Advanced Biocatalytic Oxidation for High-Purity Chiral Sulfoxide Commercial Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct chiral centers, which are critical for the biological activity of modern drugs. Patent CN109402074B introduces a groundbreaking advancement in this field by disclosing specific monooxygenase mutants that dramatically improve the stereoselective oxidation of thioether compounds. This technology addresses the long-standing challenges associated with the industrial production of chiral sulfoxides, which serve as vital intermediates for blockbuster drugs such as Modafinil and Omeprazole. By leveraging directed evolution techniques, the inventors have successfully engineered enzyme variants that possess superior catalytic activity and stability compared to their wild-type counterparts. This innovation not only enhances the enantiomeric purity of the final product but also significantly mitigates the formation of over-oxidized sulfone byproducts, a common issue in traditional chemical oxidation methods. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and cost-effective biocatalytic manufacturing processes that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical methods for synthesizing chiral sulfoxides often rely on harsh oxidizing agents and transition metal catalysts, which pose significant environmental and safety risks during large-scale manufacturing. These conventional routes frequently suffer from poor stereoselectivity, resulting in racemic mixtures that require expensive and time-consuming chiral resolution steps to isolate the desired enantiomer. Furthermore, the lack of specificity in chemical oxidation often leads to the formation of sulfone byproducts, which are difficult to separate from the target sulfoxide and can compromise the purity profile required for pharmaceutical applications. The use of heavy metals also introduces stringent regulatory hurdles regarding residual metal limits in the final active pharmaceutical ingredient, necessitating additional purification stages that drive up production costs and extend lead times. Consequently, the supply chain for high-purity chiral sulfoxides via chemical routes is often fragile and susceptible to disruptions caused by environmental compliance issues or raw material volatility.
The Novel Approach
In contrast, the novel biocatalytic approach detailed in the patent utilizes engineered Baeyer-Villiger Monooxygenases (BVMOs) to achieve highly selective oxidation under mild aqueous conditions. This method eliminates the need for toxic heavy metals and harsh reagents, thereby simplifying the workflow and reducing the environmental footprint of the manufacturing process. The specific mutants described exhibit a remarkable ability to discriminate between enantiomers, ensuring that the resulting chiral sulfoxide meets stringent purity specifications without the need for complex resolution techniques. By operating at moderate temperatures and neutral pH levels, this enzymatic process preserves the integrity of sensitive functional groups on the substrate, which is crucial for complex pharmaceutical intermediates. The transition from chemical to enzymatic catalysis represents a paradigm shift that offers a reliable chiral sulfoxide supplier the ability to deliver higher quality products with greater consistency and reduced operational risk.
Mechanistic Insights into BVMO-Catalyzed Stereoselective Oxidation
The core of this technological breakthrough lies in the precise modification of the amino acid sequence of the monooxygenase enzyme, specifically targeting key residues that influence substrate binding and catalytic efficiency. The patent identifies multiple mutation sites, including positions 190, 559, and 560, which when altered, induce conformational changes that optimize the enzyme's active site for the specific thioether substrate. These structural adjustments enhance the interaction between the flavin cofactor and the sulfur atom of the substrate, facilitating a more efficient oxygen transfer mechanism that favors the formation of the sulfoxide over the sulfone. The improved enzyme activity means that lower loading amounts of the biocatalyst are required to achieve complete conversion, which directly translates to reduced material costs and simplified downstream processing. Furthermore, the enhanced stability of these mutants allows for longer operational lifespans in industrial reactors, ensuring consistent performance over extended production runs and minimizing the frequency of enzyme replenishment.
Controlling impurity profiles is paramount in the synthesis of pharmaceutical intermediates, and the engineered mutants excel in suppressing the formation of the undesired sulfone byproduct. The mechanistic advantage stems from the mutated enzyme's ability to release the sulfoxide product more rapidly before it can undergo a second oxidation step to form the sulfone. This kinetic control is critical for maintaining high yields and reducing the burden on purification units, which often struggle to separate compounds with similar physicochemical properties. By minimizing byproduct generation at the source, the process inherently improves the overall mass balance and reduces waste generation, aligning with the principles of atom economy. For quality control teams, this means a more robust process with fewer variables, leading to batch-to-batch consistency that is essential for regulatory approval and commercial success in the global market.
