Advanced Synthesis of Lactone Ring Nematicide Intermediates for Commercial Scale Production
The development of effective nematicides is critical for modern agriculture, yet the synthesis of complex intermediates containing lactone rings has historically presented significant challenges regarding yield and cost efficiency. Patent CN108484538B introduces a breakthrough methodology for synthesizing [2-(2-methoxyphenyl)-5-oxotetrahydrofuran-3-yl]-carboxylic acid [4-(1,1,2-trifluoro-1-butene)yl] ester, a pivotal intermediate in the production of next-generation nematode control agents. This technical disclosure addresses the longstanding issue of raw material decomposition during esterification, specifically targeting the instability of 4-bromo-1,1,2-trifluoro-1-butene under traditional reaction conditions. By shifting from conventional weak base catalysis to a strategic salt-formation approach using organic strong bases, the patent demonstrates a pathway to achieve product yields consistently exceeding 85 percent, with optimized conditions reaching over 90 percent. For R&D directors and procurement specialists in the agrochemical sector, this represents a substantial opportunity to enhance the purity profile of final products while simultaneously driving down the cost of goods sold through improved material utilization. The following analysis details the mechanistic advantages and commercial implications of adopting this refined synthetic route for reliable agrochemical intermediate supply.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of this specific lactone ring-containing ester relied on conventional esterification protocols utilizing inorganic weak bases such as potassium carbonate or sodium carbonate in solvents like methanol or acetonitrile. These traditional approaches suffer from a fundamental chemical incompatibility where the alkaline conditions required for the reaction inadvertently trigger the decomposition of the sensitive 4-bromo-1,1,2-trifluoro-1-butene reagent. This decomposition not only consumes the expensive fluorinated starting material but also generates difficult-to-remove impurities that compromise the quality of the final active ingredient. Consequently, the calculated yield based on the fluorinated alkyl halide in these legacy processes is typically restricted to approximately 58.9 percent, creating a severe economic bottleneck for manufacturers. Furthermore, the incomplete conversion of the carboxylic acid necessitates complex downstream purification steps to isolate the target compound, further eroding profit margins and extending production cycles. The inability to control the decomposition of the halogenated hydrocarbon without stalling the reaction itself has been identified as the primary technical barrier limiting the widespread application of this nematicide.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally alters the reaction landscape by employing organic strong bases to pre-form a carboxylate salt before introducing the electrophilic halide. This two-stage process ensures that the nucleophilic carboxylate species is fully generated and stabilized prior to the substitution event, thereby minimizing the exposure of the sensitive 4-bromo-1,1,2-trifluoro-1-butene to harsh, uncontrolled alkaline environments. By utilizing bases such as DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) or tetramethylguanidine, the reaction proceeds with significantly higher efficiency, effectively solving the defect of incomplete reaction observed in prior art. The general chemical transformation is depicted below, illustrating the direct coupling of the lactone acid derivative with the fluorinated chain under these optimized conditions. 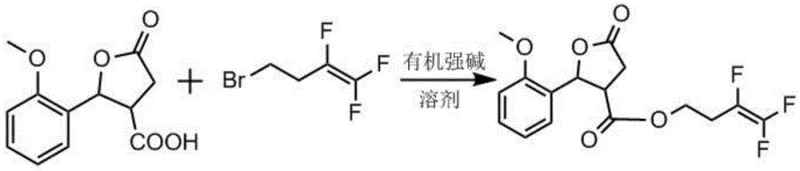
Mechanistic Insights into Organic Strong Base Catalyzed Esterification
The core of this technological advancement lies in the precise modulation of basicity to favor nucleophilic substitution over elimination or decomposition pathways. When an organic strong base like tetramethylguanidine is introduced to the 2-(2-methoxyphenyl)-5-oxotetrahydrofuran-3-carboxylic acid, it rapidly deprotonates the carboxylic acid group to form a highly reactive carboxylate anion paired with a bulky, non-nucleophilic cation. This salt formation step is crucial because it activates the acid without generating the free hydroxide ions that typically degrade fluorinated alkenes in aqueous or protic media. The subsequent addition of the bromo-butene allows for a clean SN2-type substitution where the carboxylate attacks the carbon bearing the bromine atom, displacing the halide leaving group. The reaction mechanism involving tetramethylguanidine is detailed in the figure below, highlighting the interaction between the base, the acid, and the subsequent alkylation step. 
From an impurity control perspective, this mechanism drastically reduces the formation of side products associated with the hydrolysis or polymerization of the trifluoro-butene moiety. In conventional weak base systems, the equilibrium often favors the presence of unreacted acid alongside decomposed halide fragments, leading to a messy crude profile that requires extensive chromatography or recrystallization. In contrast, the strong base method drives the reaction to near completion, as evidenced by yields reaching up to 96.8 percent in specific examples using chloroform as the solvent. The choice of solvent also plays a synergistic role; non-polar or moderately polar aprotic solvents like chloroform, toluene, or dichloromethane provide an ideal medium that solubilizes the organic salts while maintaining the stability of the fluorinated intermediate. This mechanistic clarity provides R&D teams with a robust framework for scaling the process, ensuring that the critical quality attributes of the intermediate remain consistent from kilogram to ton-scale batches.
