Advanced Synthesis of 3,3'-Diamino-4,4'-Dihydroxybiphenyl for High-Performance Polyimide Manufacturing
The rapid evolution of the flexible copper-clad laminate (FCCL) and microelectronics sectors demands precursors with exceptional thermal stability and dielectric properties. Patent CN101397258B introduces a robust and highly efficient preparation method for 3,3'-diamino-4,4'-dihydroxybiphenyl, a critical monomer for synthesizing hydroxyl-containing polyimide resins. This technical breakthrough addresses the longstanding industry challenge of balancing high purity with cost-effective manufacturing processes. By utilizing a catalytic transfer hydrogenation system involving palladium on carbon (Pd/C) and hydrazine hydrate, the process achieves product purity levels exceeding 99% without necessitating complex downstream purification steps. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, this methodology represents a significant optimization in the supply chain for high-performance polymer materials.
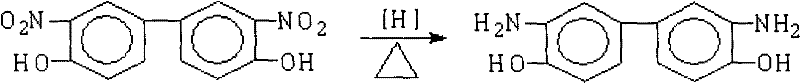
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for aromatic diamines often rely on catalytic hydrogenation using high-pressure hydrogen gas or stoichiometric metal reductions such as iron powder in acidic media. These conventional approaches present substantial operational hazards and environmental burdens that complicate cost reduction in electronic chemical manufacturing. High-pressure hydrogenation requires specialized autoclaves and rigorous safety protocols, increasing both capital expenditure and maintenance overheads for production facilities. Alternatively, metal reduction methods generate vast quantities of toxic metal sludge and acidic wastewater, creating severe disposal challenges and regulatory compliance risks. Furthermore, these older techniques frequently struggle to achieve the ultra-high purity standards required for advanced polyimide applications, often necessitating energy-intensive recrystallization or chromatographic purification steps that erode overall process yield and profitability.
The Novel Approach
The methodology disclosed in CN101397258B circumvents these inefficiencies by employing hydrazine hydrate as a liquid hydrogen donor in the presence of a heterogeneous Pd/C catalyst. This catalytic transfer hydrogenation operates under atmospheric pressure conditions within a temperature range of 70°C to 150°C, drastically simplifying the reactor requirements and enhancing operational safety. The reaction proceeds cleanly over a period of 1 to 5 hours, converting 3,3'-dinitro-4,4'-dihydroxybiphenyl directly into the target diamine with minimal byproduct formation. A distinct advantage of this novel approach is the ability to isolate the final product simply by hot filtration to remove the catalyst followed by cooling the mother liquor to induce crystallization. This elimination of post-reaction refining processes not only accelerates the production cycle but also ensures that the commercial scale-up of complex electronic intermediates remains economically viable and environmentally sustainable.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthesis lies in the efficient activation of hydrazine by the palladium surface, which facilitates the transfer of hydrogen atoms to the nitro groups of the substrate. In this mechanism, hydrazine adsorbs onto the active sites of the Pd/C catalyst, where it undergoes dehydrogenation to release nitrogen gas and reactive hydrogen species. These activated hydrogen atoms are then transferred to the nitro groups (-NO2) of the 3,3'-dinitro-4,4'-dihydroxybiphenyl, sequentially reducing them through nitroso and hydroxylamine intermediates to the final amino groups (-NH2). The use of polar organic solvents such as ethylene glycol, dimethyl sulfoxide, or alcohols plays a crucial role in stabilizing the transition states and ensuring the solubility of both the starting material and the intermediate species throughout the reaction trajectory. This homogeneous-heterogeneous interface allows for precise control over the reduction kinetics, preventing over-reduction or side reactions that could compromise the structural integrity of the biphenyl backbone.
Impurity control is inherently managed by the specificity of the Pd/C catalyst and the volatility of the nitrogen byproduct. Unlike metal reductions that introduce metallic ions into the reaction matrix, this catalytic system leaves no residual metal contaminants in the solution phase, provided the hot filtration step is executed effectively. The patent data indicates that maintaining the reaction temperature between 70°C and 150°C is critical for driving the reaction to completion while minimizing the formation of azo or azoxy coupling byproducts which can occur if the reaction stalls at the nitroso stage. The resulting crystalline product precipitates from the mother liquor with a purity of more than 99 percent, as confirmed by melting point analysis and HPLC data in the examples. This high level of chemical fidelity is essential for downstream polymerization, where even trace impurities can act as chain terminators or defects, degrading the thermal and mechanical performance of the final polyimide film.
