Advanced Synthetic Route for High-Purity 2,3,6,7-Anthracene Tetracarboxylic Dianhydride Monomers
Advanced Synthetic Route for High-Purity 2,3,6,7-Anthracene Tetracarboxylic Dianhydride Monomers
The rapid evolution of the organic electronics sector demands monomers that combine exceptional thermal stability with superior luminescent properties. Patent CN114149444B introduces a groundbreaking methodology for synthesizing 2,3,6,7-anthracene tetracarboxylic dianhydride (ANTDA), a critical building block for high-performance polyimides used in blue light-emitting applications. This novel approach addresses longstanding challenges in the field by utilizing a Diels-Alder cycloaddition strategy that significantly enhances the solubility and processability of key intermediates. By shifting away from traditional N-phenylmaleimide precursors, this technology offers a pathway to materials with wider energy gaps and higher luminous efficiency, directly supporting the development of next-generation display devices and photoresists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of anthracene-based dianhydrides has been plagued by inefficient reaction pathways and severe environmental drawbacks. Prior art, such as the method disclosed in Japanese patent document JP2008297354A, relies on the reaction of 1,2,4,5-tetramethylbenzene derivatives with N-phenylmaleimide. This conventional route suffers from critically low yields, often stagnating around 35% for the diimide intermediate, which drastically inflates raw material costs. Furthermore, the resulting N,N'-diphenyl-2,3,6,7-anthracene diimide exhibits extremely poor solubility, making central control, separation, and purification exceptionally difficult during manufacturing. Perhaps most concerning for modern supply chains is the hydrolysis step, which requires harsh conditions of 150°C for 25 hours and generates aniline as a primary wastewater component, posing significant toxicity risks and regulatory compliance burdens.
The Novel Approach
In stark contrast, the methodology outlined in CN114149444B employs 1,2,4,5-tetra(dibromomethyl)benzene and butynedicarboxylate as starting materials to forge the anthracene core. This strategic shift results in a tetraester intermediate with moderate solubility and polarity, facilitating much smoother reaction control and purification processes suitable for industrial large-scale production. The reaction yield for this key intermediate is notably improved, reaching approximately 50%, providing a more robust foundation for downstream processing. Moreover, the subsequent hydrolysis can be conducted under mild conditions, even at room temperature, which not only shortens reaction times but also ensures that the waste stream consists primarily of alcohols rather than toxic aniline, aligning perfectly with green chemistry principles and reducing environmental liability.
Mechanistic Insights into Diels-Alder Cycloaddition and Hydrolysis
The core of this innovative synthesis lies in the initial Diels-Alder reaction, where 1,2,4,5-tetra(dibromomethyl)benzene acts as the diene precursor and reacts with butynedicarboxylate as the dienophile. This cycloaddition is promoted by sodium iodide in polar aprotic solvents such as DMF or DMAc at temperatures ranging from 60°C to 120°C. The presence of sodium iodide is crucial, likely facilitating the elimination of bromide ions to generate the reactive diene species in situ, which then undergoes [4+2] cycloaddition to form the anthracene skeleton. This mechanism avoids the formation of the recalcitrant phenyl-imide bonds found in older methods, thereby preventing the solubility issues that hamper purification. The resulting tetraester maintains a structural integrity that allows for efficient isolation via recrystallization, ensuring high purity before the final conversion steps.
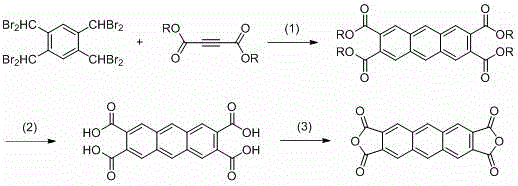
Following the cyclization, the transformation of the tetraester into the final dianhydride involves a two-stage sequence of hydrolysis and dehydration that is remarkably gentle compared to legacy protocols. The hydrolysis step can be performed using either alkaline bases like sodium hydroxide or mineral acids at temperatures as low as room temperature to 55°C. This mildness is a direct consequence of the ester linkage stability, which is far more labile than the imide bonds of the prior art, allowing for rapid cleavage without degrading the sensitive anthracene core. The final dehydration using acetic anhydride at 120°C proceeds with high efficiency, yielding the target dianhydride with purity levels exceeding 99.5%. This mechanistic elegance ensures that impurity profiles are tightly controlled, a critical factor for R&D directors focusing on the performance consistency of electronic materials.