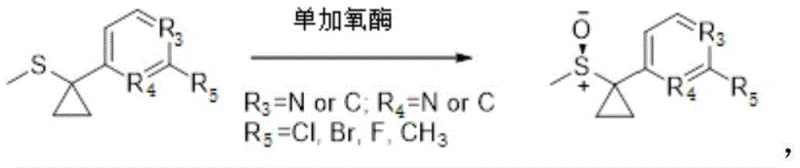
How to Synthesize Chiral Sulfoxide Efficiently
The implementation of this biocatalytic route involves a streamlined workflow that begins with the preparation of the engineered enzyme, typically expressed in robust host systems like E. coli for high-yield production. The reaction is conducted in an aqueous buffer system supplemented with necessary cofactors such as NADP+, which can be regenerated in situ using coupled enzyme systems to minimize cost. Operators must carefully control reaction parameters including temperature, which is optimally maintained between 10-50°C, and pH, which is preferably kept around 9 to maximize enzyme activity and stability. The substrate, often a complex thioether derivative, is introduced to the reaction mixture where the mutant monooxygenase facilitates the stereoselective oxygen insertion. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system with the specific thioether substrate and the engineered monooxygenase mutant enzyme solution.
- Maintain the reaction temperature between 10-50°C and pH 7-10, ensuring the presence of necessary cofactors like NADP+.
- Monitor the conversion to ensure high enantiomeric excess and minimal sulfone byproduct formation before downstream purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this mutant enzyme technology offers compelling strategic advantages that extend beyond mere technical performance. The primary benefit is the significant reduction in manufacturing costs driven by the enhanced catalytic efficiency of the mutants, which allows for lower enzyme loading while maintaining high conversion rates. This efficiency gain reduces the overall consumption of biocatalyst materials and minimizes the volume of waste generated, leading to substantial cost savings in waste disposal and raw material procurement. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling, further contributing to a lower carbon footprint and operational expenditure. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations in raw material prices.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in enzyme usage directly lower the bill of materials for each production batch. By avoiding the need for complex chiral resolution steps, the process significantly shortens the production cycle, thereby reducing labor and facility occupancy costs. The high selectivity also minimizes the loss of valuable starting materials to byproducts, improving the overall yield and economic viability of the synthesis. These cumulative efficiencies result in a more competitive pricing structure for the final chiral intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Biocatalytic processes are generally less dependent on volatile petrochemical feedstocks, offering a more sustainable and stable source of raw materials for long-term production planning. The robustness of the engineered enzymes ensures consistent output even under varying operational conditions, reducing the risk of batch failures that can disrupt supply schedules. Furthermore, the aqueous nature of the reaction simplifies logistics and storage requirements compared to hazardous chemical oxidants, enhancing safety and compliance across the supply network. This reliability is crucial for maintaining continuous production flows for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The technology is inherently scalable, having been validated from laboratory screening to potential industrial application through directed evolution strategies that prioritize stability. The reduction in hazardous waste and the absence of heavy metals simplify the environmental permitting process and reduce the regulatory burden on manufacturing sites. This alignment with green chemistry initiatives enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for global pharmaceutical companies with strict sustainability mandates. The ease of scale-up ensures that demand surges can be met without significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this monooxygenase mutant technology in industrial settings. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of adopting this biocatalytic route. Understanding these details is essential for stakeholders evaluating the transition from chemical to enzymatic synthesis for chiral sulfoxide production.
Q: How do the BVMO mutants improve upon wild-type enzymes?
A: The mutants described in patent CN109402074B exhibit significantly enhanced enzyme activity and stability compared to wild-type strains, while drastically reducing the formation of unwanted sulfone byproducts.
Q: What are the primary commercial benefits of this biocatalytic route?
A: This route offers substantial cost savings by reducing enzyme loading requirements and simplifies downstream processing due to higher selectivity, ensuring a more reliable supply chain for chiral intermediates.
Q: Is this technology scalable for industrial production?
A: Yes, the mutants are expressed in robust host cells like E. coli and function under mild conditions, making the commercial scale-up of complex pharmaceutical intermediates feasible and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic technologies described in patent CN109402074B and are well-positioned to leverage them for your specific project needs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of chiral sulfoxide meets the highest international standards. We are committed to providing a reliable chiral sulfoxide supplier partnership that combines technical expertise with commercial reliability.
We invite you to engage with our technical procurement team to discuss how this advanced enzymatic oxidation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this innovative approach for your next commercial campaign.
