Advanced Synthesis of 3-Amino-2-methyltrifluorotoluene for Scalable Pharmaceutical Production
Advanced Synthesis of 3-Amino-2-methyltrifluorotoluene for Scalable Pharmaceutical Production
The pharmaceutical industry constantly seeks robust, safe, and economically viable pathways for producing critical intermediates, particularly those serving as building blocks for potent anti-inflammatory agents. Patent CN1030073A introduces a transformative methodology for the synthesis of 3-amino-2-methyltrifluorotoluene, a pivotal compound in the manufacture of therapeutic drugs such as anilinacin and N-(2-methyl-3-trifluoromethylphenyl)anthranilic acid. This technical insight report analyzes the proprietary three-step sequence detailed in the patent, which replaces hazardous high-pressure fluorination with a streamlined nitration-methylation-reduction protocol. For R&D directors and procurement specialists, understanding this shift is crucial, as it directly impacts the purity profile of the final API and the overall cost structure of the supply chain. By leveraging inexpensive starting materials like benzotrifluoride and avoiding exotic reagents, this process establishes a new benchmark for reliability in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 3-amino-2-methyltrifluorotoluene relied heavily on methodologies described in earlier patents, such as U.S. Patent No. 3,390,170, which utilized sulfur tetrafluoride (SF4) to convert 2-methyl-3-nitrobenzoic acid into the desired trifluoromethyl derivative. This conventional approach presents severe operational hazards, requiring stainless steel high-pressure gas storage tanks and reaction temperatures exceeding 100°C for extended periods, often up to 15 hours. The inherent instability of SF4 under these conditions poses a significant safety risk, as reactor failure could lead to the catastrophic release of corrosive reaction mixtures. Furthermore, the requirement for specialized high-pressure equipment drastically increases capital expenditure and maintenance costs, creating a bottleneck for manufacturers aiming to scale production efficiently. The complexity of handling toxic fluorinating agents also necessitates rigorous safety protocols and waste treatment systems, further inflating the operational overhead associated with legacy synthesis routes.
The Novel Approach
In stark contrast, the novel approach disclosed in CN1030073A circumvents these dangers by employing a completely different retrosynthetic logic that begins with the readily available benzotrifluoride. Instead of introducing the trifluoromethyl group late in the synthesis under extreme conditions, this method utilizes the trifluoromethyl group as a pre-existing structural feature that directs subsequent functionalization. The process involves a mild nitration step followed by a sophisticated methylation using trimethylsulfoxonium halides, and concludes with a standard catalytic reduction. This strategic inversion of the synthetic sequence allows the reaction to proceed under substantially non-hazardous conditions, primarily at atmospheric pressure and moderate temperatures ranging from 0°C to 50°C. By eliminating the need for high-pressure fluorination, the novel approach not only enhances operator safety but also simplifies the engineering requirements for commercial reactors, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into the Three-Step Synthetic Sequence
The success of this synthesis relies on precise control over regioselectivity and chemoselectivity at each stage. The initial nitration of benzotrifluoride exploits the strong electron-withdrawing nature of the trifluoromethyl group, which acts as a meta-director during electrophilic aromatic substitution. By maintaining the reaction temperature between 20°C and 30°C using concentrated nitric-sulfuric acid, the process maximizes the formation of 3-nitrobenzotrifluoride while suppressing the formation of ortho or para isomers. Following isolation, the critical methylation step employs a sulfonium ylide generated in situ from trimethylsulfoxonium halide and a strong base like sodium hydride in an aprotic solvent such as DMSO. This ylide acts as a nucleophile, attacking the aromatic ring adjacent to the nitro group, effectively installing the methyl group at the 2-position through a mechanism that preserves the integrity of the sensitive trifluoromethyl moiety. Finally, the nitro group is selectively reduced to an amine using palladium on carbon (Pd/C) under hydrogen atmosphere, a transformation that is highly efficient and generates minimal byproduct waste compared to chemical reduction methods involving iron or tin.
