Scalable Production of Brivaracetam Isomer (2S, 4S) via Advanced Chemical Resolution Technology
The pharmaceutical industry continuously seeks robust methodologies for synthesizing complex chiral intermediates, particularly for antiepileptic agents like Brivaracetam. Patent CN109593055B introduces a transformative preparation method for the Brivaracetam isomer (2S, 4S), addressing critical bottlenecks in existing synthetic routes. This technology leverages a sophisticated chemical resolution strategy that bypasses the traditional reliance on preparative chromatography, a technique often plagued by high operational costs and scalability limitations. By utilizing specific chiral resolving agents such as L-(-)-dibenzoyltartaric acid, the process achieves efficient separation of stereoisomers under mild reaction conditions. This innovation represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status, as it aligns perfectly with the industry's demand for cost-effective and environmentally sustainable production pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Brivaracetam isomers has been hindered by rigorous process requirements that inflate manufacturing expenses and complicate scale-up. Prior art methods, such as those utilizing propyl Grignard reagents, demand strictly anhydrous and anaerobic environments, necessitating specialized equipment and increasing safety risks. Furthermore, many established routes rely heavily on preparative chromatographic separation to isolate the desired stereoisomer from racemic mixtures or byproduct isomers. Chromatography is inherently inefficient for large-scale production due to high solvent consumption, significant silica gel waste, and batch-to-batch variability. These factors collectively result in elevated production costs and extended lead times, making it challenging for procurement teams to maintain consistent supply chains for high-purity pharmaceutical intermediates without incurring substantial financial burdens.
The Novel Approach
The methodology disclosed in CN109593055B offers a compelling alternative by replacing chromatographic purification with a classical yet highly optimized chemical resolution process. This approach involves the formation of diastereomeric salts between the racemic intermediate and a chiral acid resolving agent. The process capitalizes on the differential solubility of these salts in alcoholic solvents, allowing the desired isomer to crystallize out while impurities remain in the solution. 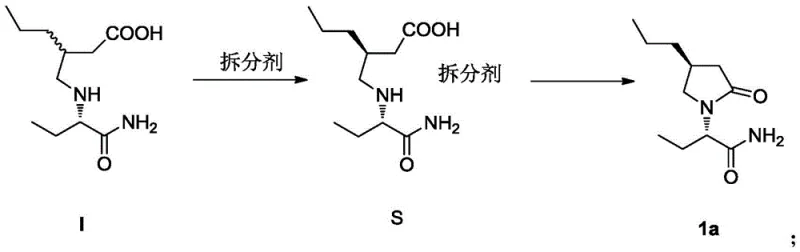 As illustrated in the reaction scheme, the racemic Compound I is treated with a resolving agent to yield the specific salt Form S, which is subsequently liberated to provide the target (2S, 4S) isomer. This shift from physical separation (chromatography) to chemical separation (crystallization) drastically simplifies the workflow, reduces solvent usage, and enhances the overall throughput, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
As illustrated in the reaction scheme, the racemic Compound I is treated with a resolving agent to yield the specific salt Form S, which is subsequently liberated to provide the target (2S, 4S) isomer. This shift from physical separation (chromatography) to chemical separation (crystallization) drastically simplifies the workflow, reduces solvent usage, and enhances the overall throughput, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Chiral Resolution and Hydrogenation
The core of this synthetic strategy lies in the precise control of stereochemistry through salt formation and catalytic reduction. The process begins with the synthesis of a key furanone precursor via the condensation of glyoxylic acid and n-valeraldehyde, catalyzed by morpholine. 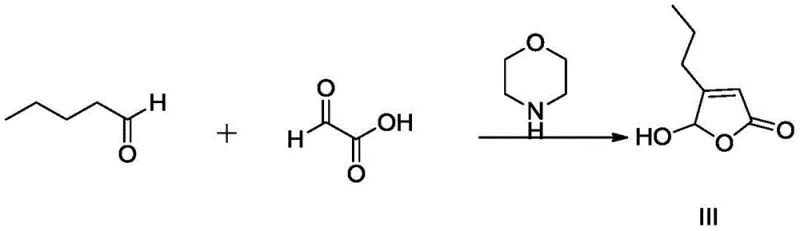 This precursor is then reacted with (S)-2-aminobutanamide to form a pyrrolidinyl intermediate, which undergoes catalytic hydrogenation using Pd/C under hydrogen pressure to generate the racemic amino acid derivative (Compound I). The critical mechanistic step occurs during the resolution phase, where the carboxylic acid group of Compound I interacts with the chiral resolving agent. The spatial arrangement of the (2S, 4S) isomer allows for a more stable crystal lattice formation with agents like L-(-)-dibenzoyltartaric acid compared to its enantiomers. This thermodynamic preference drives the selective precipitation of the target salt, effectively purifying the mixture without the need for complex separation media.
