Advanced Synthesis of 2,5-Diphenylamino Terephthalic Acid for High-Performance Pigment Production
Advanced Synthesis of 2,5-Diphenylamino Terephthalic Acid for High-Performance Pigment Production
The global demand for high-performance organic pigments, particularly quinacridone derivatives, necessitates robust and efficient supply chains for their critical precursors. Patent CN101823978A introduces a transformative methodology for preparing 2,5-diphenyl amino terephthalic acid (DATA), a pivotal intermediate in the synthesis of these premium colorants. This technical disclosure outlines a departure from conventional solvent-intensive protocols, leveraging a unique ethylene glycol and water dispersion system to enhance reaction kinetics and product isolation. The structural integrity of the final product, characterized by its red-purple pressed powder form and specific molecular weight of 348, is crucial for downstream pigment performance. 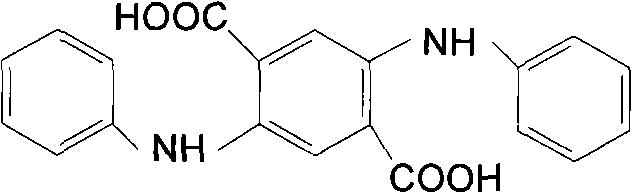 As a reliable dye intermediate supplier, understanding these mechanistic shifts is essential for securing a competitive advantage in the fine chemical market, ensuring that the transition from laboratory innovation to commercial reality is seamless and economically viable for all stakeholders involved.
As a reliable dye intermediate supplier, understanding these mechanistic shifts is essential for securing a competitive advantage in the fine chemical market, ensuring that the transition from laboratory innovation to commercial reality is seamless and economically viable for all stakeholders involved.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-diphenylamino terephthalic acid intermediates has been plagued by significant thermodynamic and kinetic barriers inherent to traditional solvent systems. Conventional processes typically rely on lower-boiling organic solvents such as methanol or ethanol, where the solubility of the key starting material, dimethyl succinosuccinate (DMSS), is severely restricted. This limitation forces the reaction into a heterogeneous solid-liquid or even solid-solid regime, drastically reducing collision frequency between reactant molecules and leading to incomplete conversions. Furthermore, the generation of water as a by-product during condensation complicates the Recovery of Organic Solvent, often requiring energy-intensive distillation columns to separate azeotropes. The necessity for pressurized operations to elevate reaction temperatures in these volatile solvents introduces substantial security hidden dangers, while the resulting low product selectivity and transformation efficiency create a bottleneck for cost reduction in pigment manufacturing. These inefficiencies culminate in a process that generates excessive wastewater and requires complex purification steps to achieve acceptable purity levels.
The Novel Approach
The innovative protocol detailed in the patent data fundamentally re-engineers the reaction environment by substituting volatile alcohols with a high-boiling ethylene glycol and water dispersion medium. This strategic shift allows the reaction system to operate at significantly elevated temperatures, specifically between 110°C and 140°C, without the need for excessive pressure, thereby enhancing the solubility of DMSS in aniline to form a transient homogeneous phase. 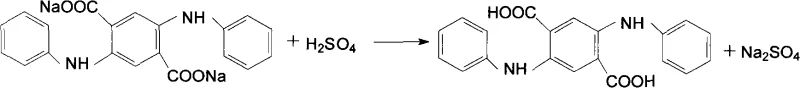 As the condensation proceeds, the product precipitates as a fine yellow solid within the suspension, facilitating easy separation and driving the equilibrium forward. This approach not only mitigates the safety risks associated with flammable solvents but also creates a high-concentration solution of the intermediate salt during the subsequent hydrolysis and oxidation stages. The result is a process that yields larger, more uniform solid crystals upon acidification, directly addressing the historical痛点 of low production efficiency and poor product purity while offering a pathway for substantial cost savings through simplified solvent recycling and reduced waste treatment burdens.
As the condensation proceeds, the product precipitates as a fine yellow solid within the suspension, facilitating easy separation and driving the equilibrium forward. This approach not only mitigates the safety risks associated with flammable solvents but also creates a high-concentration solution of the intermediate salt during the subsequent hydrolysis and oxidation stages. The result is a process that yields larger, more uniform solid crystals upon acidification, directly addressing the historical痛点 of low production efficiency and poor product purity while offering a pathway for substantial cost savings through simplified solvent recycling and reduced waste treatment burdens.
Mechanistic Insights into Condensation and Oxidative Hydrolysis
The core of this synthetic strategy lies in the precise orchestration of acid-catalyzed condensation followed by a tandem hydrolysis-oxidation sequence. In the initial phase, DMSS and aniline undergo a condensation reaction facilitated by mineral acids such as sulfuric or hydrochloric acid, forming 2,5-di-anilino-3,6-dihydro dimethyl terephthalate. The use of ethylene glycol as a dispersion agent is critical here; it acts as a thermal buffer and a solubility enhancer, allowing the reactants to interact more intimately than in traditional media. Following this, the introduction of a mineral alkali, such as sodium hydroxide or potassium carbonate, alongside a potent oxidant like m-nitrobenzene sodium sulfonate or nitrobenzene, triggers the aromatization of the cyclohexane ring. This oxidative hydrolysis converts the dihydro intermediate into the fully aromatic terephthalate salt. The stoichiometry is tightly controlled, with oxidant ratios carefully maintained between 1.05:1 and 2.5:1 relative to DMSS, ensuring complete conversion while minimizing over-oxidation by-products that could compromise the color quality of the final pigment.
