Advanced Synthesis of Alpha-Chlorovinyl Cyclopropane for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly those serving the antiretroviral market. Patent CN101624329B, published in 2012, introduces a significant technological advancement in the production of Alpha-chlorovinyl cyclopropane, a pivotal precursor for Cyclopropyne and ultimately Efavirenz. This innovation addresses long-standing safety and efficiency challenges by replacing hazardous chlorinating agents with bis(trichloromethyl) carbonate, commonly known as Triphosgene. For R&D directors and supply chain managers, this shift represents a move towards greener chemistry without compromising the structural integrity of the sensitive cyclopropane ring. The method utilizes an organic amine catalyst system that operates under mild thermal conditions, ensuring high selectivity and minimizing the formation of toxic by-products. By adopting this patented approach, manufacturers can achieve a reliable pharmaceutical intermediate supplier status while adhering to stricter environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Alpha-chlorovinyl cyclopropane relied heavily on phosphorus pentachloride (PCl5) or gaseous phosgene, both of which present severe operational hazards and environmental liabilities. As illustrated in the conventional pathway, the use of PCl5 requires rigorous temperature control during the hydrolysis step to prevent the generated HCl from attacking the strained three-membered ring. 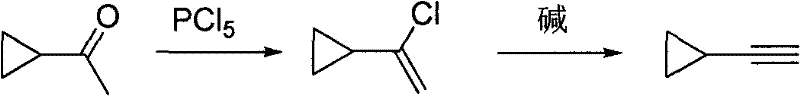 Failure to maintain low temperatures often leads to ring-opening side reactions, producing unwanted by-products like 2,5-dichloro-2-pentyne, which drastically reduces the overall yield and complicates purification. Furthermore, the hydrolysis of excess PCl5 generates substantial volumes of acidic, phosphorus-containing wastewater that is difficult and costly to treat. The corrosive nature of these reagents also accelerates equipment degradation, leading to increased maintenance downtime and higher capital expenditure for specialized corrosion-resistant reactors. These factors collectively make the traditional PCl5 route unsuitable for modern, large-scale sustainable manufacturing.
Failure to maintain low temperatures often leads to ring-opening side reactions, producing unwanted by-products like 2,5-dichloro-2-pentyne, which drastically reduces the overall yield and complicates purification. Furthermore, the hydrolysis of excess PCl5 generates substantial volumes of acidic, phosphorus-containing wastewater that is difficult and costly to treat. The corrosive nature of these reagents also accelerates equipment degradation, leading to increased maintenance downtime and higher capital expenditure for specialized corrosion-resistant reactors. These factors collectively make the traditional PCl5 route unsuitable for modern, large-scale sustainable manufacturing.
The Novel Approach
In contrast, the novel methodology disclosed in the patent employs bis(trichloromethyl) carbonate as a solid, safer phosgene equivalent, fundamentally altering the risk profile of the synthesis. This reagent reacts with cyclopropyl methyl ketone in the presence of an organic amine catalyst, such as triethylamine or quinoline, within a standard organic solvent matrix. 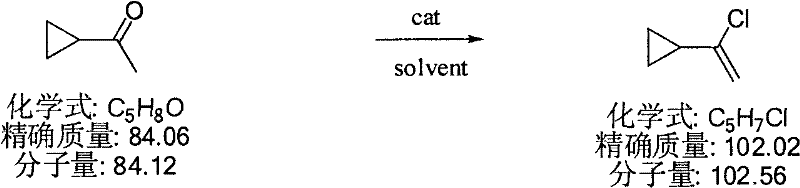 The reaction proceeds smoothly at temperatures ranging from 0 to 150°C, eliminating the need for cryogenic conditions required by older methods. The use of Triphosgene ensures that the chlorination occurs efficiently without generating free mineral acids that could compromise the cyclopropane ring stability. This approach not only simplifies the work-up procedure—often requiring only distillation—but also significantly reduces the volume of hazardous waste. For procurement teams, this translates to cost reduction in pharmaceutical intermediate manufacturing by lowering waste disposal fees and extending the lifespan of production assets through reduced corrosion.
The reaction proceeds smoothly at temperatures ranging from 0 to 150°C, eliminating the need for cryogenic conditions required by older methods. The use of Triphosgene ensures that the chlorination occurs efficiently without generating free mineral acids that could compromise the cyclopropane ring stability. This approach not only simplifies the work-up procedure—often requiring only distillation—but also significantly reduces the volume of hazardous waste. For procurement teams, this translates to cost reduction in pharmaceutical intermediate manufacturing by lowering waste disposal fees and extending the lifespan of production assets through reduced corrosion.
Mechanistic Insights into Organic Amine-Catalyzed Chlorination
The core of this synthetic breakthrough lies in the synergistic interaction between the organic amine catalyst and the Triphosgene reagent. Mechanistically, the amine acts as a nucleophilic activator, facilitating the generation of reactive chlorocarbonyl species from the solid Triphosgene under mild thermal conditions. This in-situ generation allows for a controlled release of the chlorinating agent, preventing localized hotspots of high acidity that typically trigger ring-opening events in cyclopropyl systems. The catalyst stabilizes the transition state, promoting the specific substitution at the alpha-position of the ketone while preserving the integrity of the adjacent cyclopropane ring. This precise control over the reaction trajectory is crucial for maintaining high optical and chemical purity, which is a mandatory requirement for API intermediates destined for antiretroviral therapy.
