Advanced Synthesis of Carbon-14 Double-Labeled Furmonertinib for High-Sensitivity Tracer Applications
Advanced Synthesis of Carbon-14 Double-Labeled Furmonertinib for High-Sensitivity Tracer Applications
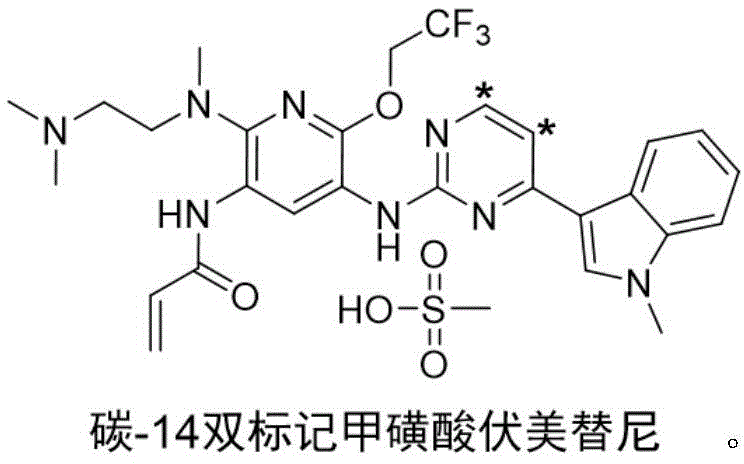
The rapid evolution of third-generation epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) has necessitated equally sophisticated analytical tools for pharmacokinetic and metabolic profiling. Patent CN113896716A introduces a groundbreaking methodology for the synthesis of radioisotope carbon-14 double-labeled furmonertinib mesylate, addressing the critical need for high-specific-activity tracers in oncology research. This innovation specifically targets the limitations of single-site labeling, offering a theoretical maximum specific activity of 124.8 mCi/mmol, which is twice that of conventional single-labeled analogues. By strategically incorporating carbon-14 isotopes at the 5-C and 6-C positions of the pyrimidine ring, this process ensures that the radioactive signal remains intact during metabolic displacement studies, thereby providing unparalleled data accuracy for drug development teams investigating resistance mechanisms in non-small cell lung cancer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional radiolabeling strategies for complex kinase inhibitors often rely on single-site incorporation, typically yielding a specific activity around 50 to 62.4 mCi/mmol. In the context of furmonertinib, where therapeutic doses are relatively low and metabolic pathways are complex, this lower specific activity presents significant analytical challenges. When conducting tracer tests in biological systems with complex compositions, the signal-to-noise ratio often becomes insufficient to detect trace metabolites accurately without increasing the administered dosage, which can alter the pharmacokinetic profile. Furthermore, labeling unstable side chains can lead to the loss of the radioactive tag during metabolism, resulting in misleading data regarding the distribution and clearance of the active pharmaceutical ingredient. These deficiencies hinder the ability of R&D directors to fully understand the ADME properties required for regulatory approval.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by employing a dual-labeling strategy on the metabolically robust pyrimidine core. By utilizing [2,3-14C2]Propiolic acid as the primary isotope raw material, the synthesis constructs the heterocyclic ring with two radioactive carbon atoms inherently integrated into the scaffold. This approach not only doubles the specific activity but also anchors the label in a region of the molecule known for its chemical and metabolic stability. The process involves a sequence of ring closing, hydroxyl chlorination, coupling, substitution, reduction, condensation, and salt formation, all optimized to preserve the isotopic integrity. This results in a tracer capable of meeting the stringent detection limits of modern radioactivity measuring instruments, facilitating precise basic research into the drug's behavior within the human body.
Mechanistic Insights into Pyrimidine Ring Construction and Functionalization
The core of this synthetic achievement lies in the efficient construction of the labeled pyrimidine ring followed by precise functionalization. The process initiates with the cyclization of [2,3-14C2]Propiolic acid with urea in polyphosphoric acid at temperatures between 90-130°C, forming the labeled uracil intermediate with high radiochemical yield. This is followed by chlorination using phosphorus oxychloride and DIPEA to generate the reactive 2,4-dichloro species. A critical step involves the Lewis acid-catalyzed coupling with N-methylindole, which establishes the crucial bond between the pyrimidine and indole moieties. The subsequent palladium-catalyzed amination connects the labeled core to the complex pyridine side chain, demonstrating the compatibility of radioactive intermediates with transition metal catalysis. Each step is meticulously designed to minimize isotopic dilution and maximize the retention of the dual labels.
Impurity control is paramount in radiochemical synthesis, particularly when dealing with high-value isotopes. The patent details rigorous purification protocols, including column chromatography and preparative high-performance liquid chromatography (HPLC), to ensure the final product achieves greater than 99% radiochemical purity. The choice of reducing agents, such as iron or zinc powder for the nitro group reduction, is balanced against the need to avoid side reactions that could scramble the isotopic label. Furthermore, the final acryloylation step is conducted at low temperatures (0-30°C) to prevent polymerization of the acrylamide group, a common degradation pathway for this class of inhibitors. This attention to mechanistic detail ensures that the resulting tracer is not only highly active but also chemically stable enough for long-term storage and distribution to global research facilities.
