Advanced Xylene-Based Synthesis of L-Dihydrophenyl Glycine Sodium Salt for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibiotic intermediates, particularly those serving the cephalosporin class. Patent CN101550089B introduces a transformative preparation method for L-dihydrophenyl glycine sodium salt, a pivotal side-chain precursor for Cephradine. This innovation addresses long-standing inefficiencies in solvent usage and product purity by replacing traditional methyl isobutyl ketone (MIBK) systems with a refined xylene-based protocol. By leveraging high-purity sodium methoxide prepared via the metal method, this process achieves a remarkable conversion rate calculated at 175% relative to dihydro phenylglycine, while maintaining product absorbency below 0.1 degree. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for semi-synthetic antibiotics, ensuring both cost efficiency and stringent quality control required for global regulatory compliance.
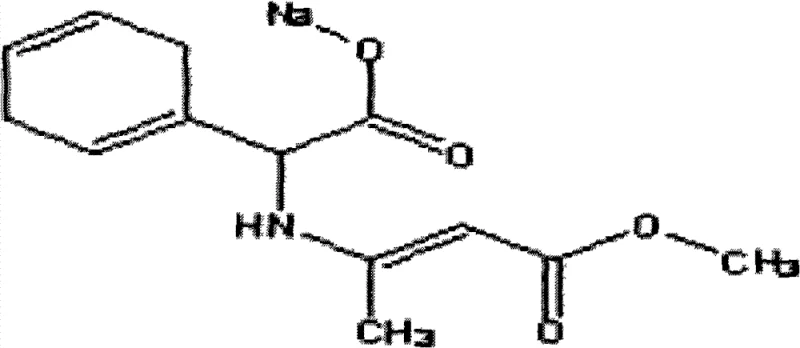
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of L-dihydrophenyl glycine sodium salt has relied heavily on MIBK as the primary reaction solvent, a practice that introduces substantial economic and technical burdens. The conventional process typically involves reacting dihydro phenylglycine with sodium metal in liquid ammonia, followed by condensation with methyl acetoacetate in MIBK. However, MIBK is a relatively expensive solvent, with market prices fluctuating significantly, and it requires a high unit consumption of approximately 0.6-0.7kg per kg of product salt. Furthermore, the removal of MIBK is notoriously difficult due to its boiling point characteristics, often leading to residual ketone contamination in the final intermediate. These ketone residues are detrimental to the subsequent synthesis of Cephradine, where they can interfere with coupling reactions and reduce the overall yield of the final antibiotic drug. Additionally, traditional methods often utilize sodium methoxide prepared via alkaline processes, which may contain higher levels of free alkali and impurities, adversely affecting the color and purity of the final sodium salt product.
The Novel Approach
The methodology disclosed in patent CN101550089B fundamentally reengineers the solvent system and reagent quality to overcome these legacy deficits. By substituting MIBK with dimethylbenzene (xylene), the process drastically reduces solvent unit consumption to a range of 0.35-0.45kg/kg, representing a massive reduction in material costs and waste generation. Crucially, the use of xylene eliminates the risk of ketone residue, thereby safeguarding the quality of downstream Cephradine production. A key differentiator in this novel approach is the specific utilization of sodium methoxide methanol solution prepared via the metal method, rather than the standard alkaline process. This ensures a higher purity reagent with minimal free alkali content, directly contributing to the exceptional optical properties of the product, evidenced by an absorbency of less than 0.1 degree. The integration of these changes results in a conversion yield reaching approximately 175% based on dihydro phenylglycine, offering a compelling value proposition for large-scale manufacturers seeking to enhance throughput without compromising specification standards.
Mechanistic Insights into Xylene-Mediated Condensation
The core of this improved synthesis lies in the precise control of the condensation reaction between dihydro phenylglycine and methyl acetoacetate within a non-polar aromatic solvent environment. In the xylene medium, the solubility profile of the reactants and the intermediate sodium salt is optimized to favor precipitation upon cooling, which drives the equilibrium forward and simplifies isolation. The reaction mechanism involves the deprotonation of the amino acid derivative by the high-purity sodium methoxide, generating a nucleophilic species that attacks the carbonyl carbon of the methyl acetoacetate. The use of the metal-method sodium methoxide is critical here; its superior purity prevents side reactions often catalyzed by trace hydroxides or carbonates found in alkali-method reagents. This purity ensures that the enamine formation proceeds cleanly, minimizing the formation of colored polymeric by-products that typically degrade the visual quality of the intermediate. The reaction is conducted under controlled thermal conditions, initially at 10-20°C to manage the exotherm during reagent addition, followed by a reflux period to ensure complete conversion before the solvent exchange and crystallization phases begin.
Impurity control is rigorously managed through temperature regulation during the distillation and isolation stages. The L-dihydrophenyl glycine sodium salt is identified as a heat-sensitive substance, prone to thermal decomposition if exposed to excessive temperatures. Specifically, if the temperature during underpressure distillation exceeds 55°C, the compound risks decomposing back into rear phenylglycine sodium salts or other degradation products, which would compromise the assay and purity profile. Therefore, the process employs a strategic vacuum distillation protocol where methanol is removed first, followed by the careful addition of xylene to induce crystallization at temperatures strictly maintained below 55°C. This thermal management, combined with the final washing step using isopropanol, effectively removes residual organic impurities and unreacted starting materials. The result is a product with a purity exceeding 99%, suitable for direct use in the sensitive coupling reactions required for cephalosporin antibiotic synthesis, ensuring that the impurity profile remains well within the tight limits demanded by pharmacopeial standards.
