Advanced Catalytic Hydrogenation for Midodrine Intermediates: Scaling Safe and Efficient Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency with stringent safety standards, particularly for cardiovascular therapeutics like Midodrine hydrochloride. Patent CN102060719A introduces a transformative methodology for preparing the critical intermediate 1-(2,5-dimethoxyphenyl)-2-aminoethanol, addressing long-standing challenges in toxicity and cost. This innovation shifts the paradigm from hazardous stoichiometric reductions to a cleaner catalytic hydrogenation process, leveraging nitromethane condensation as a key strategic step. For global procurement leaders and R&D directors, this patent represents a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials without the baggage of environmentally damaging byproducts. The technical breakthrough lies not merely in the chemical transformation but in the holistic optimization of reaction conditions, solvent systems, and workup procedures that collectively enhance the economic feasibility of large-scale manufacturing.
Furthermore, the adoption of this technology aligns with modern green chemistry principles, which are increasingly becoming a prerequisite for vendor qualification in top-tier supply chains. By eliminating the need for heavy metal waste streams associated with traditional reducing agents, manufacturers can significantly reduce their environmental footprint and regulatory compliance costs. This report analyzes the technical nuances of this patent to demonstrate how it serves as a cornerstone for cost reduction in API manufacturing, offering a competitive edge through improved safety profiles and operational simplicity. As we delve into the mechanistic details and commercial implications, it becomes evident that this synthesis route is not just a laboratory curiosity but a commercially viable solution ready for industrial deployment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for 1-(2,5-dimethoxyphenyl)-2-aminoethanol have been plagued by significant safety hazards and economic inefficiencies that hinder scalable production. As illustrated in prior art such as WO9638143, the reliance on potassium cyanide for condensation reactions introduces extreme toxicity risks, requiring specialized containment infrastructure and rigorous personnel protection protocols that drive up operational expenditures. 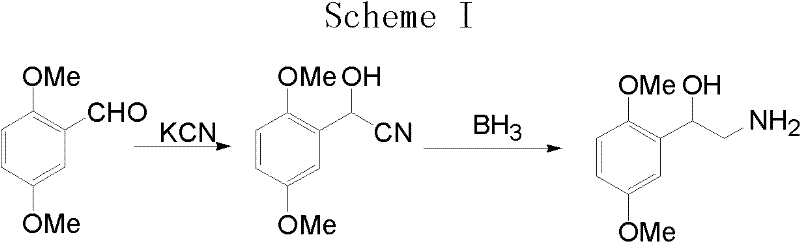 Furthermore, the subsequent reduction steps often employ borane or lithium aluminum hydride, reagents that are not only expensive but also generate substantial amounts of hazardous waste that are difficult and costly to treat. The cumulative effect of these factors results in a fragile supply chain where yield losses due to safety incidents or regulatory shutdowns can cause significant disruptions. Additionally, the use of column chromatography for purification in early-stage processes is inherently unscalable, creating a bottleneck that prevents the seamless transition from pilot plant to commercial tonnage production. These legacy methods fail to meet the modern demands for sustainable and cost-effective pharmaceutical manufacturing.
Furthermore, the subsequent reduction steps often employ borane or lithium aluminum hydride, reagents that are not only expensive but also generate substantial amounts of hazardous waste that are difficult and costly to treat. The cumulative effect of these factors results in a fragile supply chain where yield losses due to safety incidents or regulatory shutdowns can cause significant disruptions. Additionally, the use of column chromatography for purification in early-stage processes is inherently unscalable, creating a bottleneck that prevents the seamless transition from pilot plant to commercial tonnage production. These legacy methods fail to meet the modern demands for sustainable and cost-effective pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN102060719A offers a streamlined and safer alternative that fundamentally reengineers the synthesis logic to prioritize industrial applicability. By utilizing a nitroaldol condensation between 2,5-dimethoxy benzaldehyde and nitromethane, the process avoids the use of cyanide entirely, thereby removing a major category of occupational hazard from the production floor. The subsequent reduction of the nitro group is achieved through catalytic hydrogenation, a technique renowned for its atom economy and cleanliness compared to stoichiometric metal hydride reductions. This shift allows for the use of common, recoverable solvents like saturated fatty alcohols and ethanol, which simplifies solvent recovery systems and lowers raw material costs. The result is a robust process that maintains high purity standards while drastically simplifying the operational workflow, making it an ideal candidate for partners seeking a reliable pharmaceutical intermediates supplier with a focus on safety and efficiency.