This precursor is then reacted with (S)-2-aminobutanamide to form a pyrrolidinyl intermediate, which undergoes catalytic hydrogenation using Pd/C under hydrogen pressure to generate the racemic amino acid derivative (Compound I). The critical mechanistic step occurs during the resolution phase, where the carboxylic acid group of Compound I interacts with the chiral resolving agent. The spatial arrangement of the (2S, 4S) isomer allows for a more stable crystal lattice formation with agents like L-(-)-dibenzoyltartaric acid compared to its enantiomers. This thermodynamic preference drives the selective precipitation of the target salt, effectively purifying the mixture without the need for complex separation media.
Impurity control is meticulously managed through iterative recrystallization steps. The patent data indicates that multiple recrystallizations from ether solvents, such as isopropyl ether, further enhance the optical purity of the final product. This multi-stage purification ensures that trace amounts of the unwanted (2R, 4R) or other diastereomeric impurities are reduced to negligible levels. The robustness of this mechanism allows for consistent production of material meeting stringent purity specifications, a crucial factor for R&D directors evaluating the feasibility of scaling this pathway for commercial API production. The ability to monitor reaction progress via standard analytical techniques like HPLC and TLC ensures tight process control throughout the synthesis.
How to Synthesize Brivaracetam Isomer (2S, 4S) Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing the target isomer with high fidelity. It starts with the preparation of the furanone backbone, followed by amidation and hydrogenation to establish the core scaffold. The pivotal resolution step requires careful selection of the resolving agent and solvent system to maximize yield and enantiomeric excess.
- Synthesize the precursor 5-hydroxy-4-n-propyl-furan-2-one via condensation of glyoxylic acid and n-valeraldehyde.
- React the precursor with (S)-2-aminobutanamide followed by catalytic hydrogenation to obtain the racemic intermediate Compound I.
- Perform chiral resolution using L-(-)-dibenzoyltartaric acid in ethanol to isolate the desired (2S, 4S) isomer salt, followed by liberation and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution-based technology translates into tangible operational benefits that extend beyond simple yield metrics. By eliminating the dependency on preparative chromatography, manufacturers can significantly reduce the consumption of high-grade solvents and disposable chromatography media, leading to a drastic simplification of the waste management profile. This reduction in material intensity directly correlates with lower variable costs per kilogram of produced intermediate. Furthermore, the use of commercially available and inexpensive starting materials, such as n-valeraldehyde and glyoxylic acid, ensures a stable and resilient supply chain that is less susceptible to market volatility compared to routes requiring specialized organometallic reagents.
- Cost Reduction in Manufacturing: The transition from chromatographic separation to crystallization-based resolution removes one of the most expensive unit operations in fine chemical synthesis. Chromatography typically involves high capital expenditure for columns and significant ongoing costs for silica gel and solvent recovery. By replacing this with crystallization, the process utilizes standard reactor vessels and filtration equipment, which are ubiquitous in chemical plants. This structural change in the process design facilitates substantial cost savings, allowing for more competitive pricing strategies in the global market for antiepileptic drug intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than sensitive reagents like Grignard reagents enhances the robustness of the supply chain. Grignard reactions require strict exclusion of moisture and oxygen, often necessitating dedicated production lines and increasing the risk of batch failures. In contrast, the resolution and hydrogenation steps described in this patent operate under much milder and more forgiving conditions. This operational flexibility reduces the likelihood of production delays and ensures a more consistent delivery schedule, which is vital for maintaining the continuity of downstream API manufacturing.
- Scalability and Environmental Compliance: The proposed method is inherently scalable, as crystallization is a standard unit operation that scales linearly from laboratory to pilot to commercial plant sizes without the engineering complexities associated with scaling chromatography. Additionally, the reduction in solvent volume and the avoidance of silica gel waste contribute to a greener manufacturing process. This alignment with environmental compliance standards reduces the regulatory burden and disposal costs, making the process more sustainable and attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented resolution technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the practical application of the method.
Q: Why is chemical resolution preferred over chromatography for Brivaracetam isomers?
A: Chemical resolution eliminates the need for expensive preparative chromatography columns and reduces solvent waste, significantly lowering production costs and improving scalability for industrial manufacturing.
Q: What resolving agents are effective for this synthesis?
A: The patent identifies L-(-)-dibenzoyltartaric acid as the preferred resolving agent, offering superior separation efficiency compared to camphorsulfonic acid or tartaric acid variants.
Q: What is the optical purity achievable with this method?
A: Through optimized recrystallization steps, the process achieves high optical purity, with HPLC analysis confirming purity levels around 97.4% for the final (2S, 4S) isomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Isomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the chemical resolution of Brivaracetam isomers can be successfully transferred to industrial scale. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of intermediate meets the highest quality standards required for FDA-regulated markets.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage you to contact us for specific COA data and route feasibility assessments to validate the suitability of this technology for your upcoming projects.
