Optimizing Mosapride Intermediate Production via Novel Phthalimide Protection Strategy
Introduction to Advanced Mosapride Intermediate Synthesis
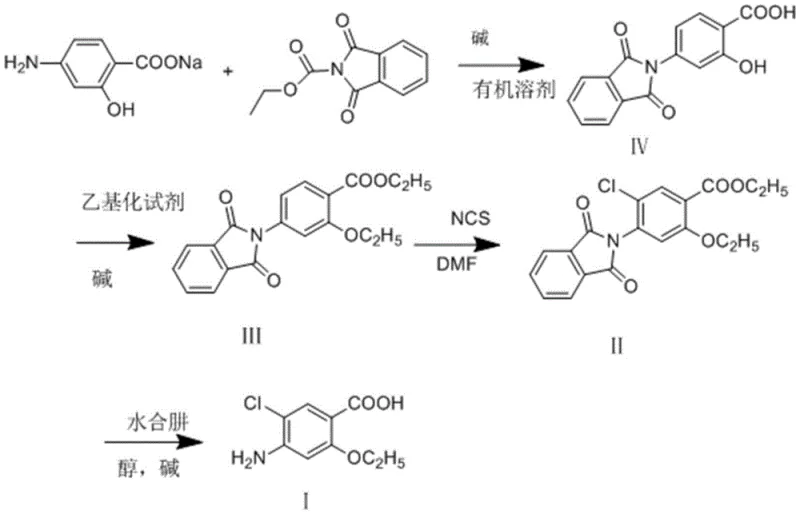
The pharmaceutical industry continuously seeks robust, cost-effective pathways for synthesizing critical active pharmaceutical ingredient (API) precursors, and the production of mosapride citrate intermediates is no exception. Patent CN110143892B introduces a groundbreaking preparation method for 2-ethoxy-4-amino-5-chlorobenzoic acid, a pivotal building block in the manufacture of mosapride, a potent gastrointestinal prokinetic agent. This innovative methodology departs from traditional, cumbersome synthetic routes by employing sodium p-aminosalicylate as a commercially abundant starting material. Through a streamlined four-step sequence involving N-ethoxycarbonylphthalimide acylation, strategic double ethylation, precise chlorination, and a final deprotection-hydrolysis cascade, the process achieves remarkable efficiency. For R&D directors and procurement specialists alike, this patent represents a significant leap forward, offering a pathway that not only enhances chemical purity but also fundamentally restructures the cost basis of production through the elimination of expensive reagents and the reduction of unit operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-ethoxy-4-amino-5-chlorobenzoic acid has been plagued by economic and operational inefficiencies that hinder large-scale adoption. Early literature, such as the method described in J. Med. Chem. (1991), relied on p-acetamido methyl salicylate as the starting scaffold. While chemically feasible, this approach suffers from the prohibitive cost and difficult availability of the initial raw materials. Furthermore, the reliance on iodoethane as the ethylating agent introduces a severe cost penalty, as iodine-based reagents are substantially more expensive than their bromine counterparts. Alternative methods, such as those disclosed in CN1526700A, attempted to mitigate raw material costs by using sodium p-amino-o-hydroxybenzoate but resulted in an elongated six-step synthesis. This increase in step count inevitably leads to cumulative yield losses, higher energy consumption due to repeated heating and cooling cycles, and increased waste generation, creating a substantial burden on both the supply chain and environmental compliance teams.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN110143892B leverages a sophisticated protecting group strategy to streamline the synthesis into just four high-yielding steps. By initiating the sequence with sodium p-aminosalicylate, a commodity chemical, the process immediately secures a stable and low-cost supply chain foundation. The introduction of the phthalimide moiety serves a dual purpose: it protects the amine functionality during subsequent harsh alkylation conditions and directs the regioselectivity of the chlorination step. This strategic design eliminates the need for complex purification between steps, as intermediates can often be isolated via simple crystallization. The replacement of iodoethane with bromoethane further underscores the economic foresight of this design, offering similar reactivity at a fraction of the cost. Consequently, this route delivers a total yield exceeding 85%, effectively doubling the efficiency of some prior art methods while drastically simplifying the operational workflow for manufacturing teams.
Mechanistic Insights into Phthalimide-Mediated Selective Functionalization
The core chemical innovation of this process lies in the precise control of reactivity afforded by the phthalimide protecting group. In the initial acylation step, sodium p-aminosalicylate reacts with N-ethoxycarbonylphthalimide under mild alkaline conditions (0-10°C). This reaction is highly exothermic and selective, forming the N-phthaloyl derivative (Compound IV) with exceptional purity (HPLC purity >99%). The phthalimide group is electron-withdrawing, which deactivates the aromatic ring towards electrophilic attack, thereby preventing unwanted poly-substitution during the subsequent chlorination phase. Moreover, the steric bulk of the phthalimide group helps to direct the incoming electrophiles to the desired positions on the benzene ring. This level of control is critical for maintaining the impurity profile within strict pharmaceutical limits, ensuring that downstream processing does not become bogged down by the removal of structural isomers or over-chlorinated byproducts.