How to Synthesize Carbon-14 Double-Labeled Furmonertinib Efficiently
The synthesis of this high-value radiotracer requires a disciplined approach to reaction conditions and purification to maintain isotopic fidelity. The process is divided into seven distinct operational stages, beginning with the formation of the labeled heterocycle and concluding with salt crystallization. Operators must strictly adhere to inert gas protection and temperature controls to prevent the loss of volatile radioactive intermediates. The following guide outlines the critical workflow derived from the patent examples, emphasizing the transition from raw isotope materials to the final mesylate salt. For detailed standard operating procedures and safety protocols regarding radioactive handling, please refer to the specific technical documentation provided below.
- Cyclization of [2,3-14C2]Propiolic acid with urea in polyphosphoric acid to form labeled Uracil.
- Chlorination and subsequent coupling with N-methylindole to construct the pyrimidine-indole core.
- Palladium-catalyzed amination, nitro reduction, acryloylation, and final salt formation with methanesulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this double-labeling technology translates into tangible strategic advantages beyond mere technical specifications. The robust nature of the synthetic route, which relies on established organic transformations rather than exotic or proprietary biocatalysts, significantly de-risks the supply chain. By utilizing commercially available precursors like propiolic acid derivatives and standard coupling reagents, the dependency on single-source specialty vendors is minimized. This diversification of the raw material base enhances supply continuity, ensuring that critical radiotracer batches can be produced consistently to support ongoing clinical and preclinical trials without interruption. The reliability of the synthesis path directly correlates to the reliability of the data generated by our partners.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the high specific activity of the final product. Because the double-labeling technique effectively doubles the signal strength per mole of compound, researchers can achieve the same detection sensitivity with potentially lower quantities of the radioactive substance, optimizing the usage of expensive carbon-14 raw materials. Furthermore, the elimination of complex metabolic instability issues reduces the need for repetitive synthesis runs caused by failed tracer studies. The streamlined purification steps, utilizing standard chromatography rather than specialized isotopic separation techniques, further contribute to substantial cost savings in the overall manufacturing lifecycle, making high-quality tracers more accessible for broad-scale research applications.
- Enhanced Supply Chain Reliability: The synthetic route is designed with scalability in mind, utilizing solvents and reagents that are staples in the fine chemical industry, such as toluene, ethanol, and acetone. This compatibility with standard chemical infrastructure means that production can be rapidly scaled or shifted between facilities without requiring bespoke equipment modifications. The robustness of the intermediates, particularly the stability of the labeled pyrimidine core, allows for safer storage and transport of key building blocks, mitigating the risks associated with the decay of short-lived isotopes or the degradation of sensitive intermediates. This flexibility ensures a steady flow of materials to meet the dynamic demands of global pharmaceutical R&D pipelines.
- Scalability and Environmental Compliance: From an environmental and operational standpoint, the process avoids the use of highly toxic heavy metals in stoichiometric quantities, relying instead on catalytic amounts of palladium which can be recovered and recycled. The waste streams generated are consistent with standard organic synthesis byproducts, simplifying the disposal and treatment protocols required for regulatory compliance. The ability to perform the synthesis in standard reactor setups, from small-scale radiochemical hoods to larger pilot plants, demonstrates the inherent scalability of the technology. This ensures that as the demand for furmonertinib metabolic data grows, the supply of the corresponding radiolabeled standards can expand in parallel without encountering significant technical bottlenecks or environmental hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of carbon-14 double-labeled furmonertinib mesylate. These insights are derived directly from the patented synthesis method and are intended to clarify the value proposition for research and procurement stakeholders. Understanding the specific advantages of this labeling strategy is essential for planning effective ADME studies and budgeting for radiotracer acquisition. We encourage technical teams to review these points to align their experimental designs with the capabilities of this advanced synthetic route.
Q: Why is double-labeling preferred for Furmonertinib metabolic studies?
A: Double-labeling at the 5-C and 6-C positions of the pyrimidine ring theoretically doubles the specific activity to 124.8 mCi/mmol, ensuring detection limits are met even in complex biological matrices without increasing drug dosage.
Q: What represents the key stability advantage of this labeling position?
A: The pyrimidine ring is metabolically stable compared to side chains, preventing the loss of the radioactive signal during metabolic displacement and ensuring accurate tracking of the parent drug molecule.
Q: Is this synthesis route scalable for commercial radiotracer production?
A: Yes, the route utilizes standard organic transformations such as Buchwald-Hartwig amination and classical acylation, which are well-suited for scale-up in specialized radiochemical facilities with appropriate shielding.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furmonertinib Mesylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation oncology therapeutics depends on the availability of high-integrity analytical standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require milligram quantities for initial screening or gram scales for comprehensive metabolic profiling, our capacity meets your needs. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle radiochemical analysis, guaranteeing that every batch of furmonertinib mesylate intermediate or labeled derivative meets the highest industry standards for specific activity and chemical purity.
We invite you to collaborate with us to optimize your supply chain for critical research materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, helping you identify efficiencies in your sourcing strategy. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can accelerate your drug development timeline while ensuring full regulatory compliance and data reliability.
