preferably proceeds at temperatures between 5°C and 35°C, with room temperature being optimal, which significantly reduces energy consumption compared to high-temperature protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkaloid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material and commercial API manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkaloid intermediate meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality assurance ensures that your drug development programs proceed without interruption due to material supply issues.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for the production of these valuable bioactive compounds.
