Advanced Manufacturing of 1,2-Bis(2-(2,6-Dimethoxyphenoxy)Ethoxy)Ethane for Energetic Materials
The development of high-energy solid propellants requires stabilizers that can effectively inhibit the autocatalytic decomposition of nitrate esters without compromising the mechanical integrity of the fuel matrix. Patent CN108002988B introduces a robust synthetic methodology for producing 1,2-bis(2-(2,6-dimethoxyphenoxy)ethoxy)ethane, a critical additive designed to enhance the chemical stability of nitrocellulose and nitroglycerin-based formulations. Unlike previous iterations that struggled with ambiguous product states and difficult purification, this invention utilizes a dicyclohexylcarbodiimide (DCC) mediated coupling strategy to achieve a definitive white powder solid with yields exceeding 84%. For R&D directors and procurement specialists in the energetic materials sector, this represents a significant leap forward in obtaining high-purity intermediates that meet stringent performance specifications while simplifying the manufacturing supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as the technique described by Patrick et al., relied on the nucleophilic substitution reaction between 2,6-dimethoxyphenol and halogenated polyethylene glycols, specifically 1,2-bis(2-chloroethoxy)ethane, using potassium carbonate as a base. This conventional pathway presents several inherent disadvantages for large-scale manufacturing, primarily due to the generation of stoichiometric amounts of inorganic salt byproducts like potassium chloride, which complicate downstream purification. Furthermore, the initial product obtained from this halide route often exists as a yellow liquid or oil that is notoriously difficult to separate and characterize, requiring multiple recrystallization steps to achieve acceptable purity. The necessity for nitrogen protection during the reaction adds operational complexity and cost, while the overall yield remains suboptimal at approximately 63%, leading to significant raw material wastage in commercial production environments.
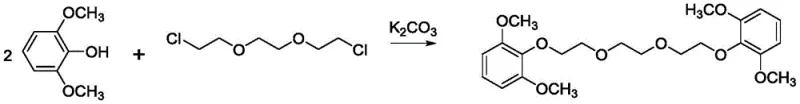
The Novel Approach
The patented innovation circumvents these challenges by employing a direct etherification strategy using triethylene glycol and DCC as a dehydrating coupling agent in an acetonitrile solvent system. This approach fundamentally alters the reaction profile by avoiding halogenated reagents entirely, thereby eliminating the formation of corrosive inorganic salts and reducing the environmental burden of waste disposal. The reaction proceeds efficiently at elevated temperatures without the need for inert gas blanketing, significantly lowering operational overheads. Most critically, the resulting crude product can be purified through a streamlined workup involving simple filtration of the urea byproduct followed by a single ether recrystallization step, yielding a well-defined white powder solid with purity levels surpassing 98% and reaction yields reaching up to 85.04%.
Mechanistic Insights into DCC-Mediated Etherification
The core of this synthetic breakthrough lies in the activation of the hydroxyl groups via the carbodiimide functionality of DCC, which facilitates the formation of the ether linkage between the phenolic oxygen and the glycol chain. In this mechanism, DCC acts as a potent dehydrating agent that promotes the condensation of the alcohol and phenol moieties, generating dicyclohexylurea (DCU) as the sole stoichiometric byproduct. The insolubility of DCU in many organic solvents allows for its facile removal via simple filtration, a distinct advantage over soluble inorganic bases that require extensive aqueous washing protocols. This mechanistic pathway ensures that the reaction mixture remains relatively clean, minimizing the formation of colored impurities often associated with oxidative degradation in base-catalyzed halide substitutions.
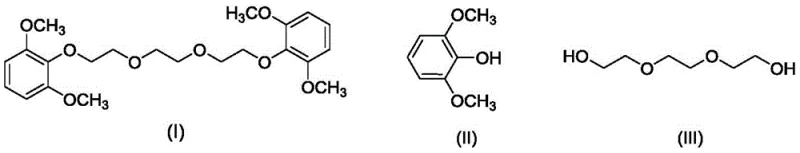
Impurity control is inherently built into this process design through the physical properties of the reaction components and the specific recrystallization conditions employed. The use of acetonitrile as the reaction solvent provides a homogeneous medium that supports the coupling efficiency, while the subsequent switch to dichloromethane for extraction allows for the selective partitioning of the organic product away from polar contaminants. The final purification step utilizes a precise temperature gradient in diethyl ether, cooling the solution to -15°C to induce the crystallization of the target molecule while keeping residual starting materials and side products in the supernatant. This rigorous control over phase transitions ensures that the final isolates possess the sharp melting point range of 59°C to 60°C required for consistent performance in propellant formulations.