How to Synthesize 3,3'-Diamino-4,4'-Dihydroxybiphenyl Efficiently
To implement this synthesis effectively, manufacturers must carefully optimize the ratio of substrate to catalyst and reducing agent. The patent specifies a weight ratio of 3,3'-dinitro-4,4'-dihydroxybiphenyl to Pd/C between 100:1 and 100:20, allowing flexibility based on catalyst activity and cost considerations. Similarly, the molar ratio of substrate to hydrazine hydrate is maintained between 1:10 and 1:30 to ensure complete conversion without excessive reagent waste. The detailed standardized synthesis steps, including specific solvent combinations and temperature profiles validated across multiple embodiments, are outlined below to guide process engineers in replicating these high-yield results.
- Charge the reactor with 3,3'-dinitro-4,4'-dihydroxybiphenyl, Pd/C catalyst (1-15% Pd loading), and a suitable organic solvent system such as ethylene glycol or alcohol mixtures.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60-85% concentration) while maintaining a molar ratio of substrate to hydrazine between 1: 10 and 1:30.
- Maintain the reaction temperature between 70°C and 150°C for 1 to 5 hours, then filter hot to remove the catalyst, cool the mother liquor to crystallize the product, and vacuum dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrazine-based reduction pathway offers tangible strategic benefits beyond mere technical feasibility. The simplification of the workup procedure directly translates to reduced labor costs and shorter batch cycle times, enhancing the overall throughput of the manufacturing facility. By eliminating the need for high-pressure equipment and toxic metal waste disposal, the process significantly lowers the barrier to entry for production and reduces long-term operational liabilities. This efficiency makes it an attractive option for securing a reliable supply of high-purity electronic chemicals in a volatile market.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the elimination of expensive purification infrastructure and the reduction of energy consumption. Since the product crystallizes directly from the reaction mixture upon cooling, there is no need for solvent swapping, column chromatography, or multiple recrystallization steps that typically consume large volumes of solvents and utilities. Furthermore, the use of hydrazine hydrate avoids the capital intensity associated with high-pressure hydrogenation units, allowing existing standard reactor setups to be utilized for production. This streamlined workflow ensures that the cost of goods sold (COGS) is minimized, providing a competitive pricing structure for downstream polyimide manufacturers without compromising on quality specifications.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including hydrazine hydrate, Pd/C, and common organic solvents like ethanol and ethylene glycol, are commodity chemicals with robust global supply chains. This availability mitigates the risk of production stoppages due to raw material shortages, a common issue with specialized reagents. Additionally, the reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors, meaning that production can be easily outsourced or scaled across multiple contract manufacturing organizations (CMOs) without requiring specialized high-pressure certification. This flexibility ensures consistent delivery schedules and reduces lead time for high-purity electronic intermediates even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process generates significantly less hazardous waste compared to traditional metal reduction methods. The primary byproduct is nitrogen gas, which is harmless and vents easily, while the spent Pd/C catalyst can be recovered and recycled for precious metal value. The absence of heavy metal sludge simplifies wastewater treatment protocols and ensures compliance with increasingly stringent environmental regulations in major manufacturing hubs. This 'green' profile not only reduces disposal costs but also aligns with the sustainability goals of major multinational electronics corporations, making the supply chain more resilient against regulatory shifts and enhancing the brand value of the final polymer products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,3'-diamino-4,4'-dihydroxybiphenyl. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on process parameters and quality outcomes.
Q: What is the typical purity level achievable with this hydrazine reduction method?
A: According to patent CN101397258B, this specific catalytic transfer hydrogenation process consistently yields 3,3'-diamino-4,4'-dihydroxybiphenyl with a purity exceeding 99%, often reaching up to 99.9% without the need for recrystallization or column chromatography.
Q: Why is hydrazine hydrate preferred over catalytic hydrogenation for this intermediate?
A: Hydrazine hydrate serves as a liquid hydrogen donor in the presence of Pd/C, eliminating the need for high-pressure hydrogen gas equipment. This significantly reduces capital expenditure on safety infrastructure and simplifies the operational workflow for batch processing in standard glass-lined or stainless steel reactors.
Q: Can this process be scaled for industrial production of polyimide precursors?
A: Yes, the patent explicitly states the method is applicable to industrial production due to its simple operation, short reaction time (1-5 hours), and minimal three-waste generation. The use of common solvents like ethylene glycol and ethanol further facilitates easy scale-up and solvent recovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Diamino-4,4'-Dihydroxybiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the performance of next-generation flexible electronics hinges on the quality of the underlying monomers. Our technical team has extensively analyzed the pathway described in CN101397258B and possesses the expertise to optimize this catalytic transfer hydrogenation for large-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications required for polyimide synthesis. Our rigorous QC labs are equipped to verify critical parameters such as melting point, amine value, and trace metal content, guaranteeing that our 3,3'-diamino-4,4'-dihydroxybiphenyl supports the high thermal stability and dielectric strength your applications demand.
We invite you to collaborate with us to leverage this efficient synthesis route for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your product performance while reducing overall material costs.
