Advanced Synthesis of 1,2-Bis(2-(4-Methoxyphenoxy)ethoxy)ethane for High-Energy Propellant Stabilization
Advanced Synthesis of 1,2-Bis(2-(4-Methoxyphenoxy)ethoxy)ethane for High-Energy Propellant Stabilization
The development of next-generation tactical high-energy solid propellants requires stabilizers that can effectively inhibit the autocatalytic decomposition of nitrate esters without interfering with the curing agents. Patent CN107935822B introduces a groundbreaking preparation method for 1,2-bis(2-(4-methoxyphenoxy)ethoxy)ethane, a critical additive designed to enhance the chemical stability of NEPE-type propellants. Unlike traditional synthetic pathways that rely on harsh halogenated reagents and strong bases, this innovative approach utilizes a dicyclohexylcarbodiimide (DCC) mediated coupling strategy. This shift represents a significant technological leap for manufacturers seeking a reliable propellant stabilizer supplier, as it addresses long-standing issues regarding environmental pollution and yield optimization in the production of energetic material intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
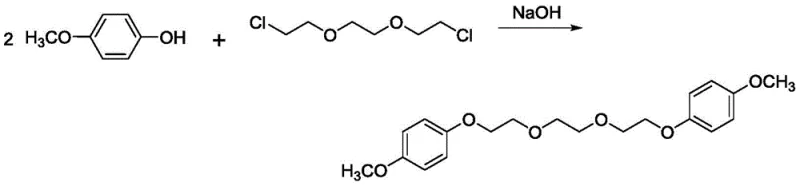
Historically, the synthesis of aromatic ether stabilizers like 1,2-bis(2-(4-methoxyphenoxy)ethoxy)ethane has relied heavily on nucleophilic substitution reactions involving halogenated precursors. As documented in prior art by Pursiainen et al. (1999), the conventional route employs 1,2-bis(2-chloroethoxy)ethane reacting with 4-methoxyphenol in the presence of strong alkali catalysts such as potassium hydroxide or sodium hydroxide. While this method can achieve yields around 96%, it presents severe drawbacks for modern industrial applications. The use of chlorine-containing reagents generates substantial amounts of inorganic salt waste, creating a heavy burden on wastewater treatment systems. Furthermore, the requirement for strong bases necessitates specialized corrosion-resistant reactor materials, increasing capital expenditure. The presence of residual chloride ions can also be detrimental to the long-term stability of the final propellant formulation, potentially accelerating degradation mechanisms.
The Novel Approach
The methodology disclosed in CN107935822B fundamentally reimagines this synthesis by replacing the halogenated electrophile with triethylene glycol and utilizing DCC as a dehydrating coupling agent. This transition from a substitution mechanism to a direct condensation eliminates the generation of stoichiometric salt byproducts. By operating in acetonitrile at moderate temperatures, the new process achieves a superior yield of up to 98% while producing a product with a sharp melting point of 67-68°C. This improvement is not merely incremental; it signifies a move towards greener chemistry that aligns with stringent global environmental regulations. For procurement teams focused on cost reduction in energetic material manufacturing, this route offers a compelling value proposition by simplifying the purification workflow and reducing the consumption of hazardous raw materials.
Mechanistic Insights into DCC-Mediated Etherification
The core innovation of this patent lies in the activation of the hydroxyl groups of triethylene glycol using dicyclohexylcarbodiimide. In this mechanism, DCC reacts with the alcohol functionality to form an O-acylisourea-like intermediate or a similar activated species that is highly susceptible to nucleophilic attack by the phenolic oxygen of 4-methoxyphenol. This activation lowers the energy barrier for the formation of the ether bond, allowing the reaction to proceed efficiently at 85°C without the need for aggressive basic conditions. The reaction stoichiometry is carefully balanced, typically employing a molar ratio of triethylene glycol to 4-methoxyphenol to DCC of approximately 1:2.2:2.4. This slight excess of the phenol and coupling agent ensures complete conversion of the glycol, minimizing the formation of mono-substituted byproducts which could act as impurities in the final stabilizer application.
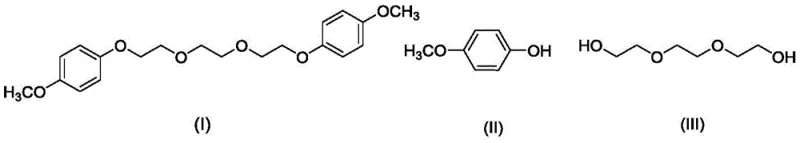
Impurity control is paramount in the synthesis of high-purity 1,2-bis(2-(4-methoxyphenoxy)ethoxy)ethane, as trace contaminants can compromise the shelf-life of sensitive propellant formulations. The patented process incorporates a robust workup procedure that leverages the solubility differences between the desired product and the urea byproduct (dicyclohexylurea) generated during the coupling. Following the reaction, the mixture is treated with dichloromethane and washed with 1M sodium hydroxide solution. This alkaline wash is crucial not for catalysis, but for removing unreacted phenolic starting materials and acidic impurities. Subsequent washing with saturated sodium chloride and drying over anhydrous magnesium sulfate ensures the removal of water and polar residues. The final crystallization from diethyl ether at low temperatures (-15°C) further refines the crystal lattice, excluding remaining organic impurities and delivering a white powdery solid with purity exceeding 98%.
