Optimizing Biotin Intermediate Production for Commercial Scale and Cost Efficiency
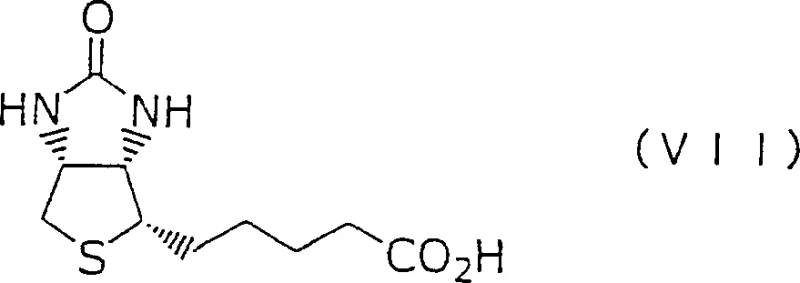
The global demand for high-purity vitamins and nutritional additives continues to drive innovation in synthetic chemistry, particularly for essential compounds like Biotin, also known as Vitamin H. Patent CN1273453C introduces a groundbreaking method for preparing biotin synthesis intermediates that offers significant advantages for industrial application. This technology discloses a general formula for novel thienoimidazole compounds, characterized by subjecting specific precursors to a ring conversion process. By leveraging this advanced synthetic route, manufacturers can overcome traditional bottlenecks associated with lengthy production cycles and complex purification requirements. The patent details a robust framework for generating key intermediates, specifically compounds of Formula (I) and Formula (III), which serve as critical building blocks in the value chain. For R&D directors and procurement specialists, understanding the nuances of this intellectual property is vital for securing a competitive edge in the fine chemical market. The method not only promises enhanced yield profiles but also aligns with modern sustainability goals by reducing the overall step count. As we delve into the technical specifics, it becomes clear that this innovation represents a pivotal shift towards more efficient vitamin manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biotin and its precursors has been plagued by inefficient methodologies that hinder large-scale commercial viability. Conventional routes often rely on thienoimidazole compounds that require extensive processing, including multiple protection and deprotection steps that add unnecessary complexity to the workflow. A major drawback identified in prior art is the necessity for complicated optical resolution during the intermediate stages, which drastically reduces overall throughput and increases material costs. These traditional methods frequently involve harsh reaction conditions that can compromise the integrity of sensitive functional groups, leading to lower purity profiles and higher impurity loads. Furthermore, the reliance on specific chiral starting materials that are difficult to source can create supply chain vulnerabilities, causing delays and price volatility for downstream manufacturers. The cumulative effect of these inefficiencies is a production process that is both economically burdensome and environmentally taxing, failing to meet the rigorous demands of modern pharmaceutical and nutritional ingredient standards. Consequently, there has been a persistent industry need for a streamlined alternative that can bypass these inherent structural limitations.
The Novel Approach
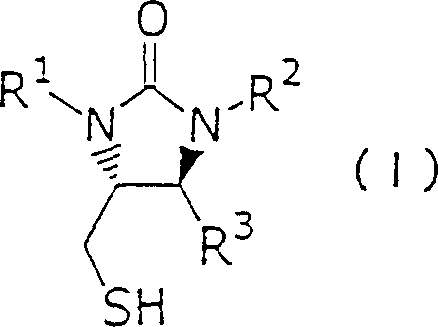
In stark contrast to legacy techniques, the method disclosed in CN1273453C utilizes a strategic ring transformation of compounds represented by general Formula (II-a) to generate the desired intermediates with superior efficiency. This novel approach allows for the direct construction of the core thienoimidazole scaffold without the need for the cumbersome optical resolution steps that characterize older pathways. By employing specific substituents such as benzyl, benzhydryl, or trityl groups on the nitrogen atoms, the process ensures high stereochemical control throughout the synthesis. The ability to convert precursors into compounds of Formula (I) or Formula (III) through a controlled ring conversion mechanism significantly shortens the production timeline. This reduction in synthetic steps translates directly into lower operational expenditures and a reduced carbon footprint, making it an attractive option for cost-conscious procurement managers. Moreover, the flexibility of the method allows for various substituents on the benzene rings, providing chemists with the versatility to optimize solubility and reactivity profiles for specific downstream applications. This represents a substantial leap forward in process chemistry, offering a scalable solution for the reliable biotin intermediate supplier market.
