Revolutionizing Aryl Phosphorus Compound Production with Base-Promoted C-H Activation Technology
Revolutionizing Aryl Phosphorus Compound Production with Base-Promoted C-H Activation Technology
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing processes. Patent CN115716849A introduces a groundbreaking synthetic methodology that challenges the decades-old reliance on transition metal catalysis for constructing aryl phosphorus bonds. This innovation utilizes a base-promoted activation strategy involving N,N,N-trimethyl-N-arylmethylammonium iodide and phosphine oxyhydrogen reagents to achieve efficient phosphorylation. For R&D directors and procurement specialists in the fine chemical sector, this represents a paradigm shift away from expensive palladium systems toward a more accessible, metal-free protocol. The technology enables the direct functionalization of aromatic C-H sites under remarkably mild conditions, offering a robust pathway for producing high-value intermediates used in pharmaceuticals, agrochemicals, and material science without the burden of heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl phosphorus compounds has been dominated by methods requiring toxic phosphorus halides or transition metal-catalyzed cross-coupling reactions, such as the Hirao coupling. These traditional approaches often necessitate the use of unstable organometallic reagents like organolithium or Grignard reagents, which pose significant safety hazards and environmental risks due to their sensitivity to moisture and air. Furthermore, modern palladium or nickel-catalyzed protocols typically demand high reaction temperatures, sometimes exceeding 160°C, to activate relatively inert aryl C-X bonds. This high energy consumption not only increases operational costs but also places severe stress on reaction equipment and safety protocols. Additionally, the inevitable presence of transition metal catalysts introduces a critical purification challenge, requiring extensive downstream processing to remove trace heavy metals to meet stringent regulatory standards for pharmaceutical and electronic grade materials.
The Novel Approach
In stark contrast, the methodology disclosed in CN115716849A leverages a unique base-promoted mechanism that activates the benzylic position of quaternary ammonium salts to facilitate aromatic C-H phosphorylation. This approach completely bypasses the need for pre-functionalized aryl halides or expensive transition metal catalysts. By utilizing cheap and readily available inorganic bases such as cesium carbonate or potassium tert-butoxide, the reaction proceeds efficiently at mild temperatures ranging from 40°C to 60°C. This drastic reduction in thermal energy requirements translates directly into lower utility costs and enhanced process safety. The simplicity of the operation, combined with the use of stable ammonium salt precursors, makes this route highly attractive for large-scale manufacturing where reproducibility and safety are paramount concerns for supply chain managers.
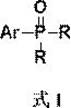
Mechanistic Insights into Base-Promoted C-H Phosphorylation
The core innovation of this technology lies in its ability to activate the aromatic ring through a benzylic activation pathway rather than direct oxidative addition into a carbon-halogen bond. The reaction initiates with the base-mediated deprotonation or activation of the N,N,N-trimethyl-N-arylmethylammonium iodide at the benzylic position. This generates a reactive intermediate that subsequently facilitates the activation of the adjacent aromatic C-H site, allowing for the nucleophilic attack by the phosphine oxyhydrogen reagent. This mechanism avoids the high energy barriers associated with breaking strong C-X bonds found in traditional cross-coupling. For process chemists, understanding this mechanistic nuance is crucial as it dictates the selection of solvents and bases. The patent specifies solvents like dimethyl sulfoxide, anisole, or acetonitrile, which stabilize the ionic intermediates formed during the base promotion, ensuring high conversion rates without the formation of complex metal-ligand byproducts that often plague transition metal catalysis.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity profile of the final product. In conventional palladium-catalyzed reactions, side reactions such as homocoupling of the aryl halide or beta-hydride elimination can lead to difficult-to-remove impurities. Moreover, residual palladium levels must be reduced to parts-per-million (ppm) levels, often requiring specialized scavengers or recrystallization steps that reduce overall yield. The base-promoted method described here eliminates the source of metal contamination entirely. The primary byproducts are typically inorganic salts and trimethylamine derivatives, which are significantly easier to separate via aqueous workup or standard column chromatography. This results in a cleaner crude product and simplifies the purification workflow, thereby enhancing the overall process mass intensity (PMI) and making the synthesis more environmentally benign and economically viable for commercial production.

How to Synthesize Aryl Phosphorus Compounds Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of execution in standard chemical manufacturing facilities. The process involves mixing the quaternary ammonium salt precursor with the phosphine oxide reagent and a stoichiometric amount of base in a polar aprotic solvent. The reaction is conducted under an inert atmosphere to prevent oxidation of the phosphorus species, although the mild conditions reduce the sensitivity compared to organometallic routes. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and workup techniques are critical for maximizing yield and purity. The following section outlines the generalized workflow derived from the patent examples, serving as a foundational guide for process development teams looking to adapt this technology for specific target molecules.
