Advanced Manufacturing of 3,3'-Diamino-4,4'-bis(4-cyanophenoxy)biphenyl for High-Performance Polyimides
The rapid evolution of the electronics industry, particularly in the sectors of flexible copper clad laminates (FCCL) and very large scale integration (VLSI), demands materials with exceptional thermal stability and mechanical strength. At the forefront of this material science revolution is the aromatic dibasic primary amine known as 3,3'-diamino-4,4'-bis(4-cyanophenoxy)biphenyl, a critical monomer for synthesizing high-performance polyimide resins. The preparation method detailed in patent CN101225059A represents a significant technological breakthrough, offering a robust pathway to produce this complex molecule with industrial viability. Unlike traditional synthetic routes that often struggle with impurity profiles and difficult purification steps, this novel approach leverages a precise nucleophilic aromatic substitution strategy. By optimizing reaction parameters such as temperature, solvent systems, and molar ratios, the process ensures the production of high-purity intermediates essential for next-generation electronic insulation materials and liquid crystal alignment agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines containing ether bonds and cyano groups has been plagued by significant technical hurdles that impact both cost and quality. Conventional methods often rely on harsh reaction conditions that can lead to the degradation of sensitive functional groups, resulting in a complex impurity spectrum that is difficult to separate. Furthermore, many existing processes utilize solvent systems that are challenging to recover, leading to substantial environmental waste and increased operational costs for manufacturers. The inability to effectively remove inorganic salts and by-products without losing significant amounts of the target product has been a persistent bottleneck. These inefficiencies not only drive up the price of the final polyimide resin but also compromise the dielectric properties required for high-frequency electronic applications. Consequently, there has been a pressing need for a streamlined synthesis method that addresses these purification and yield challenges simultaneously.
The Novel Approach
The methodology disclosed in CN101225059A introduces a refined one-pot synthesis strategy that dramatically simplifies the production workflow while enhancing product quality. This innovative route utilizes a specific combination of an aprotic polar solvent and an entrainer, such as the DMF-toluene system, to facilitate the reaction under nitrogen protection. The core of this advancement lies in the controlled reaction environment where 3,3'-diamino-4,4'-dihydroxybiphenyl reacts with 4-halobenzonitrile in the presence of a salt-forming agent. 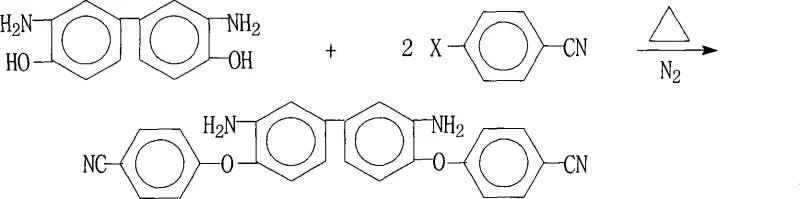 This specific configuration allows for the efficient displacement of the halogen atom by the phenoxide ion, driven by the electron-withdrawing nature of the cyano group. The process includes a crucial hot filtration step that removes insoluble inorganic salts before the product crystallizes, thereby preventing contamination and ensuring a purity level exceeding 99%. This approach not only improves the yield significantly, reaching up to 96.3% in optimized examples, but also enables the recycling of solvents, aligning with modern green chemistry principles.
This specific configuration allows for the efficient displacement of the halogen atom by the phenoxide ion, driven by the electron-withdrawing nature of the cyano group. The process includes a crucial hot filtration step that removes insoluble inorganic salts before the product crystallizes, thereby preventing contamination and ensuring a purity level exceeding 99%. This approach not only improves the yield significantly, reaching up to 96.3% in optimized examples, but also enables the recycling of solvents, aligning with modern green chemistry principles.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The chemical transformation at the heart of this synthesis is a classic yet meticulously optimized nucleophilic aromatic substitution (SNAr) reaction. The mechanism initiates with the deprotonation of the hydroxyl groups on the 3,3'-diamino-4,4'-dihydroxybiphenyl backbone by the salt-forming agent, typically potassium carbonate or potassium hydroxide. This deprotonation generates a highly reactive phenoxide anion, which serves as the nucleophile in the subsequent step. The presence of the cyano group on the benzene ring of the 4-halobenzonitrile is critical, as it acts as a powerful electron-withdrawing group that activates the aromatic ring towards nucleophilic attack. This activation lowers the energy barrier for the formation of the Meisenheimer complex, a key intermediate in the SNAr pathway. The reaction proceeds through the addition of the phenoxide to the carbon bearing the halogen, followed by the elimination of the halide ion, ultimately forming the stable ether linkage that characterizes the final bis(4-cyanophenoxy) structure.
Controlling the impurity profile in such a complex molecule requires a deep understanding of the reaction kinetics and thermodynamics involved in this catalytic cycle. The use of an entrainer like toluene plays a dual role: it forms an azeotrope with water produced during the salt formation, driving the equilibrium towards the completion of the deprotonation step, and it helps maintain a homogeneous reaction phase at elevated temperatures. The temperature range of 120°C to 200°C is carefully selected to provide sufficient thermal energy for the substitution to occur without triggering side reactions such as the hydrolysis of the nitrile group or the decomposition of the amine functionalities. Furthermore, the molar ratio of the salt-forming agent to the dihydroxybiphenyl is maintained between 1.2:1 and 3.2:1 to ensure complete conversion of the phenolic hydroxyls while minimizing excess base that could complicate downstream purification. This precise control over the reaction environment is what enables the production of high-purity material suitable for sensitive electronic applications.