Mechanistic Insights into Nitroaldol Condensation and Catalytic Hydrogenation
The core chemical innovation rests on the precise control of the Henry reaction conditions, where the interaction between the aldehyde and nitromethane is mediated by alkali catalysts in a specific solvent environment. The patent specifies the use of saturated fatty alcohols or mixtures with water, which likely play a dual role in solubilizing reactants and moderating the exothermic nature of the condensation. Temperature control between -10°C and 20°C is critical to minimizing side reactions such as polymerization or over-nitration, ensuring that the intermediate 1-(2,5-dimethoxyphenyl)-2-nitroethyl alcohol is formed with high selectivity. This selectivity is paramount for the R&D Director, as it directly correlates to the impurity profile of the final product, reducing the burden on downstream purification units. The careful neutralization step following condensation further ensures that residual base does not interfere with the subsequent hydrogenation catalyst, preserving its activity and longevity throughout the reaction cycle.
Following the formation of the nitro intermediate, the catalytic hydrogenation step employs transition metals such as nickel, palladium, or platinum supported on carbon or alumina to facilitate the reduction of the nitro group to an amine. 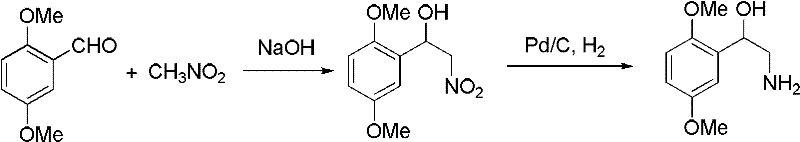 This heterogeneous catalysis approach offers distinct advantages over homogeneous reductions, primarily in the ease of catalyst separation and reuse, which contributes to significant cost reduction in API manufacturing. The reaction proceeds under hydrogen pressure ranging from 0.1 to 50 MPa, allowing for flexibility in reactor design and throughput optimization. The presence of acid additives during this step helps to stabilize the amine product as a salt, preventing potential degradation or side reactions that could compromise the high-purity midodrine intermediate specifications. Understanding these mechanistic subtleties allows process chemists to fine-tune parameters for maximum yield and minimal waste, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds without technical hurdles.
This heterogeneous catalysis approach offers distinct advantages over homogeneous reductions, primarily in the ease of catalyst separation and reuse, which contributes to significant cost reduction in API manufacturing. The reaction proceeds under hydrogen pressure ranging from 0.1 to 50 MPa, allowing for flexibility in reactor design and throughput optimization. The presence of acid additives during this step helps to stabilize the amine product as a salt, preventing potential degradation or side reactions that could compromise the high-purity midodrine intermediate specifications. Understanding these mechanistic subtleties allows process chemists to fine-tune parameters for maximum yield and minimal waste, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds without technical hurdles.
How to Synthesize 1-(2,5-Dimethoxyphenyl)-2-aminoethanol Efficiently
Implementing this synthesis route requires a disciplined approach to process parameters to replicate the high yields and purity reported in the patent data. The procedure begins with the preparation of the nitro-alcohol intermediate under strictly controlled thermal conditions, followed by a filtration and drying step that prepares the solid for the reduction phase. Operators must ensure that the hydrogenation reactor is properly purged of oxygen to prevent safety hazards and catalyst deactivation, a standard protocol in modern fine chemical facilities. The final recrystallization from mixed solvent systems like toluene and isopropyl ether is crucial for achieving the target purity levels required for pharmaceutical applications.