How to Synthesize 1,2-Bis(2-(4-Methoxyphenoxy)ethoxy)ethane Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize the formation of dicyclohexylurea precipitates which can complicate filtration. The process begins with the dissolution of reactants in acetonitrile, followed by the controlled addition of the coupling agent to manage exotherms. Detailed operational protocols, including specific temperature ramps and filtration techniques, are essential for reproducibility on a larger scale. The following guide outlines the standardized steps derived from the patent examples to ensure consistent production quality.
- Combine 4-methoxyphenol and triethylene glycol in acetonitrile at 20°C, then add dicyclohexylcarbodiimide (DCC) and heat to 85°C for 24 hours to facilitate coupling.
- Filter the reaction mixture to remove solids, distill off acetonitrile under reduced pressure, and extract the residue with dichloromethane followed by washing with 1M NaOH and brine.
- Evaporate the solvent to obtain a yellow solid, dissolve in diethyl ether, filter insolubles, cool to -15°C for crystallization, and filter to isolate the white powdery product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this DCC-mediated synthesis offers distinct strategic advantages over legacy chlorinated routes. The primary benefit is the drastic simplification of the waste management profile. By eliminating the production of stoichiometric quantities of sodium chloride and avoiding chlorinated organic waste, manufacturers can significantly reduce the costs associated with hazardous waste disposal and environmental compliance. This reduction in regulatory burden translates directly into improved operational margins and a more resilient supply chain that is less vulnerable to changes in environmental legislation.
- Cost Reduction in Manufacturing: Although the coupling reagent DCC is more expensive per kilogram than sodium hydroxide, the overall process economics are favorable due to the elimination of multiple downstream processing steps. The absence of corrosive salts reduces maintenance costs for reactor vessels and piping, extending the lifespan of capital equipment. Furthermore, the higher yield of 98% means less raw material is wasted per unit of finished product, optimizing the utilization of 4-methoxyphenol and triethylene glycol. This efficiency gain allows for a more competitive pricing structure for the final stabilizer without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically triethylene glycol and 4-methoxyphenol, are commodity chemicals with robust global supply chains. Unlike specialized halogenated intermediates which may be subject to supply constraints or geopolitical trade restrictions, these feedstocks are readily available from multiple vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Additionally, the milder reaction conditions reduce the safety risks associated with handling strong bases and reactive halides, leading to fewer unplanned shutdowns and a more consistent delivery schedule for customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward because it avoids the engineering challenges posed by highly exothermic alkali reactions. The use of acetonitrile as a solvent allows for efficient heat transfer and easy recovery through distillation. From an environmental perspective, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). This compliance is increasingly critical for suppliers serving the defense and aerospace sectors, where contractors are mandated to source materials from environmentally responsible manufacturers. The ability to provide a "greener" stabilizer enhances the marketability of the final propellant system.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced stabilizer. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the technology's capabilities.
Q: How does the DCC-mediated method improve upon the traditional Pursiainen synthesis route?
A: The novel DCC-mediated route eliminates the use of hazardous chlorinated reagents like 1,2-bis(2-chloroethoxy)ethane and strong alkaline catalysts such as sodium hydroxide. This significantly reduces corrosive waste streams and simplifies downstream purification, resulting in a cleaner product profile with yields reaching up to 98% compared to the 96% typical of the older halogenated method.
Q: What are the critical purity specifications for this propellant stabilizer?
A: For application in tactical high-energy solid propellants, the stabilizer must exhibit exceptional purity to prevent unintended catalytic decomposition of nitrate esters. The patented process achieves purity levels exceeding 98%, with specific melting points between 67-68°C, ensuring consistent performance and stability during long-term storage of energetic formulations.
Q: Is this synthesis route scalable for industrial production of energetic material additives?
A: Yes, the process utilizes commodity chemicals like triethylene glycol and operates at moderate temperatures (85°C) in standard solvents like acetonitrile. The absence of highly corrosive reagents and the straightforward crystallization workup make it highly amenable to commercial scale-up, reducing the engineering complexity often associated with halogenated ether syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Bis(2-(4-Methoxyphenoxy)ethoxy)ethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance stabilizers play in the reliability of modern energetic systems. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless. We are committed to delivering 1,2-bis(2-(4-methoxyphenoxy)ethoxy)ethane with stringent purity specifications, supported by our rigorous QC labs that utilize advanced NMR and HPLC techniques to verify every batch. Our facility is equipped to handle the specific solvent recovery and crystallization requirements of this DCC-mediated process, guaranteeing a consistent supply of this vital intermediate.
We invite R&D directors and procurement specialists to engage with us for a Customized Cost-Saving Analysis tailored to your specific formulation needs. By optimizing the supply chain for this key stabilizer, we can help you reduce overall program costs while enhancing the safety profile of your products. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
