Advanced Synthesis of Emtricitabine Intermediates: Enhancing Safety and Scalability for Global API Production
Advanced Synthesis of Emtricitabine Intermediates: Enhancing Safety and Scalability for Global API Production
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational safety, particularly for critical antiviral agents like Emtricitabine. Patent CN101391997B introduces a significant methodological advancement in the synthesis of the key Emtricitabine intermediate, specifically (2R, 5S)-5-(5'-fluoro-cytosine-1-yl)-1, 3-oxathiolane-2-carboxylic acid-L-menthyl ester. This technical disclosure addresses long-standing challenges associated with traditional chlorination reagents by substituting hazardous thionyl chloride with the more manageable bis(trichloromethyl) carbonate, also known as Triphosgene. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, this shift represents a pivotal opportunity to optimize manufacturing protocols. The patent outlines a pathway that not only maintains high stereochemical integrity but also drastically improves the environmental profile of the production process, aligning with modern green chemistry standards required by top-tier global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of nucleoside analog intermediates has heavily relied on thionyl chloride for the activation of hydroxyl groups into chloro-leaving groups. While chemically effective, this reagent presents severe logistical and safety hurdles for large-scale operations. Thionyl chloride is a volatile, corrosive liquid that releases substantial quantities of sulfur dioxide gas upon reaction, necessitating expensive and complex off-gas treatment systems to meet environmental regulations. Furthermore, the handling of such toxic materials increases the risk profile for operators and requires specialized corrosion-resistant equipment, driving up capital expenditure. In the context of synthesizing sensitive chiral molecules, the harsh conditions often associated with thionyl chloride can sometimes lead to racemization or the formation of difficult-to-remove impurities, complicating downstream purification and potentially impacting the overall yield of the high-purity pharmaceutical intermediate.
The Novel Approach
The innovative strategy detailed in the patent leverages bis(trichloromethyl) carbonate as a superior chlorinating agent to overcome these deficiencies. As a crystalline solid at ambient temperatures, this reagent offers inherent advantages in terms of storage stability and precise metering, which are critical for reproducible batch processing. Unlike its liquid counterparts, the decomposition of bis(trichloromethyl) carbonate primarily yields carbon dioxide, a benign gas that does not require aggressive scrubbing, thereby simplifying the engineering controls needed for the reactor setup. 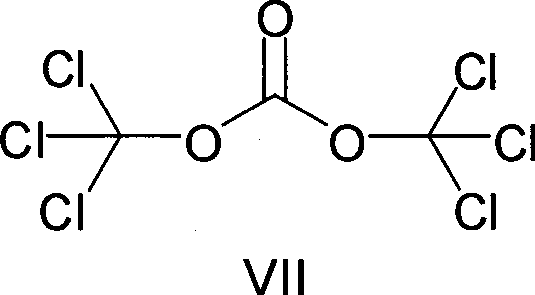 This substitution effectively transforms a hazardous unit operation into a safer, more controllable process step. By mitigating the risks associated with toxic gas evolution and equipment corrosion, this novel approach facilitates a smoother path toward commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality while reducing the operational burden on manufacturing facilities.
This substitution effectively transforms a hazardous unit operation into a safer, more controllable process step. By mitigating the risks associated with toxic gas evolution and equipment corrosion, this novel approach facilitates a smoother path toward commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality while reducing the operational burden on manufacturing facilities.
Mechanistic Insights into Triphosgene-Mediated Chlorination and Glycosylation
The core of this synthetic breakthrough lies in the efficient conversion of the chiral hydroxyl precursor into the corresponding chloro-intermediate without compromising stereochemistry. The reaction utilizes a catalytic amount of N,N-dimethylformamide (DMF) to activate the bis(trichloromethyl) carbonate, generating a highly reactive Vilsmeier-Haack type species in situ. This activated complex selectively targets the secondary hydroxyl group on the oxathiolane ring of the L-menthyl ester substrate, facilitating a clean substitution to form the chloro-derivative. The mild reaction conditions, typically maintained between 0°C and 50°C, are crucial for preserving the chiral centers at the 2 and 5 positions of the oxathiolane ring, preventing epimerization which would otherwise lead to diastereomeric impurities that are notoriously difficult to separate. This mechanistic precision ensures that the resulting chloro-compound retains the high optical purity required for the subsequent biological activity of the final antiviral drug.