Impurity control is achieved through a sophisticated adsorption and decolorization step prior to the final acidification. The patent specifies the use of activated carbon, diatomite, or molecular sieves to scavenge trace organic impurities and colored by-products from the high-strength terephthalate salt solution. This step is paramount for R&D directors focused on purity specifications, as removing these contaminants in the liquid phase prevents them from co-crystallizing with the product. Subsequent neutralization with acid at a controlled pH range of 2 to 6 induces the precipitation of the target 2,5-diphenylamino terephthalic acid. The high concentration of the precursor salt solution, a direct benefit of the ethylene glycol system, ensures that the nucleation and growth of crystals occur under optimal conditions, yielding a product with superior physical properties and consistent quality suitable for high-grade applications in engineering plastics and automotive coatings.
How to Synthesize 2,5-Diphenylamino Terephthalic Acid Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature profiling to maximize yield and safety. The process begins with the batch mixing of DMSS and aniline in the ethylene glycol-water dispersion, followed by a heating phase to ensure complete dissolution before catalyst addition. The subsequent condensation and oxidative hydrolysis steps must be monitored closely to maintain the specified pH and temperature windows, ensuring the reaction proceeds to completion without degradation. Detailed standard operating procedures regarding stirring speeds, cooling rates for crystallization, and filtration parameters are essential for reproducibility.
- Mix DMSS and aniline in an ethylene glycol and water dispersion system, then heat to dissolve and initiate acid-catalyzed condensation.
- Perform simultaneous hydrolysis and oxidation by adding mineral alkali and an oxidant at elevated temperatures to form the terephthalate salt.
- Purify the solution via adsorption, separate solids, and acidify the mother liquor to crystallize the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible benefits that extend beyond mere chemical yield. By eliminating the reliance on large volumes of volatile organic solvents like methanol, the process inherently reduces the complexity and cost of solvent recovery infrastructure. The ability to recycle the ethylene glycol and water dispersion medium significantly lowers raw material consumption and minimizes the volume of hazardous waste requiring treatment, aligning with increasingly stringent environmental regulations. This reduction in waste handling and solvent loss translates directly into a more predictable and stable cost structure for high-purity dye intermediates, shielding the supply chain from the volatility of petrochemical-derived solvent markets.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous volatile solvents removes the need for complex explosion-proof recovery systems, drastically lowering capital expenditure and operational overhead. Furthermore, the high conversion efficiency and the ability to recycle unreacted starting materials from the solid-liquid separation step minimize raw material waste, leading to substantial cost savings in the overall production budget without compromising on output quality.
- Enhanced Supply Chain Reliability: The use of readily available and stable raw materials such as aniline, DMSS, and ethylene glycol ensures a robust supply base that is less susceptible to market fluctuations compared to specialized solvents. The simplified process flow, which avoids high-pressure operations and reduces reaction times through improved solubility, enhances production throughput, allowing for faster turnaround times and more reliable delivery schedules for critical pigment intermediates.
- Scalability and Environmental Compliance: The process design facilitates easy scale-up from pilot batches to multi-ton commercial production using standard stirred tank or tubular reactors, avoiding the engineering challenges associated with high-pressure autoclaves. Additionally, the significant reduction in wastewater generation and the ease of treating the aqueous glycol effluent ensure compliance with environmental standards, reducing the risk of regulatory shutdowns and enhancing the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the practical advantages of this method for potential partners and technical teams evaluating the process for adoption.
Q: How does the new ethylene glycol dispersion method improve upon traditional methanol-based synthesis?
A: Traditional methods suffer from limited DMSS solubility in methanol and difficult solvent recovery due to water by-products. The new method utilizes the high boiling point of ethylene glycol to create a homogeneous reaction phase at higher temperatures, significantly improving reaction efficiency and simplifying downstream separation.
Q: What represents the critical quality control point in this synthesis route?
A: The adsorption and decolorization step prior to acidification is critical. By removing impurities from the high-concentration terephthalate salt solution before crystallization, the process ensures the final solid crystal has superior purity and color properties compared to traditional low-concentration methods.
Q: Is this process scalable for industrial pigment manufacturing?
A: Yes, the process avoids high-pressure operations associated with volatile solvents and utilizes standard stirred tank or tubular reactors. The ability to recycle the ethylene glycol and water dispersion medium makes it highly suitable for large-scale commercial production with reduced environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diphenylamino Terephthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of high-performance pigments relies on the consistent supply of ultra-pure intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,5-diphenylamino terephthalic acid meets the exacting standards required for quinacridone pigment synthesis, providing our clients with a secure and high-quality foundation for their downstream applications.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthesis route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the fine chemical and pigment industries.