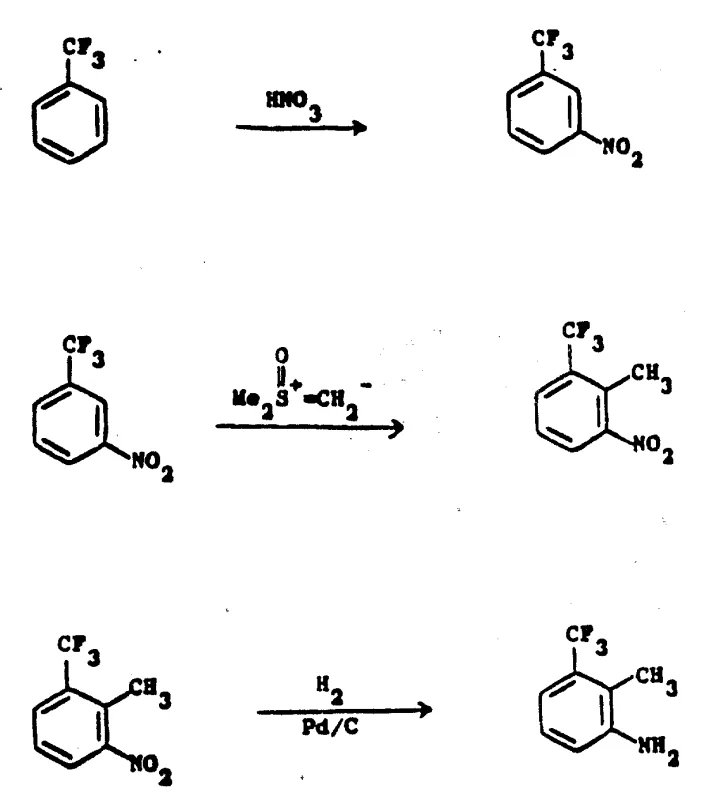
Understanding the impurity profile is vital for R&D teams aiming for high-purity pharmaceutical intermediates. The use of catalytic hydrogenation in the final step is particularly advantageous because it avoids the introduction of heavy metal salts that are difficult to remove and often require complex purification workflows. The mild conditions of the methylation step, conducted below 30°C under an inert nitrogen atmosphere, prevent thermal degradation of the nitro intermediate, ensuring a cleaner crude product before distillation. This attention to reaction parameters ensures that the final 3-amino-2-methyltrifluorotoluene meets stringent purity specifications required for downstream coupling reactions in drug synthesis. The mechanistic elegance of using a sulfonium ylide for aromatic methylation provides a unique solution to the steric and electronic challenges posed by the trifluoromethyl substituent, offering a reproducible pathway that minimizes batch-to-batch variability.
How to Synthesize 3-Amino-2-methyltrifluorotoluene Efficiently
Implementing this synthesis requires careful adherence to the specific reaction conditions outlined in the patent to ensure optimal yield and safety. The process is divided into three distinct operational units: nitration, methylation, and reduction, each requiring specific solvent systems and temperature controls. Operators must ensure that the nitration mixture is quenched properly into ice water to precipitate the nitro intermediate, and that the methylation step is strictly moisture-free to maintain the activity of the sulfonium ylide. The following guide summarizes the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Nitrate benzotrifluoride using concentrated nitric-sulfuric acid at 20-30°C to obtain 3-nitrobenzotrifluoride.
- React 3-nitrobenzotrifluoride with trimethylsulfoxonium halide in DMSO under inert atmosphere to form 3-nitro-2-methylbenzotrifluoride.
- Reduce the nitro group of the intermediate to an amino group using catalytic hydrogenation with Pd/C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; benzotrifluoride is a commodity chemical available in bulk quantities globally, whereas the precursors for the old SF4 method were specialized and hazardous. This shift significantly reduces the risk of supply disruption and allows for more flexible sourcing strategies, ensuring continuous production even during market fluctuations. Furthermore, the elimination of high-pressure reactors lowers the barrier to entry for contract manufacturing organizations, increasing the number of qualified suppliers capable of producing this intermediate at scale.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and dangerous reagents like sulfur tetrafluoride, which require specialized containment and disposal protocols. By replacing high-energy, high-pressure steps with ambient pressure reactions, the energy consumption per kilogram of product is significantly lowered, leading to direct utility cost savings. Additionally, the use of catalytic hydrogenation instead of stoichiometric metal reductions reduces the volume of solid waste generated, thereby lowering waste treatment costs and environmental compliance burdens. These factors combine to create a leaner cost structure that allows for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The safety profile of this method directly correlates with supply chain resilience. Facilities operating under atmospheric pressure and moderate temperatures face lower risks of unplanned shutdowns due to safety incidents or regulatory inspections related to high-pressure vessels. The stability of the intermediates, such as 3-nitro-2-methyltrifluorotoluene, allows for safer storage and transportation between different processing sites if a multi-vendor strategy is employed. This robustness ensures that lead times for high-purity pharmaceutical intermediates remain consistent, supporting the just-in-time manufacturing models adopted by many modern drug developers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward because it utilizes unit operations common in the fine chemical industry, such as standard stirred-tank reactors and filtration units. The absence of corrosive fluorinating gases simplifies the material selection for equipment, reducing capital investment in Hastelloy or specialized alloys. From an environmental perspective, the process generates fewer hazardous byproducts, aligning with green chemistry principles and facilitating easier permitting for new production lines. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet growing market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN1030073A, providing clarity on the practical aspects of adoption. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does this synthesis method improve safety compared to traditional SF4 processes?
A: Unlike traditional methods utilizing sulfur tetrafluoride under high pressure and temperatures exceeding 100°C, this novel route operates at atmospheric pressure and moderate temperatures, eliminating the risk of vessel rupture and corrosive release.
Q: What represents the key regioselectivity challenge in the nitration step?
A: The trifluoromethyl group is a strong meta-director. The process leverages this electronic effect to ensure high selectivity for the 3-nitro isomer, minimizing ortho/para impurities that are difficult to separate downstream.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of common reagents like concentrated sulfuric acid and standard catalytic hydrogenation equipment makes this route highly scalable, avoiding the specialized high-pressure reactors required by older methodologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-2-methyltrifluorotoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that 3-amino-2-methyltrifluorotoluene plays in the development of next-generation anti-inflammatory therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your overall production budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of this essential building block.
