Advanced Synthesis of N-(β-Aminoacrylate) Indoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical landscape is constantly evolving, driven by the need for novel scaffolds that can address resistant bacterial strains and complex therapeutic challenges. Patent CN114702430A introduces a groundbreaking methodology for the synthesis of N-(β-aminoacrylate) substituted indole compounds, a class of molecules that merges the pharmacological potential of indole alkaloids with the versatile reactivity of beta-amino acid derivatives. This innovation addresses a long-standing synthetic hurdle: the selective functionalization of the indole nitrogen (N1) position, which has historically been overshadowed by the higher reactivity of the carbon positions. By leveraging a selective addition reaction between indoles and azaallenes, this technology opens new avenues for creating high-value pharmaceutical intermediates with potent antibacterial properties. The structural versatility of this approach allows for extensive modification of the R1, R2, and R3 groups, enabling medicinal chemists to fine-tune biological activity while maintaining a robust synthetic pathway suitable for industrial application.
As a leading entity in the fine chemical sector, understanding the implications of such patents is crucial for maintaining a competitive edge in the supply of reliable pharmaceutical intermediate suppliers. The ability to access these complex heterocyclic structures efficiently translates directly into accelerated drug discovery timelines and more cost-effective production pipelines. The patent details a comprehensive library of derivatives, demonstrating that the method is not limited to a single substrate but possesses broad universality. This generality is a key indicator of a mature chemical process, suggesting that the underlying mechanism is robust enough to tolerate various electronic and steric environments. For procurement and R&D teams alike, this represents a significant opportunity to diversify their portfolio of bioactive building blocks without incurring the prohibitive costs often associated with exotic heterocycle synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of indole rings has been heavily biased towards the C2 and C3 positions due to their inherent nucleophilicity in electrophilic aromatic substitution reactions. Attempting to modify the N1 position often requires harsh conditions, protecting group strategies that add multiple steps to the synthesis, or the use of expensive transition metal catalysts that pose challenges for removal and regulatory compliance in final drug products. Furthermore, conventional methods for introducing beta-amino acid motifs often suffer from poor stereocontrol or require complex multistep sequences that drastically reduce overall yield. These limitations create bottlenecks in the cost reduction in API manufacturing, as each additional step introduces material loss, increased solvent waste, and extended processing time. The inability to directly and selectively couple indole nitrogen with acrylate precursors has restricted the chemical space available for exploring new antibacterial agents based on this privileged scaffold.
The Novel Approach
The methodology disclosed in CN114702430A circumvents these historical constraints by utilizing a direct nucleophilic addition of the indole N1 atom to an azaallene system. This strategy eliminates the need for pre-functionalization of the indole nitrogen or the use of rare earth catalysts. The reaction proceeds under relatively mild conditions, with temperatures ranging from -60°C to 150°C depending on the specific substrate reactivity, and utilizes common organic bases as catalysts. This shift from complex metal catalysis to simple base promotion is a paradigm shift that simplifies the operational complexity of the synthesis. By merging the indole and beta-amino acrylate units in a single convergent step, the process significantly shortens the synthetic route. This efficiency is critical for achieving high-purity pharmaceutical intermediates at a scale that meets commercial demand, as fewer steps inherently mean fewer opportunities for impurity generation and easier downstream purification.
Mechanistic Insights into Base-Catalyzed N1-Selective Addition
The core of this technological advancement lies in the mechanistic pathway where the indole nitrogen acts as a nucleophile attacking the electrophilic central carbon of the azaallene moiety. The presence of a base catalyst, such as DBU or potassium carbonate, facilitates the deprotonation of the indole N-H bond, generating a more reactive indolyl anion species. This anion then undergoes a conjugate addition to the electron-deficient azaallene system, forming the new C-N bond that defines the N-(β-aminoacrylate) structure. The reaction is highly sensitive to the electronic nature of the substituents on both the indole ring and the azaallene, allowing for fine-tuning of reaction kinetics. The patent data indicates that the reaction can proceed in a variety of polar and non-polar solvents, suggesting a mechanism that is adaptable to different solubility profiles of the starting materials. This mechanistic flexibility is a strong indicator of process robustness, ensuring that the reaction can be optimized for different scales without fundamental changes to the chemistry.
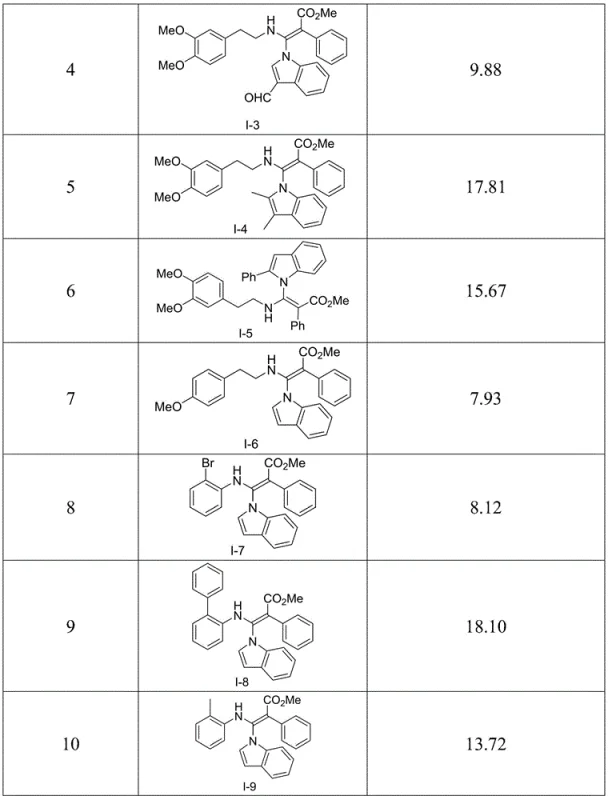
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring product quality. Harsh acidic or basic conditions often lead to polymerization of the acrylate species or degradation of the sensitive indole ring. However, the controlled basic environment described in the patent minimizes these side reactions. The use of stoichiometric amounts of catalyst (ranging from 0.05% to 100%) allows for precise control over the reaction rate, preventing runaway exotherms that could compromise safety and purity. Furthermore, the byproduct profile is likely clean, consisting primarily of unreacted starting materials which can be easily separated via standard chromatographic techniques. This high level of control over the reaction trajectory ensures that the resulting commercial scale-up of complex pharmaceutical intermediates yields material that meets stringent regulatory specifications for residual solvents and heavy metals, a critical factor for any supplier aiming to serve the global pharmaceutical market.
