Revolutionizing Organometallic Synthesis: Safe Production of Ferrocene-Phosphazene Hybrids for Global Markets
Revolutionizing Organometallic Synthesis: Safe Production of Ferrocene-Phosphazene Hybrids for Global Markets
The development of advanced functional materials often hinges on the ability to combine distinct chemical moieties into a single, stable architecture. Patent CN102276659A introduces a groundbreaking methodology for synthesizing monoacyl ferrocene Schiff base aminocyclotriphosphazene, a hybrid compound that merges the redox-active properties of ferrocene with the thermal stability and versatility of the cyclotriphosphazene matrix. This innovation addresses critical bottlenecks in the production of high-performance organometallic intermediates used in electrochemistry, medicine, and catalysis. By circumventing the need for hazardous organolithium reagents, this technology offers a robust pathway for creating complex molecular structures that were previously difficult to manufacture safely and economically. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable specialty chemical supplier capable of delivering high-value intermediates with improved safety profiles and consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of ferrocene groups into phosphazene matrices has relied heavily on classical organometallic strategies that pose substantial operational risks and cost inefficiencies. Traditional literature, such as reports in the Journal of the American Chemical Society, describes routes involving the generation of ferrocenyl lithium at cryogenic temperatures using butyllithium. These processes are inherently dangerous due to the pyrophoric nature of the reagents and the strict requirement for moisture-free, low-temperature environments. Furthermore, the handling of such highly active species necessitates specialized equipment and rigorous safety protocols, which drastically inflate capital expenditure and operational costs. The complexity of these routes often leads to lower yields and difficulties in purification, creating supply chain vulnerabilities for manufacturers seeking high-purity organometallic intermediates. Consequently, the industry has long sought alternative synthetic pathways that mitigate these risks while maintaining structural integrity.
The Novel Approach
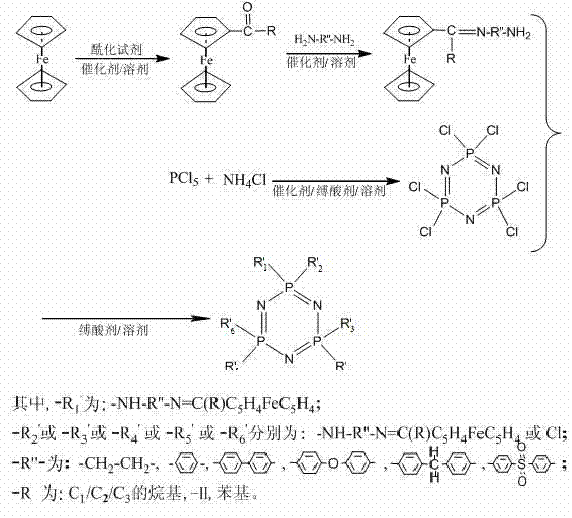
The patented methodology presented in CN102276659A fundamentally reengineers the synthesis workflow by replacing hazardous lithiation steps with a sequence of mild acylation and condensation reactions. This novel approach begins with the acylation of ferrocene to form monoacyl ferrocene, followed by condensation with a diamine to generate an amino-terminated Schiff base. This intermediate is then coupled with hexachlorocyclotriphosphazene through a nucleophilic substitution reaction. The entire process can be conducted at ambient or moderately elevated temperatures, utilizing common organic solvents and readily available reagents. This shift not only enhances operator safety but also simplifies the engineering requirements for production facilities. By avoiding extreme conditions, the process facilitates easier scale-up and reduces the total cost of ownership for manufacturing these complex derivatives. This strategic pivot enables cost reduction in fine chemical manufacturing by streamlining the workflow and minimizing waste associated with hazardous reagent quenching.
Mechanistic Insights into Nucleophilic Substitution on Cyclotriphosphazene
The core chemical transformation in this synthesis involves the nucleophilic substitution of chlorine atoms on the hexachlorocyclotriphosphazene ring by the amino groups of the ferrocene Schiff base. This reaction is driven by the high electrophilicity of the phosphorus atoms in the phosphazene ring, which are activated by the adjacent electronegative nitrogen atoms. The presence of an acid-binding agent, such as triethylamine, is crucial to neutralize the hydrogen chloride byproduct generated during the substitution, thereby driving the equilibrium towards the desired product. The reaction typically proceeds at temperatures between 60°C and 80°C, allowing for controlled substitution without degrading the sensitive Schiff base linkage. This mechanistic pathway ensures that the ferrocene moiety is securely anchored to the phosphazene core, preserving the redox activity of the iron center while imparting the thermal stability of the inorganic backbone. Understanding this mechanism is vital for R&D teams aiming to optimize reaction kinetics and minimize the formation of polymeric by-products.
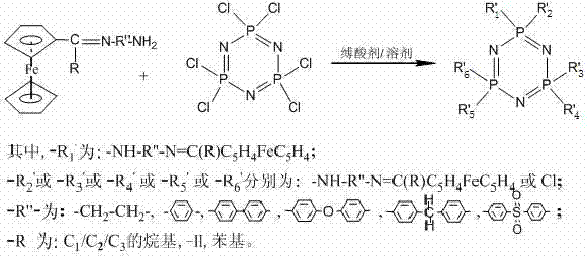
Impurity control is another critical aspect of this mechanistic design, particularly regarding the degree of substitution on the phosphazene ring. The stoichiometry of the reactants can be tuned to favor mono-substitution or multi-substitution, allowing for the customization of the final product's properties. The patent specifies molar ratios ranging from 1:1.0 to 1:6.0 between the phosphazene and the Schiff base, providing flexibility in designing derivatives with specific functional densities. Rigorous purification steps, including washing with acetone and water followed by vacuum drying, are employed to remove unreacted starting materials and salts. This attention to detail in the workup phase ensures that the final high-purity organometallic intermediates meet the stringent specifications required for applications in solid electrolytes and optoelectronic materials. Such precise control over the impurity profile is essential for maintaining the performance consistency of the end-use products.
