Revolutionizing Chiral Amine Production: Scalable Synthesis of (R)-Rivastigmine Intermediates
The pharmaceutical industry's relentless pursuit of efficient chiral synthesis has found a significant breakthrough in patent CN110668976A, which details a novel methodology for producing (R)-Rivastigmine and its critical optical isomer intermediates. This technology addresses the longstanding challenges associated with the manufacturing of Alzheimer's disease therapeutics by shifting away from inefficient resolution techniques toward a direct, catalytic asymmetric approach. By leveraging a specialized Iridium complex catalyst, the disclosed process facilitates an asymmetric reductive amination reaction that establishes the crucial chiral center with exceptional stereocontrol. This innovation is particularly vital for global supply chains seeking a reliable pharmaceutical intermediate supplier capable of delivering high-enantiomeric purity without the inherent waste of racemic splitting. The strategic implementation of this chemistry not only enhances the economic viability of production but also aligns with modern green chemistry principles by improving atom economy and reducing the reliance on stoichiometric chiral auxiliaries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer drugs like Rivastigmine relied heavily on the resolution of racemic mixtures, a process fundamentally flawed by its maximum theoretical yield ceiling of 50%. As illustrated in prior art such as WO2005061446, while resolution reagents might be inexpensive, the discarding of the unwanted enantiomer represents a massive inefficiency in raw material utilization and waste generation. Furthermore, alternative chemical synthesis routes reported in literature, such as those involving enzymatic kinetic resolution, often suffer from extended synthetic sequences that drastically increase processing time and operational costs. These legacy methods frequently necessitate the use of hazardous and corrosive reagents, including acetic anhydride and methylsulfonyl chloride, which pose significant safety risks to operators and complicate environmental compliance protocols. The cumulative effect of these drawbacks is a supply chain vulnerable to volatility, where cost reduction in API manufacturing is stifled by low overall yields and complex purification requirements.
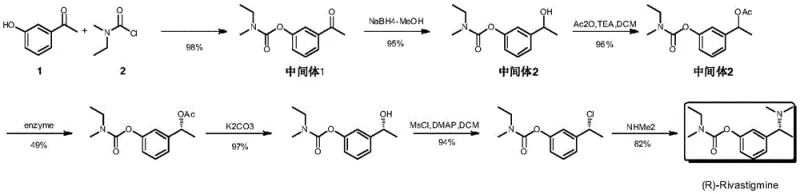
The Novel Approach
In stark contrast to these legacy limitations, the methodology outlined in CN110668976A introduces a streamlined four-step synthesis that bypasses the need for resolution entirely. The core of this innovation lies in the direct construction of the chiral amine scaffold through an Iridium-catalyzed asymmetric reductive amination of a ketone precursor. This approach allows for the conversion of readily available starting materials into the desired (R)-configuration intermediate with high efficiency and selectivity. By integrating a metal-catalyzed debenzylation and a subsequent Eschweiler-Clarke methylation, the process achieves a total yield approaching 80%, effectively doubling the material throughput compared to resolution strategies. This paradigm shift enables the commercial scale-up of complex pharmaceutical intermediates by simplifying the workflow and eliminating the bottleneck of separating enantiomers. The result is a robust manufacturing protocol that delivers high-purity chiral amine intermediates suitable for immediate downstream processing into the final active pharmaceutical ingredient.
Mechanistic Insights into Ir-Catalyzed Asymmetric Reductive Amination
The heart of this synthetic advancement is the sophisticated coordination chemistry employed during the asymmetric reductive amination step. The reaction utilizes a chiral Iridium complex, specifically designed with bulky binaphthyl-based ligands, to create a highly sterically constrained environment around the metal center. When the achiral ketone substrate interacts with the benzhydrylamine and hydrogen gas in the presence of this catalyst, the chiral pocket of the Iridium complex directs the hydride transfer to occur exclusively from one face of the imine intermediate. This precise spatial control ensures that the newly formed carbon-nitrogen bond possesses the desired (R)-stereochemistry with an enantiomeric excess (ee) value reaching up to 98%. The inclusion of additives such as titanium esters and organic acids further modulates the catalyst's activity, stabilizing the transition state and preventing non-selective background reactions that could erode optical purity.