Following the chlorination, the process proceeds to a condensation step with N,O-bis(trimethylsilyl)-5-fluorocytosine. The silylation of the cytosine base enhances its nucleophilicity and solubility in organic media, promoting an efficient SN2-type displacement of the chloride leaving group. The use of an acid binding agent, such as triethylamine, is essential to neutralize the hydrochloric acid generated during the coupling, preventing the degradation of the acid-sensitive oxathiolane ring. 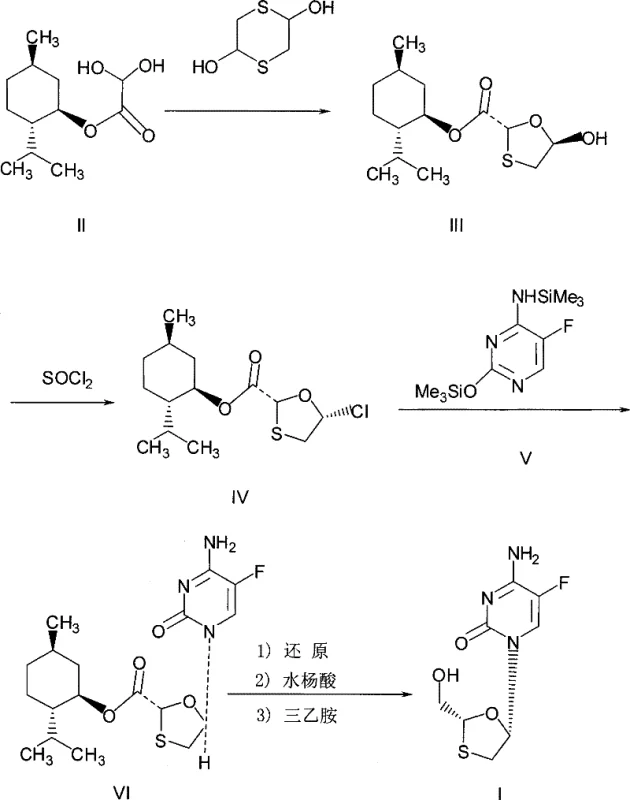 The final hydrolysis step removes the chiral auxiliary (L-menthol) and silyl protecting groups under controlled alkaline conditions. This sequence is designed to minimize side reactions, ensuring that the final product exhibits the stringent purity specifications demanded for API intermediates, with experimental data indicating HPLC purity levels consistently above 99% and chiral purity exceeding 99%.
The final hydrolysis step removes the chiral auxiliary (L-menthol) and silyl protecting groups under controlled alkaline conditions. This sequence is designed to minimize side reactions, ensuring that the final product exhibits the stringent purity specifications demanded for API intermediates, with experimental data indicating HPLC purity levels consistently above 99% and chiral purity exceeding 99%.
How to Synthesize (2R,5S)-5-(5'-fluoro-cytosine-1-yl)-1,3-oxathiolane-2-carboxylic acid-L-menthyl ester Efficiently
The patented methodology offers a streamlined protocol that can be adapted for both batch and continuous processing environments. The procedure begins with the dissolution of the chiral hydroxy-ester in a suitable organic solvent such as methylene chloride or toluene, followed by the controlled addition of the chlorinating agent. The reaction temperature is carefully managed to optimize the formation of the chloro-intermediate while suppressing degradation. Subsequently, the silylated base is introduced to effect the glycosidic bond formation. The versatility of this route allows for a 'one-pot' variation where reagents are added sequentially without isolating the unstable chloro-intermediate, further enhancing process efficiency.