- Mix N,N,N-trimethyl-N-arylmethylammonium iodide, phosphine oxyhydrogen compound, and a cheap inorganic base in an organic solvent.
- Stir the reaction mixture under an inert gas environment at a mild temperature range of 40°C to 60°C for approximately 3 hours.
- Remove the solvent via reduced pressure distillation and purify the crude product using column chromatography to obtain the target aryl phosphorus compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-promoted synthesis technology offers substantial strategic advantages beyond mere technical feasibility. The most immediate impact is seen in the raw material cost structure. By replacing expensive palladium catalysts and specialized ligands with commodity inorganic bases like potassium methoxide or cesium carbonate, the direct material cost of the reaction is significantly reduced. Furthermore, the elimination of heavy metals removes the need for costly metal scavenging resins and extensive analytical testing for residual metals, streamlining the quality control budget. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and lower utility bills, which aligns with modern sustainability goals and corporate social responsibility mandates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of high-value transition metal catalysts with low-cost inorganic bases. Traditional methods often require catalyst loadings that, while low in percentage, represent a significant portion of the bill of materials due to the high price of precious metals. By removing this cost center entirely, manufacturers can achieve a leaner cost structure. Additionally, the simplified purification process reduces solvent consumption and waste disposal costs associated with metal-contaminated waste streams. The overall effect is a more competitive pricing model for the final aryl phosphorus intermediates, allowing downstream customers to optimize their own production costs without compromising on quality or performance specifications.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of widely available and stable starting materials. Quaternary ammonium salts and phosphine oxides are generally more stable and easier to source than sensitive organometallic reagents or specialized aryl halides that may have limited suppliers. The robustness of the reaction conditions, operating at near-ambient temperatures, reduces the risk of batch failures due to thermal runaway or equipment malfunction. This reliability ensures consistent delivery schedules and reduces the need for excessive safety stock. For global supply chains, the ability to manufacture these intermediates in diverse geographic locations without requiring specialized high-pressure or high-temperature reactors enhances logistical flexibility and mitigates regional supply disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, particularly regarding heat transfer and safety. The mild exothermic nature and low temperature range of this base-promoted reaction make it inherently safer and easier to scale from kilogram to multi-ton production. The absence of toxic phosphorus halides and heavy metals simplifies environmental compliance and waste treatment. Facilities can operate with standard ventilation and waste management systems, avoiding the need for specialized containment required for highly toxic or pyrophoric reagents. This ease of scale-up accelerates the time-to-market for new products relying on these aryl phosphorus motifs, enabling faster response to market demands in the pharmaceutical and agrochemical sectors while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-promoted synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on substrate scope, reaction conditions, and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing manufacturing pipelines or for procurement professionals assessing the long-term viability of suppliers utilizing this advanced chemistry.
Q: What are the primary advantages of this base-promoted method over traditional palladium-catalyzed routes?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts like palladium or nickel. This method operates at significantly lower temperatures (40-60°C vs 160°C) and uses cheap inorganic bases, drastically reducing raw material costs and eliminating the need for complex heavy metal removal steps.
Q: What types of substrates are compatible with this synthesis protocol?
A: The protocol demonstrates high compatibility with a wide range of aromatic systems, including substituted naphthyl groups, benzothiophenes, benzofurans, and various phenyl derivatives with electron-donating or withdrawing groups such as methoxy, chloro, and trifluoromethyl substituents.
Q: How does this method impact the purity profile of the final organophosphorus product?
A: By avoiding transition metal catalysts, the risk of heavy metal contamination in the final product is effectively removed. This results in a cleaner impurity profile, which is critical for pharmaceutical and electronic applications where trace metal limits are strictly regulated.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Phosphorus Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free C-H activation technologies in the production of high-value fine chemical intermediates. Our technical team has extensively analyzed the capabilities of this base-promoted pathway and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of aryl phosphorus compound meets the exacting standards required for pharmaceutical and electronic applications. We are committed to delivering consistent quality and supply continuity for our global partners.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project requirements. Whether you need custom synthesis of complex naphthyl-phosphine oxides or large-scale production of benzothiophene derivatives, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations. Let us help you optimize your supply chain and reduce manufacturing costs with our advanced, sustainable chemical solutions.
