Advanced Synthesis of Sulfur-Functionalized Calix[4]arenes for Industrial and Biomedical Applications
Advanced Synthesis of Sulfur-Functionalized Calix[4]arenes for Industrial and Biomedical Applications
The landscape of supramolecular chemistry has been significantly advanced by the development of specialized calixarene derivatives, particularly those functionalized with sulfur groups for enhanced metal ion recognition. Patent CN101337918A discloses a robust and highly efficient preparation method for 25,26,27,28-tetramercaptoethoxy-calix[4]arene, a compound that bridges the gap between theoretical host-guest chemistry and practical industrial application. This specific sulfur-containing derivative offers superior chemical activity compared to traditional oxygen-based analogues, primarily due to the unique electronic properties of the sulfur atom which facilitate stronger interactions with transition metals. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a critical pathway to obtaining high-purity intermediates that are essential for next-generation sensors, biomimetic catalysts, and environmental remediation technologies. The methodology outlined ensures that the production of these complex macrocycles is not only chemically feasible but also economically viable for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of calixarenes at the lower rim has presented significant challenges regarding regioselectivity and reaction severity. Traditional methods often require harsh conditions, such as extreme temperatures or the use of highly reactive reagents that can degrade the delicate macrocyclic cavity or lead to unpredictable polymerization. Furthermore, direct introduction of thiol groups is notoriously difficult due to the susceptibility of thiols to oxidation and the potential for forming disulfide bridges, which complicates purification and lowers overall yield. Many conventional routes rely on expensive or difficult-to-source precursors, creating supply chain bottlenecks that hinder the commercial adoption of these valuable materials. The lack of a standardized, mild protocol has often forced research teams to rely on small-batch, custom syntheses that are ill-suited for the consistent quality required in pharmaceutical or electronic grade applications.
The Novel Approach
The methodology described in CN101337918A overcomes these historical barriers through a clever three-step sequence that prioritizes mild conditions and high conversion rates. By utilizing 25,26,27,28-tetrahydroxyethoxy-calix[4]arene as a stable starting material, the process avoids the instability associated with direct thiolation. The strategy employs a mesylation step to activate the hydroxyl groups, followed by a nucleophilic substitution with a sulfur source, and finally a gentle hydrolysis to reveal the free thiol functionality. This approach ensures that the integrity of the calixarene cup is maintained throughout the synthesis while achieving yields as high as 90% or above. The use of common solvents like dichloromethane and acetonitrile, combined with operation at normal pressure, drastically reduces the engineering controls required for production, making this a superior route for reliable specialty chemical supplier operations aiming for cost reduction in fine chemical intermediates manufacturing.
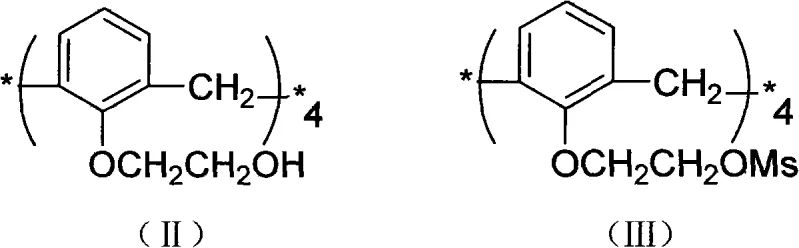
Mechanistic Insights into Stepwise Functionalization and Thio-Substitution
The core of this synthetic breakthrough lies in the precise control of nucleophilic substitution reactions on the calixarene lower rim. In the first critical stage, the hydroxyl groups of the starting material are converted into mesylates (methanesulfonates) using methanesulfonyl chloride in the presence of a base like triethylamine or pyridine at 0°C. This transformation is crucial because the mesylate group is an excellent leaving group, far superior to the hydroxyl group itself, which primes the molecule for the subsequent sulfur introduction. The reaction is conducted at low temperatures to prevent side reactions and ensure that the substitution occurs exclusively at the desired positions without affecting the methylene bridges of the macrocycle. This step effectively turns a relatively inert alcohol into a highly reactive electrophile, setting the stage for the introduction of the sulfur functionality with high fidelity and minimal impurity formation.
Following the activation, the second stage involves the displacement of the mesylate groups by potassium thioacetate in anhydrous acetonitrile at 80°C. This nucleophilic attack introduces the sulfur atoms in a protected thioacetate form, which is a strategic move to prevent premature oxidation of the thiols. The thioacetate group is stable enough to withstand the reaction conditions yet labile enough to be removed in the final step. The final hydrolysis using sodium methoxide in methanol cleaves the acetyl protecting groups under mild basic conditions, releasing the free mercapto (-SH) groups. This mechanistic pathway ensures that the final product, 25,26,27,28-tetramercaptoethoxy-calix[4]arene, possesses four evenly spaced thiol groups capable of acting as soft Lewis bases, providing exceptional binding affinity for soft Lewis acids like heavy metal ions.