- Perform chlorination of (2R,5S)-5-hydroxy-1,3-oxathiolane-2-carboxylic acid-L-menthyl ester using bis(trichloromethyl) carbonate and DMF catalyst in an organic solvent at 0-50°C.
- Condense the resulting chloro-intermediate with N,O-bis(trimethylsilyl)-5-fluorocytosine in the presence of an acid binding agent like triethylamine.
- Execute hydrolysis and workup by pouring the reaction mixture into water and hexane containing triethylamine, followed by filtration and drying to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this triphosgene-based synthesis translates into tangible operational benefits beyond mere chemical yield. The transition from a hazardous liquid reagent to a stable solid significantly de-risks the supply chain by simplifying logistics; solid reagents are easier to ship, store, and handle without the need for specialized containment infrastructure required for corrosive liquids. This inherent safety profile reduces the regulatory burden and insurance costs associated with manufacturing hazardous intermediates. Moreover, the elimination of sulfur dioxide emissions removes the need for complex and maintenance-intensive scrubbing systems, leading to substantial cost savings in facility operations and waste management. These factors collectively contribute to a more resilient and cost-effective manufacturing model for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of thionyl chloride with bis(trichloromethyl) carbonate eliminates the necessity for expensive corrosion-resistant reactors and extensive gas scrubbing equipment, directly lowering capital and maintenance expenditures. The solid nature of the reagent allows for precise automated dosing, minimizing material waste and improving batch-to-batch consistency, which is vital for reducing the cost of goods sold in competitive API markets. Additionally, the potential for a one-pot synthesis reduces the number of isolation and purification steps, saving on solvent consumption and labor hours, thereby driving down the overall production cost without sacrificing yield or quality.
- Enhanced Supply Chain Reliability: Utilizing a stable solid reagent mitigates the risks associated with the transportation and storage of volatile liquids, ensuring a more reliable flow of raw materials to the production site. The simplified process workflow reduces the likelihood of unplanned downtime caused by equipment failure due to corrosion or safety incidents, guaranteeing consistent delivery schedules for clients. This reliability is paramount for pharmaceutical companies managing tight production timelines for antiretroviral therapies, where supply continuity is critical for patient access and regulatory compliance.
- Scalability and Environmental Compliance: The process is inherently scalable due to the ease of handling solid reagents and the absence of toxic gas evolution, making it ideal for transitioning from pilot plant to multi-ton commercial production. The generation of carbon dioxide as a byproduct instead of sulfur dioxide aligns with increasingly strict environmental regulations, facilitating easier permitting and reducing the ecological footprint of the manufacturing site. This environmental compatibility not only future-proofs the production facility against tightening regulations but also enhances the corporate sustainability profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is bis(trichloromethyl) carbonate preferred over thionyl chloride for this synthesis?
A: Bis(trichloromethyl) carbonate (Triphosgene) is a solid at room temperature, making it significantly safer to transport, store, and meter compared to the toxic and corrosive liquid thionyl chloride. Furthermore, its decomposition byproducts are primarily carbon dioxide, eliminating the generation of sulfur dioxide waste gas which requires complex scrubbing systems.
Q: What are the typical purity and yield metrics for this patented process?
A: According to the experimental data in patent CN101391997B, the process consistently achieves HPLC purity levels exceeding 99% and chiral purity above 99%. The isolated yields for the key intermediate typically range between 66% and 69% across various embodiments, demonstrating robust reproducibility suitable for industrial application.
Q: Can this synthesis be adapted for large-scale commercial production?
A: Yes, the patent explicitly highlights the suitability for industrial production. The ability to use a 'one-pot' synthetic approach simplifies the operational workflow by reducing unit operations. Additionally, the use of a solid chlorinating agent facilitates precise automated dosing, which is critical for maintaining quality control during commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Emtricitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the triphosgene-mediated synthesis are seamlessly translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify chiral and chemical purity at every stage of production. Our dedication to quality assurance ensures that every batch of intermediate meets the exacting standards required for downstream API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a successful and sustainable partnership in the production of life-saving antiviral medications.