- Condense 2,5-dimethoxy benzaldehyde with nitromethane using an alkali catalyst in saturated fatty alcohol solvent at -10°C to 20°C.
- Perform catalytic hydrogenation on the resulting nitroethyl alcohol using a metal catalyst (Ni, Pd, or Pt) under hydrogen pressure.
- Purify the crude product via recrystallization using organic solvents such as toluene or isopropyl ether to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology offers tangible benefits that extend beyond simple chemical yield, impacting the overall total cost of ownership for the intermediate. By eliminating the need for highly regulated and dangerous reagents like potassium cyanide and sodium azide, facilities can reduce their insurance premiums and waste disposal fees, leading to substantial cost savings over the lifecycle of the product. The simplified workflow also means shorter batch cycles and reduced equipment downtime, which enhances the overall capacity utilization of the manufacturing plant. This efficiency translates into a more resilient supply chain capable of meeting tight deadlines without compromising on quality or safety standards. Furthermore, the use of catalytic hydrogenation aligns with global sustainability goals, making the supply chain more attractive to environmentally conscious stakeholders and regulatory bodies.
- Cost Reduction in Manufacturing: The replacement of expensive stoichiometric reducing agents like lithium aluminum hydride with catalytic hydrogenation significantly lowers raw material expenses. Since the catalyst can be recovered and reused, the recurring cost per kilogram of product is drastically reduced compared to traditional methods. Additionally, the avoidance of complex chromatographic purification steps reduces solvent consumption and labor costs, contributing to a leaner manufacturing budget. These cumulative savings allow for more competitive pricing structures without sacrificing margin, providing a strategic advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as 2,5-dimethoxy benzaldehyde and nitromethane ensures that raw material sourcing is stable and less prone to geopolitical disruptions. The robustness of the catalytic process means fewer batch failures due to sensitivity issues, guaranteeing a consistent output of high-purity active ingredients. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers can maintain their own production schedules without interruption. A stable supply of key intermediates is the backbone of a dependable pharmaceutical supply chain.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, avoiding unit operations that are difficult to scale such as column chromatography. The reduction in hazardous waste generation simplifies environmental compliance and permits management, reducing the risk of regulatory fines or shutdowns. This eco-friendly profile supports the long-term viability of the manufacturing site and aligns with corporate social responsibility initiatives. Scalability ensures that as demand for Midodrine grows, the production capacity can be expanded seamlessly to meet market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent's background and beneficial effects. Understanding these aspects helps stakeholders make informed decisions about adopting this route for their supply chains. The answers reflect the specific advantages of the catalytic hydrogenation method over legacy technologies, focusing on safety, purity, and cost.
Q: Why is the catalytic hydrogenation method preferred over cyanide-based routes for Midodrine intermediates?
A: Traditional methods utilizing potassium cyanide pose severe toxicity risks and require expensive waste treatment. The catalytic hydrogenation route described in CN102060719A eliminates hazardous cyanide reagents, significantly improving operational safety and environmental compliance while maintaining high yield.
Q: What are the purity specifications achievable with this novel synthesis route?
A: Patent data indicates that through optimized recrystallization processes, HPLC purity levels exceeding 98% can be consistently achieved. This high purity reduces the burden on downstream purification steps during final API synthesis.
Q: How does this method impact the commercial scalability of Midodrine production?
A: By replacing expensive and dangerous reducing agents like lithium aluminum hydride with catalytic hydrogenation, the process becomes more cost-effective and safer to operate at large scales. The use of common solvents and mild reaction conditions facilitates easier technology transfer and commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2,5-Dimethoxyphenyl)-2-aminoethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale patent data to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 1-(2,5-dimethoxyphenyl)-2-aminoethanol meets the highest international standards. Our infrastructure is designed to handle complex chemistries safely, leveraging the very catalytic hydrogenation techniques described in leading patents to deliver superior value to our partners.
We invite you to collaborate with us to optimize your supply chain for Midodrine production and achieve your cost and quality targets. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us be your partner in driving innovation and efficiency in pharmaceutical manufacturing.