How to Synthesize 2,3,6,7-Anthracene Tetracarboxylic Dianhydride Efficiently
The synthesis protocol described in the patent provides a clear, scalable roadmap for producing this high-value monomer. The process begins with the precise stoichiometric mixing of the dibromomethyl benzene derivative and the butynedicarboxylate ester in the presence of a sodium iodide catalyst. Following the cycloaddition, the intermediate is isolated and subjected to hydrolysis, where pH control is vital to precipitate the tetracarboxylic acid cleanly. Finally, the acid is dehydrated to close the anhydride rings. For detailed operational parameters, including specific solvent volumes, stirring rates, and crystallization techniques required to replicate these results in a pilot plant setting, please refer to the standardized synthesis guide below.
- Perform a Diels-Alder reaction between 1,2,4,5-tetra(dibromomethyl)benzene and butynedicarboxylate using sodium iodide in DMF or DMAc at 60-120°C.
- Hydrolyze the resulting tetraester intermediate under mild alkaline or acidic conditions at room temperature to obtain the tetracarboxylic acid.
- Dehydrate the tetracarboxylic acid using acetic anhydride at elevated temperatures to finalize the dianhydride structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the dramatic simplification of the purification workflow; because the tetraester intermediate possesses moderate solubility, it does not require the aggressive solvents or extreme conditions needed to process the insoluble diimides of the past. This ease of handling reduces the complexity of equipment requirements and minimizes the potential for batch-to-batch variability, ensuring a more reliable supply of high-purity raw materials for downstream polymerization. Furthermore, the shift to a cleaner waste profile fundamentally alters the cost structure of waste management and regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of harsh hydrolysis conditions (dropping from 150°C to room temperature) results in substantial energy savings and reduces the wear and tear on reactor vessels. Additionally, the removal of aniline from the waste stream eliminates the need for specialized and costly hazardous waste treatment protocols associated with toxic aromatic amines. The higher yield of the initial cyclization step also means less raw material is wasted per kilogram of final product, directly lowering the cost of goods sold (COGS) for this electronic chemical.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials like butynedicarboxylate esters ensures a robust supply base that is less susceptible to the bottlenecks often seen with specialized maleimide derivatives. The mild reaction conditions also imply that the process can be run in a wider variety of standard chemical manufacturing facilities without requiring exotic high-pressure or high-temperature equipment, thereby increasing the number of qualified suppliers capable of producing this material and reducing lead time for high-purity anthracene derivatives.
- Scalability and Environmental Compliance: The generation of alcohol-based wastewater instead of aniline-laden effluent significantly eases the burden on environmental health and safety (EHS) departments. This makes the commercial scale-up of complex polyimide monomers much more feasible in regions with strict environmental regulations. The process inherently supports sustainable manufacturing goals, reducing the carbon footprint associated with heating and waste neutralization, which is increasingly a prerequisite for qualifying as a vendor to major multinational electronics corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this monomer into your existing polyimide production lines.
Q: What are the key advantages of this new synthesis method over prior art?
A: The new method utilizes butynedicarboxylate instead of N-phenylmaleimide, resulting in intermediates with moderate solubility that are easier to purify. Additionally, the hydrolysis step occurs at room temperature rather than 150°C, significantly reducing energy consumption and eliminating toxic aniline waste.
Q: What is the expected yield for the key Diels-Alder step?
A: According to the patent data, the reaction yield for the formation of the 2,3,6,7-anthracene tetracarboxylic acid tetraester intermediate is relatively high, reaching approximately 50%, which is a substantial improvement over the 35% yield observed in conventional N-phenylmaleimide routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. The use of common solvents like DMF and DMAc, combined with mild hydrolysis conditions and the generation of less toxic alcohol-based waste streams, makes it highly suitable for commercial scale-up of complex polyimide monomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,6,7-Anthracene Tetracarboxylic Dianhydride Supplier
As the demand for advanced display materials continues to surge, securing a stable source of high-performance monomers like ANTDА is critical for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your R&D and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of electronic chemical we deliver meets the exacting standards required for OLED and semiconductor applications.
We invite you to engage with our technical team to explore how this optimized synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in energy and waste treatment costs for your operations. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume requirements.