How to Synthesize 1,2-Bis(2-(2,6-Dimethoxyphenoxy)Ethoxy)Ethane Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for laboratory and pilot-scale production, emphasizing precise temperature control and sequential solvent exchanges to maximize recovery. The process begins with the activation of the coupling partners in acetonitrile, followed by a strategic workup that leverages the solubility differences between the product and the urea byproduct. Operators must adhere strictly to the thermal profiles during the recrystallization phase, as the cooling rate directly influences the crystal habit and purity of the final white powder. Detailed standardized operating procedures for each unit operation, including specific molar ratios and washing sequences, are essential to replicate the reported 84% yield and >98% purity consistently across different batches.
- Coupling Reaction: Stir 2,6-dimethoxyphenol and triethylene glycol in acetonitrile with DCC at 85°C for 48-60 hours to form the ether linkage.
- Workup and Extraction: Filter off solid byproducts, extract with dichloromethane, and wash sequentially with sodium hydroxide and saturated sodium chloride solutions.
- Purification: Dissolve the crude oil in ether, filter, and perform controlled cooling crystallization at -15°C to isolate the white powder solid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this DCC-mediated process offers substantial logistical and economic benefits by simplifying the raw material portfolio and reducing waste management costs. The elimination of chlorinated polyethylene glycols removes the dependency on specialized halogenated intermediates, replacing them with commodity-grade triethylene glycol which is widely available and cost-effective. Additionally, the removal of inorganic salt byproducts significantly reduces the volume of aqueous waste streams requiring treatment, aligning with increasingly strict environmental regulations and lowering the total cost of ownership for manufacturing facilities. The robustness of the reaction, which does not require nitrogen protection, further decreases utility consumption and equipment complexity, facilitating easier technology transfer to larger production vessels.
- Cost Reduction in Manufacturing: The process achieves a dramatic improvement in atom economy and yield compared to prior art, directly translating to lower raw material consumption per kilogram of finished stabilizer. By avoiding the use of expensive halogenated reagents and reducing the number of recrystallization cycles from three to one, the operational expenditure associated with solvents and energy is significantly curtailed. The high yield of over 84% ensures that less feedstock is wasted, providing a clear margin advantage for high-volume production runs of energetic material additives.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key building blocks, 2,6-dimethoxyphenol and triethylene glycol, are established bulk chemicals with stable global supply chains. The reduced sensitivity of the reaction to atmospheric moisture, evidenced by the lack of need for nitrogen purging, minimizes the risk of batch failures due to equipment leaks or inert gas supply interruptions. This reliability ensures consistent delivery schedules for downstream propellant manufacturers who depend on a steady flow of high-quality stabilizers for their production lines.
- Scalability and Environmental Compliance: The generation of solid dicyclohexylurea as a byproduct simplifies waste handling compared to the saline wastewater produced by the potassium carbonate method, making regulatory compliance more straightforward. The process is inherently scalable because the filtration steps required to remove the urea are easily adapted to industrial filter presses, avoiding the bottlenecks often encountered in liquid-liquid extraction of emulsions. This scalability supports the seamless transition from kilogram-scale R&D batches to multi-ton commercial production without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the comparative data and experimental examples provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this new standard for stabilizer production. The answers reflect the specific improvements in yield, purity, and physical form that distinguish this method from historical precedents.
Q: What are the primary advantages of the DCC-mediated route over the traditional alkyl halide method?
A: The DCC-mediated route eliminates the need for hazardous chlorinated reagents and avoids the formation of inorganic salt byproducts like potassium chloride. This results in a clearer post-processing workflow, a definitive white solid product state rather than an ambiguous oil, and a significant yield increase from approximately 63% to over 84%.
Q: How does this synthesis method improve the stability of solid propellants?
A: This compound acts as a superior stabilizer by effectively absorbing nitrogen oxides and free radicals generated during nitrate ester decomposition. The high purity (>98%) achieved through the patented recrystallization process ensures consistent performance without the risk of crystallization migration or unwanted reactions with isocyanate curing agents.
Q: Is this process scalable for industrial production of energetic material additives?
A: Yes, the process is highly scalable as it utilizes commodity raw materials like triethylene glycol and avoids the need for inert gas protection (nitrogen) during the reaction. The solid byproduct (DCU) is easily removed by filtration, simplifying the scale-up compared to methods requiring extensive aqueous washing to remove inorganic salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Bis(2-(2,6-Dimethoxyphenoxy)Ethoxy)Ethane Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise to translate this patented DCC-mediated synthesis into a commercially viable manufacturing process tailored to your specific volume requirements. Our facility is equipped to handle complex etherification chemistries with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,2-bis(2-(2,6-dimethoxyphenoxy)ethoxy)ethane meets the exacting standards required for high-performance energetic applications.
We invite you to engage with our technical procurement team to discuss how this optimized route can drive value for your organization through improved efficiency and cost effectiveness. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior manufacturing method. Please contact us today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to delivering high-quality chemical solutions for the global market.