Mechanistic Insights into Ring Transformation and Cyclization
The core of this technological advancement lies in the precise mechanistic execution of the ring transformation and subsequent cyclization steps. The process begins with the activation of the starting material, typically a compound of Formula (II-a), which undergoes a structural rearrangement to form the cyclic urea derivative found in Formula (I). This transformation is facilitated by the careful selection of reaction conditions, including the use of inert atmospheres such as nitrogen or argon to prevent oxidative degradation of sensitive sulfur-containing moieties. The reaction kinetics are optimized by maintaining temperatures between 80°C and 100°C, which provides sufficient thermal energy to overcome activation barriers without promoting unwanted side reactions. Solvent choice plays a critical role, with polar aprotic solvents like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) being preferred to stabilize transition states and ensure homogeneous reaction mixtures. The presence of specific bases, ranging from alkali metal carbonates to organic amines, further drives the equilibrium towards the desired product by neutralizing acidic byproducts generated during the ring closure. This meticulous control over the reaction environment ensures that the stereochemistry at the asymmetric carbon centers is preserved, which is essential for the biological activity of the final biotin product.
Following the initial ring transformation, the pathway involves critical cyclization and epimerization steps to establish the final stereochemical configuration required for biological efficacy. The conversion of intermediates like Formula (I-a) into the fused ring system of Formula (III) requires precise activation, often utilizing coupling agents such as DCC or EDC·HCl to facilitate amide bond formation. The epimerization step is particularly crucial, as it ensures that the thermodynamically stable cis-fused ring system is obtained, which is a prerequisite for the subsequent side-chain attachment. This stage may be conducted under basic conditions using organic bases like DBU or triethylamine, or alternatively through thermal treatment alone, depending on the specific substituents employed. The ability to control the ratio of cis to trans isomers through these mechanistic adjustments allows for the production of high-purity intermediates with minimal chromatographic purification. Such high levels of stereocontrol are vital for R&D teams focused on impurity profiling, as it reduces the burden on downstream purification processes and ensures consistent quality across batches. The integration of these mechanistic insights into a cohesive process flow demonstrates the robustness of the patented technology.
How to Synthesize Biotin Intermediate Efficiently
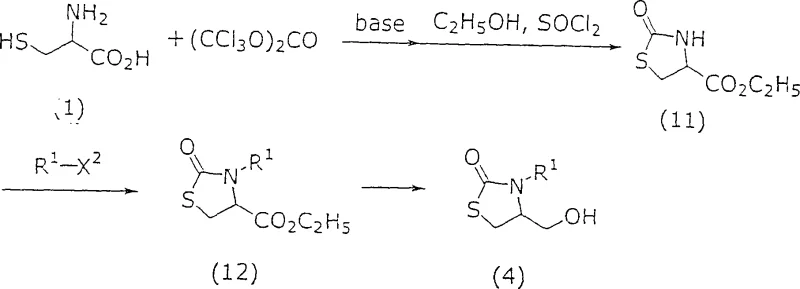
Implementing this synthesis route requires a thorough understanding of the operational parameters outlined in the patent to ensure reproducibility and safety on a commercial scale. The process is designed to be adaptable, allowing manufacturers to select from a range of commercially available starting materials such as cysteine derivatives and various chloroformates. The initial steps involve the formation of the thiazolidine ring, which serves as the foundation for the subsequent ring expansion to the imidazolidine system. Operators must pay close attention to the stoichiometry of reagents, particularly the base and activating agents, to maximize conversion rates while minimizing waste. The protocol emphasizes the importance of temperature control during the exothermic addition of reagents, ensuring that the reaction mixture remains within the optimal thermal window to prevent decomposition. Detailed standard operating procedures should be established to handle the specific solvents and reagents safely, adhering to all relevant environmental and safety regulations. For those seeking to adopt this technology, the following guide provides a high-level overview of the critical stages involved in transforming raw materials into high-value intermediates.