How to Synthesize L-Dihydrophenyl Glycine Sodium Salt Efficiently
Implementing this optimized route requires strict adherence to the sequential addition of reagents and precise thermal management to maximize the 175% theoretical yield potential. The process begins with the preparation of the reaction mixture using xylene and high-concentration sodium methoxide solution, followed by the controlled addition of methyl acetoacetate. Operators must monitor the clarity of the reaction solution closely, as the transition from turbidity to clarity indicates the formation of the soluble intermediate prior to crystallization. The subsequent distillation steps are critical for solvent exchange, requiring a switch from atmospheric to underpressure distillation to protect the heat-sensitive product. Detailed standard operating procedures regarding the specific flow rates, vacuum levels, and cooling ramps are essential for reproducibility at the commercial scale. For a comprehensive breakdown of the exact operational parameters and safety protocols, please refer to the standardized synthesis guide below.
- Prepare high-purity sodium methoxide methanol solution using the metal method and mix with dihydro phenylglycine in xylene solvent at 10-20°C.
- Add methyl acetoacetate slowly while heating, maintaining reaction clarity, followed by reflux to ensure complete condensation.
- Perform vacuum distillation to remove methanol, induce crystallization with additional xylene, and filter the product at temperatures below 55°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this xylene-based methodology offers profound strategic advantages beyond simple yield improvements. The shift away from MIBK not only lowers the direct cost of raw materials but also simplifies the waste treatment infrastructure, as xylene is easier to recover and recycle in standard solvent recovery units. The elimination of ketone residues removes a critical bottleneck in the downstream production of Cephradine, reducing the risk of batch failures in the final API manufacturing stage. This reliability translates into a more stable supply chain, where intermediates consistently meet the rigorous specifications required by major pharmaceutical partners. Furthermore, the reduced solvent consumption per kilogram of product significantly lowers the logistical burden of solvent transport and storage, enhancing the overall sustainability profile of the manufacturing operation. These factors collectively contribute to a more resilient and cost-effective supply chain for cephalosporin intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive MIBK with xylene, combined with a reduction in solvent unit consumption from 0.7kg/kg to roughly 0.4kg/kg, drives substantial savings in variable production costs. The use of high-yield chemistry means that less starting material is wasted, further improving the cost-per-kilogram metric. Additionally, the avoidance of complex purification steps to remove ketone residues reduces energy consumption and labor hours associated with reprocessing. These cumulative efficiencies allow for a more competitive pricing structure without sacrificing margin, making the intermediate more attractive in a price-sensitive generic drug market.
- Enhanced Supply Chain Reliability: By producing an intermediate with negligible ketone content, manufacturers eliminate a major source of variability for their customers producing Cephradine. This consistency reduces the likelihood of supply disruptions caused by out-of-specification batches. The robustness of the xylene solvent system also allows for greater flexibility in sourcing, as xylene is a widely available commodity chemical with a stable global supply network. This ensures that production schedules can be maintained even during periods of raw material volatility, providing a secure and dependable source of critical antibiotic precursors for long-term contracts.
- Scalability and Environmental Compliance: The process is designed for seamless scale-up, utilizing standard reactor configurations and distillation equipment common in fine chemical facilities. The lower solvent load reduces the volume of hazardous waste generated per ton of product, simplifying compliance with increasingly stringent environmental regulations. The ability to operate at moderate temperatures and pressures enhances process safety, reducing the risk of thermal runaways. This combination of scalability and environmental stewardship makes the technology ideal for expanding production capacity to meet growing global demand for semi-synthetic antibiotics while maintaining a green manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this method for commercial production. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for process engineers.
Q: Why is xylene preferred over MIBK for this synthesis?
A: Xylene significantly reduces solvent unit consumption to 0.35-0.45kg/kg compared to MIBK, lowers costs, and eliminates residual ketone impurities that negatively impact downstream Cephradine production.
Q: How does the metal method for sodium methoxide improve quality?
A: Sodium methoxide prepared via the metal method has higher purity and lower free alkali content compared to the alkali process, resulting in product absorbency of less than 0.1 degree and superior color.
Q: What are the critical temperature controls during distillation?
A: Temperature must not exceed 55°C during underpressure distillation because the sodium salt is heat-sensitive and can decompose into rear phenylglycine salts if overheated.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Dihydrophenyl Glycine Sodium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering L-dihydrophenyl glycine sodium salt with stringent purity specifications and rigorous QC labs testing every batch for absorbency, assay, and residual solvents. Our facility is equipped to handle the specific thermal and solvent requirements of this xylene-based process, guaranteeing a product that consistently meets the <0.1 degree absorbency standard required for premium Cephradine synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your production efficiency and reduce overall costs. Let us be your trusted partner in securing a reliable and high-quality supply of essential cephalosporin intermediates.