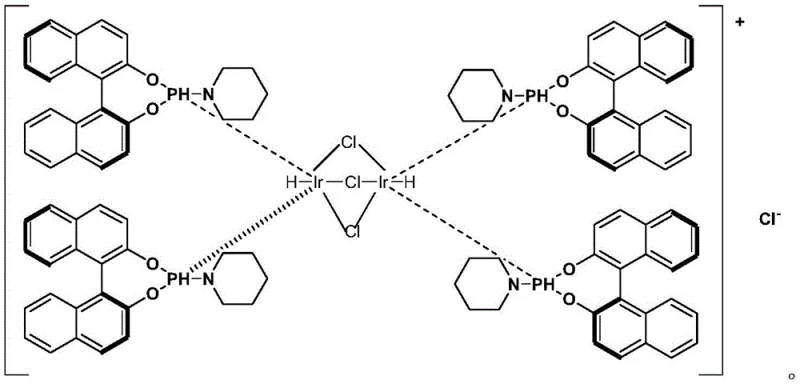
Beyond stereocontrol, the mechanistic design of this route inherently minimizes the formation of difficult-to-remove impurities. Traditional routes often generate regioisomers or over-alkylated byproducts that require extensive chromatographic purification, which is impractical on a multi-ton scale. In this catalytic cycle, the use of benzhydrylamine as a protecting group serves a dual purpose: it acts as the nitrogen source for the reductive amination and subsequently protects the amine during the reaction, only to be cleanly removed later via catalytic hydrogenolysis. This strategy prevents the formation of secondary amine impurities during the initial coupling and ensures that the final methylation step proceeds cleanly to the tertiary amine without quaternary salt formation. Such rigorous impurity control is essential for meeting the stringent regulatory standards required for central nervous system (CNS) drugs, where trace contaminants can have profound biological effects.
How to Synthesize (R)-Rivastigmine Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the critical asymmetric hydrogenation phase. The process begins with the preparation of the ketone carbamate precursor, followed by the key stereoselective step under controlled hydrogen pressure and temperature. The subsequent deprotection and methylation steps are designed to be operationally simple, utilizing standard heterogeneous catalysts and common reagents. For R&D teams looking to replicate or adapt this chemistry, the detailed reaction conditions regarding solvent selection, catalyst loading, and pressure ranges are critical for maximizing yield and ee. The following guide outlines the standardized operational framework derived from the patent examples to ensure reproducible results in a pilot or production setting.
- Condense 3-hydroxyacetophenone with N-ethyl-N-methylcarbamoyl chloride to form the acetophenone carbamate precursor.
- Perform asymmetric reductive amination using a chiral Iridium complex catalyst and benzhydrylamine under hydrogen pressure to establish chirality.
- Execute catalytic debenzylation followed by Eschweiler-Clarke methylation to finalize the (R)-Rivastigmine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates directly into enhanced operational resilience and financial efficiency. By eliminating the 50% yield loss inherent in resolution processes, manufacturers can effectively double their output from the same quantity of raw materials, leading to substantial cost savings in raw material procurement. Furthermore, the avoidance of corrosive and hazardous reagents simplifies waste treatment protocols and reduces the regulatory burden associated with handling dangerous chemicals. This streamlined approach not only lowers the cost of goods sold (COGS) but also mitigates supply risks by reducing dependency on specialized chiral resolving agents that may have limited availability. Consequently, this method supports reducing lead time for high-purity chiral amines by shortening the overall production cycle and minimizing purification bottlenecks.
- Cost Reduction in Manufacturing: The transition from resolution to asymmetric catalysis fundamentally alters the cost structure of production by maximizing atom economy. Since the theoretical yield is no longer capped at 50%, the effective cost per kilogram of the active intermediate is drastically reduced without requiring cheaper raw materials. Additionally, the high selectivity of the Iridium catalyst minimizes the formation of byproducts, which reduces the consumption of solvents and silica gel required for purification. This efficiency gain allows for a more competitive pricing strategy in the generic pharmaceutical market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on robust, commercially available catalysts and standard reagents ensures a stable supply chain that is less susceptible to disruptions. Unlike enzymatic processes that may require specific fermentation capabilities or sensitive biocatalysts, this chemical synthesis can be performed in standard stainless steel reactors equipped for hydrogenation. This versatility allows for flexible manufacturing across multiple sites, ensuring continuity of supply even if one facility faces operational challenges. The simplicity of the workflow also facilitates faster technology transfer between CDMO partners and internal manufacturing units.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily manageable in large-scale vessels. The elimination of toxic reagents like methylsulfonyl chloride significantly improves the environmental profile of the manufacturing process, aligning with increasingly strict global environmental regulations. This 'green' aspect not only reduces waste disposal costs but also enhances the corporate sustainability profile of the manufacturer, which is becoming a key criterion for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for stakeholders evaluating the potential for licensing or adopting this method for their own production pipelines.
Q: What is the primary advantage of this Ir-catalyzed route over traditional resolution methods?
A: Traditional resolution methods are limited to a maximum theoretical yield of 50%, whereas this asymmetric catalytic route achieves high yields exceeding 80% overall with >98% ee, significantly improving atom economy.
Q: Does this synthesis method avoid hazardous reagents found in previous literature?
A: Yes, unlike earlier enzymatic or chemical routes that utilized corrosive acetic anhydride and methylsulfonyl chloride, this method employs safer catalytic hydrogenation and methylation conditions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: The process utilizes robust catalytic systems and common solvents like dichloromethane and alcohols, making it highly adaptable for commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Rivastigmine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Iridium-catalyzed synthesis in meeting the growing global demand for high-quality Alzheimer's therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >98% ee. We are committed to delivering consistent quality and supply security for complex chiral intermediates, leveraging our deep technical expertise to optimize every step of the manufacturing process.
We invite pharmaceutical partners to collaborate with us to fully realize the economic and technical benefits of this advanced synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and accelerate your time to market for next-generation CNS medications.