Furthermore, the choice of solvent plays a pivotal role in modulating the reaction kinetics and solubility of the intermediates. The patent highlights a broad scope of compatible solvents, including halogenated alkanes, esters, and aromatic hydrocarbons, providing flexibility for process optimization. By selecting solvents like ethyl acetate or toluene, manufacturers can further align the process with green chemistry principles, moving away from chlorinated solvents where possible. The mechanism ensures that side reactions are kinetically suppressed, resulting in a cleaner crude product profile. This reduction in impurity load simplifies downstream purification, thereby enhancing the overall throughput and reducing the consumption of energy-intensive distillation steps. Such mechanistic elegance is what defines a high-purity pharmaceutical intermediate suitable for global regulatory submission.
How to Synthesize Alpha-Chlorovinyl Cyclopropane Efficiently
To implement this superior synthetic route, operators must adhere to specific stoichiometric ratios and thermal profiles outlined in the patent data. The process begins with the precise charging of cyclopropyl methyl ketone and the selected organic amine catalyst into a reactor, followed by the controlled addition of the Triphosgene solution. Detailed standardized synthesis steps see the guide below.
- Charge cyclopropyl methyl ketone and an organic amine catalyst (such as triethylamine or quinoline) into a reaction vessel equipped with mechanical stirring.
- Heat the mixture to a temperature range of 20-100°C and slowly add a solution of bis(trichloromethyl) carbonate in an organic solvent.
- Maintain the reaction temperature for 4 to 12 hours to ensure complete conversion, followed by vacuum distillation to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition to this Triphosgene-based methodology offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials; Triphosgene is a stable solid that is easier to transport and store compared to gaseous phosgene or moisture-sensitive PCl5. This stability reduces the risk of supply disruptions caused by hazardous material transport restrictions, ensuring a more continuous flow of production. Additionally, the elimination of phosphorus waste streams removes a significant bottleneck in wastewater treatment capacity, allowing facilities to operate at higher utilization rates without exceeding environmental discharge limits. These operational efficiencies collectively contribute to substantial cost savings and enhanced supply chain reliability.
- Cost Reduction in Manufacturing: The adoption of this catalytic system eliminates the need for expensive corrosion-resistant equipment mandated by strong mineral acids, leading to lower capital investment and maintenance costs. By avoiding the complex quenching and neutralization steps associated with PCl5, the process consumes fewer auxiliary chemicals and reduces labor hours per batch. The high selectivity of the reaction minimizes the loss of valuable starting materials, directly improving the material cost efficiency of the final product. Consequently, the total cost of ownership for producing this key intermediate is significantly optimized.
- Enhanced Supply Chain Reliability: Utilizing a solid chlorinating agent like Triphosgene mitigates the logistical challenges associated with hazardous gas cylinders or highly reactive liquid reagents. This ease of handling allows for larger batch sizes and more flexible scheduling, as the process is less susceptible to delays caused by safety inspections or specialized transport requirements. The robustness of the reaction conditions also means that production can be scaled up with greater confidence, reducing the lead time for high-purity pharmaceutical intermediates. Suppliers can thus guarantee more consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of organic solvents that can often be recovered and recycled, aligning with modern sustainability goals. The absence of phosphorus effluents simplifies compliance with increasingly stringent environmental regulations, reducing the administrative burden and potential fines associated with waste management. This eco-friendly profile enhances the marketability of the product to multinational corporations with strict vendor sustainability codes. Furthermore, the mild reaction conditions facilitate safer scale-up from pilot plants to multi-ton commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation.
Q: Why is Triphosgene preferred over PCl5 for this synthesis?
A: Triphosgene (bis(trichloromethyl) carbonate) is a solid, safer alternative to gaseous phosgene and avoids the harsh acidic conditions of PCl5. Unlike PCl5, which generates significant phosphorus-containing wastewater and risks opening the sensitive cyclopropane ring during hydrolysis, Triphosgene offers milder reaction conditions and easier waste management.
Q: What is the primary application of Alpha-Chlorovinyl Cyclopropane?
A: This compound serves as a critical precursor for Cyclopropyne, which is a key building block in the synthesis of Efavirenz, a widely used non-nucleoside reverse transcriptase inhibitor for treating HIV/AIDS.
Q: How does the new catalytic method improve yield stability?
A: By utilizing organic amine catalysts like 1,3-dimethyl-2-imidazolidinone or triethylamine at controlled temperatures (0-150°C), the process minimizes side reactions such as ring-opening. This results in consistently high yields (reported up to 94% in examples) compared to the variable and lower yields of traditional phosphorus-based methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Chlorovinyl Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that Alpha-chlorovinyl cyclopropane plays in the global supply of antiretroviral medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major pharmaceutical contracts. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the absence of ring-opened impurities. Our facility is equipped to handle the specific safety requirements of Triphosgene chemistry, guaranteeing a safe and compliant manufacturing environment for our partners.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential efficiencies gained by switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to secure a stable, high-quality supply of this essential pharmaceutical building block.