Following the protection step, the mechanism proceeds through a double ethylation where both the phenolic hydroxyl group and the carboxylic acid are alkylated using bromoethane. This transformation converts the polar, hydrogen-bonding acid and phenol into lipophilic ether and ester functionalities, significantly altering the solubility profile of the intermediate (Compound III). This change is advantageous for purification, as the neutral organic intermediate can be easily separated from inorganic salts via aqueous workup. The final deprotection step utilizes hydrazine hydrate, which acts as a powerful nucleophile to cleave the phthalimide ring via the formation of phthalhydrazide, simultaneously releasing the free amine. Concurrently, the basic conditions facilitate the hydrolysis of the ethyl ester back to the carboxylic acid. This telescoped deprotection-hydrolysis maneuver is a masterstroke of process chemistry, combining two distinct chemical transformations into a single operational unit, thereby saving time, solvent, and energy.
How to Synthesize 2-Ethoxy-4-amino-5-chlorobenzoic Acid Efficiently
The synthesis of this high-value intermediate is predicated on a sequence of robust, scalable reactions that prioritize safety and yield. The process begins with the careful control of pH and temperature during the acylation to ensure complete conversion of the starting amine. Following isolation, the ethylation step requires a polar aprotic solvent like DMF to solubilize the ionic intermediates and facilitate the SN2 displacement of the bromide. The chlorination step is notably mild, utilizing N-chlorosuccinimide (NCS) rather than hazardous chlorine gas, which enhances plant safety. Finally, the deprotection is conducted in an alcoholic medium, allowing for the precipitation of the final product upon acidification. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized synthesis guide below.
- Perform phthalic acylation of sodium p-aminosalicylate with N-ethoxycarbonylphthalimide in alkaline aqueous solution.
- Execute double ethylation using bromoethane in a polar aprotic solvent to form the ethyl ester.
- Conduct regioselective chlorination using N-chlorosuccinimide (NCS) in DMF.
- Complete deprotection and hydrolysis simultaneously using hydrazine hydrate in ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative advantages that extend far beyond simple chemical yield. The primary driver of value is the radical reduction in raw material costs achieved by substituting scarce, high-priced reagents with commodity chemicals. By eliminating the dependency on iodoethane and p-acetamido methyl salicylate, manufacturers can insulate their production costs from the volatility of the iodine market and secure a more predictable budget for long-term contracts. Furthermore, the reduction in reaction steps from six to four directly correlates to a decrease in labor hours, utility consumption, and equipment occupancy time. This efficiency gain allows for a higher throughput per annum without the need for capital expenditure on new reactors, effectively increasing the asset utilization rate of existing manufacturing facilities.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the substitution of expensive reagents with cost-effective alternatives. The switch from iodoethane to bromoethane represents a significant line-item saving, as bromine derivatives are generally produced at a much larger industrial scale and carry a lower price tag. Additionally, the high yield of the first step (over 95%) ensures that the most expensive starting material is not wasted, maximizing the return on investment for every kilogram of input. The simplified workup procedures, which rely on crystallization rather than complex chromatography or distillation, further reduce the operational expenditure associated with solvent recovery and waste treatment, leading to substantial overall cost optimization.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of sodium p-aminosalicylate, a widely available feedstock with a mature global supply network. Unlike specialized intermediates that may have single-source suppliers, this starting material can be sourced from multiple vendors, mitigating the risk of supply disruptions. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without compromising quality, ensures consistent batch-to-batch reliability. This stability is crucial for maintaining uninterrupted production schedules for the final API, preventing costly delays in the pharmaceutical value chain and ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for green manufacturing initiatives. The avoidance of corrosive chlorine gas and the use of solid NCS reduces the hazard profile of the plant, lowering insurance and safety compliance costs. The process generates fewer byproducts and utilizes solvents that are easier to recover and recycle, aligning with increasingly stringent environmental regulations. The simplicity of the isolation steps, primarily involving filtration and washing, means that the process can be scaled from pilot kilograms to multi-ton commercial production with minimal engineering challenges, ensuring a smooth transition from R&D to full-scale manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines.
Q: Why is sodium p-aminosalicylate preferred over p-acetamido methyl salicylate?
A: Sodium p-aminosalicylate is significantly cheaper and more readily available than p-acetamido methyl salicylate. Furthermore, the new route avoids the use of expensive ethylating agents like iodoethane, replacing them with cost-effective bromoethane, thereby drastically reducing raw material costs.
Q: How does this process improve overall yield compared to prior art?
A: By utilizing a phthalimide protecting group strategy, the process achieves high regioselectivity and minimizes side reactions. The total yield reaches approximately 85.1%, with the initial acylation step alone achieving yields over 95%, which is superior to the multi-step, lower-yield processes disclosed in earlier patents.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the process is designed for scalability. It utilizes mild reaction temperatures (mostly below 100°C), common solvents like DMF and ethanol, and simple workup procedures involving crystallization and filtration, making it ideal for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethoxy-4-amino-5-chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-ethoxy-4-amino-5-chlorobenzoic acid meets the highest international standards required for API synthesis. We understand the critical nature of this intermediate in the mosapride supply chain and are dedicated to providing a seamless, reliable sourcing experience.
We invite you to collaborate with us to unlock the full potential of this advanced synthetic route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us partner with you to drive efficiency, reduce costs, and secure your supply chain for the future of gastrointestinal therapeutics.