How to Synthesize Lactone Ring Nematicide Efficiently
The operational protocol derived from this patent offers a streamlined workflow that balances reaction kinetics with thermal management to maximize output. The process begins with the dissolution of the lactone carboxylic acid in a selected organic solvent, followed by the controlled addition of the organic strong base at temperatures ranging from 0 to 80 degrees Celsius to ensure complete salt formation. Once the carboxylate is established, the fluorinated bromide is introduced, and the mixture is heated to between 40 and 110 degrees Celsius, typically under reflux, for a duration of 1 to 72 hours depending on the specific base and solvent combination employed. The workup procedure is notably simple, involving a standard acid wash to neutralize excess base, followed by an alkali wash to remove acidic impurities, and finally, vacuum distillation to isolate the pure ester product. For a comprehensive breakdown of the specific molar ratios, temperature profiles, and processing times validated in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve 2-(2-methoxyphenyl)-5-oxotetrahydrofuran-3-carboxylic acid in a solvent such as chloroform or toluene and add an organic strong base like tetramethylguanidine to form a salt.
- Add 4-bromo-1,1,2-trifluoro-1-butene to the reaction mixture and perform a substitution reaction under reflux conditions for 48 to 60 hours.
- Perform acid washing with hydrochloric acid followed by alkali washing with sodium carbonate solution, separate the layers, and remove the solvent via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis method translates directly into enhanced operational resilience and significant cost structure improvements. The primary economic driver is the drastic reduction in raw material waste, particularly regarding the expensive 4-bromo-1,1,2-trifluoro-1-butene, which is no longer lost to decomposition side reactions. By pushing yields from the historical average of roughly 59 percent to over 90 percent, manufacturers can effectively nearly double the output from the same quantity of input materials, resulting in substantial cost savings in nematicide manufacturing without requiring capital investment in new equipment. Furthermore, the simplified post-treatment process, which eliminates the need for complex purification steps to remove decomposition byproducts, reduces solvent consumption and energy usage during the isolation phase. This efficiency gain shortens the overall batch cycle time, allowing facilities to increase throughput and respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The shift to high-yield chemistry fundamentally alters the cost basis of the intermediate by maximizing the utility of the most expensive reagent. Eliminating the loss of fluorinated raw materials means that the cost per kilogram of the final active ingredient is significantly lowered, providing a competitive pricing advantage in the global agrochemical market. Additionally, the use of recoverable solvents like chloroform and toluene, combined with a straightforward aqueous workup, minimizes waste disposal costs and reduces the burden on environmental compliance teams. The qualitative improvement in process efficiency ensures that production budgets are utilized more effectively, freeing up capital for other strategic initiatives within the organization.
- Enhanced Supply Chain Reliability: A more robust chemical process inherently leads to a more reliable supply chain, as the risk of batch failure due to incomplete reaction or impurity spikes is markedly reduced. The ability to consistently achieve high purity specifications means that downstream formulation partners receive material that meets rigorous quality standards without delay. This reliability is critical for maintaining continuous production schedules for the final nematicide product, preventing stockouts that could impact agricultural seasons. Moreover, the flexibility to use various organic strong bases and solvents provides procurement teams with options to source materials based on availability and price, mitigating the risk of supply disruptions for any single reagent.
- Scalability and Environmental Compliance: The method is explicitly designed for large-scale industrial production, utilizing unit operations that are standard in fine chemical manufacturing plants. The absence of exotic catalysts or extreme pressure conditions simplifies the engineering requirements for scale-up, allowing for a smooth transition from pilot plant to commercial manufacturing. From an environmental standpoint, the reduction in byproduct formation aligns with green chemistry principles by lowering the E-factor of the process. This makes the technology attractive for companies aiming to reduce their carbon footprint and meet increasingly stringent regulatory requirements regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the organic strong base method over traditional weak base protocols, focusing on yield optimization and impurity control.
Q: Why is the yield of conventional synthesis methods for this nematicide so low?
A: Conventional methods using weak bases like potassium carbonate often result in incomplete reactions and significant decomposition of the expensive 4-bromo-1,1,2-trifluoro-1-butene原料,leading to yields as low as 58.9%.
Q: What represents the key innovation in patent CN108484538B?
A: The key innovation is the use of organic strong bases such as DBU or tetramethylguanidine to form a carboxylate salt prior to substitution, which stabilizes the reaction environment and prevents the decomposition of the fluorinated alkyl halide.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes common solvents like chloroform and toluene and achieves yields exceeding 90%, making it highly scalable and cost-effective for commercial manufacturing compared to previous techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lactone Ring Nematicide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN108484538B for the agrochemical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield route are fully realized in a practical manufacturing setting. Our facility is equipped with rigorous QC labs and stringent purity specifications protocols that guarantee every batch of lactone ring intermediate meets the exacting standards required for modern pesticide formulations. We are committed to leveraging this advanced chemistry to deliver high-purity agrochemical intermediates that empower our clients to bring more effective crop protection solutions to market faster.
We invite global agrochemical enterprises to collaborate with us to explore the full commercial potential of this technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about optimizing your nematicide supply chain with a partner dedicated to innovation and quality.