![Chemical structure of the intermediate 25,26,27,28-tetrathioacetoxyethoxy-calix[4]arene (IV) showing the protected sulfur groups](/insights/img/tetramercaptoethoxy-calixarene-synthesis-supplier-20260308125651-03.png)
How to Synthesize 25,26,27,28-Tetramercaptoethoxy-Calix[4]arene Efficiently
The synthesis of this high-value calixarene derivative is streamlined into three distinct operational phases that can be easily integrated into standard batch reactor workflows. The process begins with the activation of the starting phenol derivative, followed by the sulfur incorporation, and concludes with the deprotection. Each step utilizes reagents that are readily available on the global chemical market, eliminating the need for custom synthesis of starting materials. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles optimized for maximum yield and purity, are provided in the guide below for technical teams evaluating process feasibility.
- Prepare 25,26,27,28-tetramethanesulfonyloxyethoxy-calix[4]arene (III) by reacting the tetrahydroxy precursor with methanesulfonyl chloride and triethylamine at 0°C.
- Synthesize 25,26,27,28-tetrathioacetoxyethoxy-calix[4]arene (IV) via nucleophilic substitution using potassium thioacetate in anhydrous acetonitrile at 80°C.
- Complete the synthesis by hydrolyzing the thioacetate groups with sodium methoxide in methanol, followed by acidification to isolate the final tetramercapto product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reliance on commercially available raw materials means that supply continuity is robust, reducing the risk of production stoppages due to precursor shortages. The simplicity of the process, which operates at normal pressure and moderate temperatures, translates directly into lower capital expenditure requirements for reactor infrastructure and reduced energy consumption during manufacturing. Furthermore, the high yields reported in the patent examples indicate a material-efficient process that minimizes waste generation, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities. These factors combine to create a supply chain profile that is both resilient and cost-effective.
- Cost Reduction in Manufacturing: The elimination of exotic catalysts and the use of standard organic solvents significantly lowers the bill of materials. By avoiding high-pressure equipment and extreme thermal conditions, the operational costs associated with energy and maintenance are substantially reduced. The high conversion efficiency means less raw material is wasted, optimizing the cost per kilogram of the final active ingredient. This economic efficiency makes the commercial scale-up of complex calixarene derivatives much more attractive for budget-conscious manufacturing strategies.
- Enhanced Supply Chain Reliability: Since all starting materials, including the calixarene precursor, methanesulfonyl chloride, and potassium thioacetate, are commodity chemicals, sourcing is straightforward and competitive. This diversity of supply sources mitigates the risk of single-supplier dependency. The robustness of the reaction conditions also implies that the process is less sensitive to minor variations in utility supply, ensuring consistent batch-to-bquality and reliable delivery schedules for downstream customers requiring high-purity OLED material or pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates manageable waste streams that can be treated using standard effluent protocols, avoiding the need for specialized hazardous waste disposal associated with heavy metal catalysts. The ability to run reactions at atmospheric pressure simplifies the engineering scale-up from pilot plant to full commercial production. This ease of scaling ensures that suppliers can rapidly respond to increases in market demand without lengthy requalification periods, securing the supply of critical functional materials for the electronics and environmental sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of sulfur-functionalized calixarenes. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners.
Q: What are the key advantages of this sulfur-containing calixarene synthesis?
A: The process utilizes commercially available raw materials, operates at normal pressure with mild temperature conditions (0°C to 90°C), and achieves high yields up to 90% or above, significantly simplifying scale-up compared to traditional harsh methods.
Q: What are the primary applications of 25,26,27,28-tetramercaptoethoxy-calix[4]arene?
A: Due to the low electronegativity and high excitability of sulfur atoms, this derivative exhibits strong recognition capabilities for transition metal and heavy metal ions, making it ideal for life sciences, biomimetic catalysis, toxic element removal, and environmental protection sensors.
Q: Is the synthetic route scalable for industrial production?
A: Yes, the patent explicitly states that the raw materials are widely sourced and sufficient, the process is simple, and each reaction step is operated at normal pressure, facilitating convenient scale preparation and reducing operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 25,26,27,28-Tetramercaptoethoxy-Calix[4]arene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced supramolecular hosts like 25,26,27,28-tetramercaptoethoxy-calix[4]arene in fields ranging from heavy metal remediation to biomimetic sensing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market reality is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of this complex intermediate meets the exacting standards required for high-tech applications. We understand that consistency is key, and our process engineering team is dedicated to maintaining the high yields and purity profiles demonstrated in the patented literature.
We invite you to collaborate with us to leverage this efficient synthetic technology for your specific application needs. Whether you require custom kilo-lab quantities for R&D or multi-ton supply for industrial deployment, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can accelerate your product development timeline while optimizing your overall manufacturing costs.