How to Synthesize N-(β-Aminoacrylate) Indoles Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the careful selection of the indole substrate and the azaallene precursor, ensuring that they are dry and free from inhibitors that might interfere with the base catalysis. The reaction is typically conducted under an inert atmosphere, such as nitrogen, to prevent oxidation of the sensitive amine or enamine functionalities. Following the mixing of reagents and catalyst in a suitable solvent like acetonitrile or toluene, the mixture is heated to the optimal temperature determined by the specific substrate combination. Monitoring the reaction progress via TLC or HPLC is essential to determine the endpoint, ensuring maximum conversion before workup. The detailed standardized synthesis steps for optimizing this process are provided in the guide below.
- Mix the indole substrate (Formula II), 3-(phenylethylimino)-2-aryl-acrylate compound (Formula III), and a base catalyst (e.g., DBU, K2CO3) in a suitable organic solvent under nitrogen protection.
- Stir the reaction mixture at a controlled temperature ranging from -60°C to 150°C until TLC analysis indicates complete consumption of the starting materials.
- Isolate the target N-(β-aminoacrylate) substituted indole compound (Formula I) through standard workup procedures such as extraction and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike processes that rely on proprietary ligands or scarce transition metals, this method utilizes commodity chemicals such as potassium carbonate, triethylamine, and common organic solvents. This accessibility ensures that the reducing lead time for high-purity pharmaceutical intermediates is achievable, as there is no dependency on single-source suppliers for exotic catalysts. The robustness of the reaction across a wide temperature range also implies that the process is forgiving of minor fluctuations in plant conditions, enhancing overall manufacturing reliability and reducing the risk of batch failures that can disrupt supply continuity.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a significant opportunity for cost optimization. Precious metals like palladium or rhodium not only carry high upfront costs but also require rigorous and costly removal processes to meet ppm-level specifications in final drug substances. By replacing these with inexpensive organic or inorganic bases, the cost of goods sold (COGS) is substantially lowered. Additionally, the convergent nature of the synthesis, which combines two complex fragments in a single step, reduces the total number of unit operations. Fewer reaction steps translate directly to lower labor costs, reduced energy consumption for heating and cooling, and decreased solvent usage, all of which contribute to a leaner and more economically efficient manufacturing process that maximizes margin potential.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as substituted indoles and acrylate derivatives which are widely produced for various chemical industries, mitigates the risk of supply shortages. In a global market prone to logistical disruptions, having a synthesis route that depends on bulk commodities rather than specialized fine chemicals is a strategic asset. The broad substrate scope demonstrated in the patent means that if one specific indole derivative becomes unavailable, structurally similar analogs can often be substituted with minimal process re-optimization. This flexibility ensures business continuity and allows for rapid adaptation to changing market demands or raw material availability, securing the supply chain against volatility and ensuring consistent delivery to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, utilizing standard solvents and temperatures that are compatible with existing stainless steel reactor infrastructure. There is no requirement for specialized high-pressure equipment or cryogenic conditions that would necessitate significant capital expenditure for new plant installations. Furthermore, the use of simpler catalysts and the potential for solvent recycling aligns well with green chemistry principles. Reducing the environmental footprint of chemical manufacturing is increasingly a requirement for doing business with major pharmaceutical companies. By minimizing hazardous waste and avoiding toxic heavy metals, this process facilitates easier regulatory approval and waste disposal, streamlining the path from bench-scale discovery to multi-ton commercial production while adhering to strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines and for procurement teams assessing the long-term viability of the supply source.
Q: What is the primary technical breakthrough of this synthesis method?
A: The primary breakthrough is the achievement of stereoselective functionalization at the indole N1-position via selective addition to azaallenes, overcoming the historical difficulty of modifying this specific site compared to the more reactive C2 or C3 positions.
Q: Are the catalysts used in this process expensive or rare?
A: No, the process utilizes readily available and inexpensive catalysts such as potassium carbonate, triethylamine, or DBU, which significantly lowers the raw material costs compared to methods requiring precious transition metals.
Q: What is the biological application of these compounds?
A: These N-(β-aminoacrylate) substituted indole compounds exhibit significant antibacterial activity, particularly against Gram-negative bacilli, making them valuable candidates for developing new antibacterial drugs or intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(β-Aminoacrylate) Indole Supplier
The technological potential of N-(β-aminoacrylate) substituted indoles is immense, offering a new frontier in the development of antibacterial therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this patented chemistry into reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest industry standards. Our commitment to quality assurance means that you can rely on us for consistent, high-performance materials that support your regulatory filings and product efficacy.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our manufacturing capabilities can support your project goals. Let us help you navigate the complexities of fine chemical sourcing with a partner dedicated to innovation and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