How to Synthesize Monoacyl Ferrocene Schiff Base Aminocyclotriphosphazene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable compounds with high reproducibility. The process is divided into three distinct stages: the preparation of the phosphazene core, the synthesis of the ferrocene Schiff base ligand, and the final coupling reaction. Each stage utilizes standard laboratory glassware and heating mantles, making it accessible for both pilot-scale and commercial production environments. The detailed procedure emphasizes the importance of inert atmospheres and anhydrous conditions during the initial acylation and substitution steps to prevent hydrolysis of the reactive intermediates. For a comprehensive guide on the specific temperature ramps, stirring rates, and workup procedures, please refer to the standardized synthesis steps provided below.
- Synthesize hexachlorocyclotriphosphazene by reacting phosphorus pentachloride with ammonium chloride in an inert solvent with an acid-binding agent at 100-180°C.
- Prepare monoacyl ferrocene via acylation, then react with a diamine to form an amino-terminated ferrocene Schiff base intermediate.
- Perform nucleophilic substitution by reacting the ferrocene Schiff base with hexachlorocyclotriphosphazene in the presence of an acid scavenger at 60-80°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for advanced materials. The elimination of pyrophoric reagents like butyllithium removes a major bottleneck in logistics and storage, as these materials often require special shipping classifications and dedicated storage facilities. By switching to stable, shelf-stable reagents, companies can significantly reduce their inventory holding costs and insurance premiums. Furthermore, the simplified reaction conditions allow for the use of standard multipurpose reactors, increasing asset utilization rates and reducing the need for dedicated cryogenic infrastructure. This operational flexibility translates directly into substantial cost savings and enhanced supply chain reliability, ensuring that production schedules are not disrupted by reagent availability or equipment maintenance issues.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive and hazardous organolithium reagents with commodity chemicals such as acyl chlorides and diamines. This substitution drastically lowers the raw material bill of materials while simultaneously reducing the costs associated with hazardous waste disposal. The milder reaction temperatures also result in lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and reduced utility expenses. Additionally, the higher selectivity of the nucleophilic substitution minimizes the formation of difficult-to-separate by-products, improving overall yield and reducing the load on downstream purification units. These factors combine to create a highly efficient manufacturing process that delivers significant economic value.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials that are not subject to the same geopolitical or logistical constraints as specialized organometallic reagents. Ferrocene and phosphorus pentachloride are produced at large scales globally, ensuring a steady flow of inputs even during market fluctuations. The robustness of the synthesis method also means that production can be easily transferred between different manufacturing sites without the need for extensive requalification of specialized equipment. This flexibility allows for diversified sourcing strategies and reduces the risk of single-point failures in the supply network. Consequently, customers can enjoy reducing lead time for high-purity ferrocene derivatives and more predictable delivery schedules.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with lithiation. The reaction kinetics are manageable in large vessels, allowing for the commercial scale-up of complex phosphazene derivatives without compromising safety. From an environmental standpoint, the process generates less hazardous waste, primarily consisting of amine hydrochloride salts which are easier to treat than organolithium quench residues. This aligns with increasingly strict environmental regulations and corporate sustainability goals. The ability to produce these materials with a lower environmental impact enhances the marketability of the final products to eco-conscious industries such as green energy and sustainable electronics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of monoacyl ferrocene Schiff base aminocyclotriphosphazene. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the operational benefits and performance characteristics of this innovative synthetic route for potential partners and technical stakeholders.
Q: How does this synthesis method improve safety compared to traditional organolithium routes?
A: Traditional methods often require highly reactive and pyrophoric reagents like butyllithium at cryogenic temperatures. This patented process utilizes mild acylation and condensation reactions at ambient or moderate temperatures, significantly reducing operational hazards and eliminating the need for specialized low-temperature equipment.
Q: What are the key purity specifications for the ferrocene-phosphazene intermediate?
A: High purity is critical for electrochemical and catalytic applications. The process includes rigorous purification steps such as recrystallization and vacuum sublimation, ensuring the removal of linear by-products and unreacted starting materials to meet stringent quality standards for advanced material synthesis.
Q: Is this process scalable for industrial production of specialty catalysts?
A: Yes, the reaction conditions avoid extreme parameters and utilize standard organic solvents and reagents. The elimination of sensitive organometallic reagents allows for straightforward scale-up in standard stainless steel reactors, facilitating reliable commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monoacyl Ferrocene Schiff Base Aminocyclotriphosphazene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented technology in advancing the fields of electrochemistry and catalysis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic synthesis, including inert atmosphere operations and rigorous QC labs that enforce stringent purity specifications. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical and specialty chemical industries, providing you with a competitive edge in your respective markets.
We invite you to collaborate with us to leverage this advanced synthesis method for your next generation of functional materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your production costs.