- Prepare the starting compound of Formula (II-a) and select an appropriate solvent system such as N,N-dimethylformamide or dimethyl sulfoxide.
- Conduct the ring transformation reaction under an inert gas flow like nitrogen or argon, maintaining temperatures between 80°C to 100°C for optimal conversion.
- Isolate the resulting Formula (I) intermediate through standard workup procedures including hydrolysis and crystallization to ensure high purity for downstream biotin synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers profound benefits for procurement managers and supply chain heads looking to optimize their vendor networks. The primary advantage lies in the significant reduction of manufacturing costs achieved through the elimination of expensive and time-consuming optical resolution steps. By streamlining the synthetic route, companies can reduce the consumption of raw materials and solvents, leading to substantial cost savings in the overall production budget. This efficiency gain is particularly relevant in the context of cost reduction in vitamin manufacturing, where margin pressures are often high. Furthermore, the use of readily available starting materials and standard reagents enhances supply chain reliability, reducing the risk of disruptions caused by the scarcity of specialized chiral catalysts. The robustness of the process also means that production timelines can be shortened, allowing for faster response times to market demand fluctuations. These factors combined create a more resilient supply chain capable of sustaining long-term commercial partnerships.
- Cost Reduction in Manufacturing: The elimination of complex optical resolution steps removes a major cost driver from the production process, allowing for more competitive pricing structures without compromising quality. The simplified workflow reduces labor hours and equipment usage, contributing to lower overheads and improved profit margins for manufacturers. Additionally, the higher yields associated with this method mean that less raw material is required to produce the same amount of final product, further driving down the cost of goods sold. This economic efficiency makes the technology highly attractive for large-scale production facilities aiming to maximize return on investment.
- Enhanced Supply Chain Reliability: By relying on common chemical reagents and avoiding proprietary chiral auxiliaries, the supply chain becomes less vulnerable to single-source dependencies and geopolitical disruptions. The flexibility of the synthesis allows for the substitution of various protecting groups, providing procurement teams with the agility to source materials from multiple vendors. This diversification strategy ensures continuous production capability even when specific raw materials face temporary shortages. Consequently, manufacturers can guarantee more consistent lead times for high-purity biotin intermediates, strengthening their reputation as a dependable partner in the global market.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and full-scale production. The reduction in synthetic steps inherently lowers the volume of chemical waste generated, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The use of standard solvents that can be recovered and recycled further minimizes the environmental footprint of the manufacturing operation. This commitment to green chemistry principles not only reduces disposal costs but also enhances the brand image of companies adopting this technology as responsible corporate citizens.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of this biotin intermediate synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits for potential adopters. Understanding these details is crucial for making informed decisions about integrating this method into existing production lines. The responses cover aspects ranging from reaction conditions to scalability, providing a comprehensive overview of the technology's capabilities.
Q: How does this new method improve upon conventional biotin intermediate synthesis?
A: The novel approach eliminates the need for complicated optical resolution steps in the intermediate stages, which are traditionally labor-intensive and costly, thereby streamlining the overall production workflow.
Q: What are the key reaction conditions for the ring transformation step?
A: The reaction is typically conducted in the absence of oxygen using solvents like DMF or DMSO, with temperatures ranging from 0°C to 200°C, specifically optimized around 80°C to 100°C for industrial efficiency.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly highlights the method's industrial applicability, noting that it allows for low-cost production and avoids the lengthy processes associated with older synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biotin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of fine chemical manufacturing. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale supply. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques. By leveraging advanced technologies like the one described in CN1273453C, we can offer our partners a reliable biotin intermediate supplier relationship built on technical excellence and operational reliability. Our infrastructure is designed to handle complex chemistries with precision, guaranteeing the consistency and quality required by top-tier pharmaceutical and nutritional companies.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements with a Customized Cost-Saving Analysis. Whether you need specific COA data for your quality assurance protocols or route feasibility assessments for new product development, we are ready to provide the detailed information you need. Partnering with us means gaining access to a supply chain that prioritizes innovation, efficiency, and long-term value creation. Let us help you optimize your production strategy and secure a competitive advantage in the global market for high-value nutritional ingredients.