How to Synthesize 3,3'-Diamino-4,4'-bis(4-cyanophenoxy)biphenyl Efficiently
Implementing this synthesis route on an industrial scale requires strict adherence to the procedural parameters outlined in the patent to ensure reproducibility and safety. The process begins with the careful charging of the reactor with the defined molar ratios of reactants and solvents under an inert nitrogen atmosphere to prevent oxidation of the amine groups. Operators must monitor the temperature ramp-up closely, moving from ambient conditions to the target range of 120°C to 200°C over a controlled period to manage exotherms and ensure uniform mixing. The reaction duration, typically spanning 10 to 30 hours, is critical for achieving full conversion, and sampling may be required to monitor the disappearance of starting materials. Following the reaction, the workup procedure involving hot filtration and vacuum concentration is essential to isolate the crude product before the final crystallization step yields the pure white solid. For a comprehensive breakdown of the standardized operating procedures and safety protocols, please refer to the detailed guide below.
- Charge the reactor with 3,3'-diamino-4,4'-dihydroxybiphenyl and 4-halobenzonitrile (molar ratio 1: 2.0-2.4) along with a salt-forming agent like potassium carbonate in an aprotic polar solvent and entrainer.
- Heat the mixture under nitrogen protection to a temperature range of 120°C to 200°C and maintain reaction for 10 to 30 hours to ensure complete conversion.
- Perform hot filtration to remove inorganic salts, concentrate the mother liquor under reduced pressure, and induce crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers substantial strategic advantages for procurement managers and supply chain directors looking to optimize their raw material sourcing. The simplicity of the one-pot reaction design eliminates the need for multiple intermediate isolation steps, which traditionally consume significant time and resources. This streamlining of the manufacturing process directly translates to reduced operational expenditures and a more predictable production schedule. Furthermore, the ability to recover and recycle the solvent system, particularly the expensive aprotic polar solvents, contributes to a more sustainable and cost-effective supply chain model. By minimizing waste generation and improving overall process efficiency, manufacturers can offer more competitive pricing without compromising on the stringent quality standards required by the electronics industry.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily derived from the elimination of complex purification stages and the high efficiency of the reaction itself. By utilizing a salt-forming agent that generates easily filterable by-products, the method avoids the need for extensive chromatographic purification or multiple recrystallizations that often result in significant yield loss. The high conversion rate ensures that raw materials are utilized effectively, reducing the cost per kilogram of the final active pharmaceutical ingredient or electronic chemical. Additionally, the recovery of solvents like toluene and DMF allows for a closed-loop system that drastically cuts down on raw material consumption and waste disposal costs, providing a clear path to margin improvement for large-scale producers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain stability by reducing the dependency on hard-to-source reagents or specialized catalysts. The starting materials, such as 3,3'-diamino-4,4'-dihydroxybiphenyl and 4-halobenzonitrile, are commercially available and can be sourced from multiple suppliers, mitigating the risk of single-source bottlenecks. The straightforward nature of the reaction conditions, which do not require extreme pressures or exotic catalysts, means that production can be easily scaled across different manufacturing sites without significant retooling. This flexibility ensures consistent availability of the high-purity monomer, allowing downstream customers to maintain their own production schedules for polyimide films and adhesives without interruption.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste management intensify, this process offers a compliant pathway for scaling production. The use of a nitrogen-protected environment minimizes the release of volatile organic compounds, and the efficient solvent recovery system ensures that hazardous waste generation is kept to a minimum. The solid by-products generated during the hot filtration step are primarily inorganic salts, which are easier to handle and dispose of compared to complex organic sludge. This environmental compatibility not only reduces the liability associated with waste treatment but also aligns with the corporate sustainability goals of major electronics manufacturers who prioritize green supply chains.
Frequently Asked Questions (FAQ)
To further clarify the technical specifications and operational details of this synthesis method, we have compiled a list of frequently asked questions based on the patent data. These inquiries address common concerns regarding reaction conditions, solvent choices, and product quality metrics that are critical for R&D teams evaluating this technology. Understanding these nuances is essential for successfully transferring this laboratory-scale method to a commercial production environment. The answers provided below are derived directly from the experimental examples and claims found in the intellectual property documentation.
Q: What is the optimal molar ratio for the reactants in this synthesis?
A: According to patent CN101225059A, the optimal molar ratio of 3,3'-diamino-4,4'-dihydroxybiphenyl to 4-halobenzonitrile is between 1:2.0 and 1:2.4 to ensure high yield and minimize unreacted starting materials.
Q: Which solvents are recommended for this reaction process?
A: The process utilizes a mixed solvent system comprising an aprotic polar solvent such as N,N-dimethylformamide (DMF) or N-methylpyrrolidone (NMP), combined with an entrainer like toluene or o-dichlorobenzene to facilitate water removal.
Q: What purity levels can be achieved with this preparation method?
A: By employing hot filtration followed by crystallization, the method achieves product purity exceeding 99%, which is critical for high-performance electronic applications like FCCL and VLSI passivation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Diamino-4,4'-bis(4-cyanophenoxy)biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of electronic materials and specialty polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global supply chains. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require this specific aromatic diamine for polyimide resin synthesis or custom derivatives for liquid crystal applications, our technical expertise guarantees a supply of material that meets the highest industry standards for thermal and electrical performance.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific application requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized process can reduce your overall manufacturing costs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to validate the quality and compatibility of our materials with your existing formulations. By choosing NINGBO INNO PHARMCHEM, you are securing a partnership dedicated to innovation, quality, and long-term supply chain reliability.
