Advanced Synthesis of Tetramethyldiphenylsulfone Derivatives for High-Performance Polyimide Manufacturing
The rapid advancement of high-performance materials in the aerospace and electronic microelectronics sectors has created an urgent demand for aromatic polyimides with superior thermal stability and mechanical strength. Patent CN101235005A introduces a groundbreaking preparation method for 4,4'-bis(2,4-dinitrophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone, a pivotal intermediate in the synthesis of highly branched aromatic polyimide monomers. This specific chemical structure enables the production of new polyimide materials that exhibit exceptional resistance to high temperatures, corrosion, and organic solvents, making them ideal for demanding applications such as liquid crystal displays (LCDs) and advanced electrical insulation systems. The innovation lies not only in the molecular architecture but also in the robust industrial scalability of the synthesis route, which addresses long-standing challenges in purity and yield that have historically hindered the mass adoption of these specialty chemicals.
For R&D Directors and technical decision-makers, understanding the provenance and reliability of this synthetic route is crucial for integrating it into existing supply chains. The patent details a process that moves beyond theoretical laboratory success to demonstrate practical viability for commercial scale-up of complex electronic chemical intermediates. By leveraging a specific nucleophilic aromatic substitution mechanism, the method ensures that the final product meets the stringent purity specifications required for downstream polymerization processes. This level of quality control is essential for maintaining the integrity of the final polyimide films, where even trace impurities can compromise dielectric properties or thermal performance. Consequently, this technology represents a significant leap forward for manufacturers seeking a reliable agrochemical intermediate supplier or partner in the electronic materials space who can guarantee consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diphenylsulfone derivatives has been plagued by inefficiencies that drive up costs and complicate supply chain logistics for procurement managers. Traditional methods often suffer from incomplete reactions due to poor solubility of reactants or inadequate removal of byproducts, leading to lower yields and a complex mixture of impurities that are difficult to separate. In many conventional processes, the use of single-solvent systems fails to effectively manage the water generated during the etherification reaction, which can reverse the reaction equilibrium and limit conversion rates. Furthermore, older techniques frequently require harsh conditions or expensive catalysts that introduce heavy metal contaminants, necessitating costly purification steps that erode profit margins. These limitations result in inconsistent batch quality, extended lead times for high-purity electronic chemical intermediates, and significant waste generation that poses environmental compliance risks for modern manufacturing facilities.
The Novel Approach
The patented methodology described in CN101235005A overcomes these historical barriers through a sophisticated dual-solvent system and precise stoichiometric control. By employing a mixture of water-insoluble organic solvents and strong polar aprotic organic solvents, the process creates an optimal environment for the nucleophilic attack while facilitating the continuous removal of water via azeotropic distillation. This strategic approach drives the reaction equilibrium towards the product side, ensuring high conversion rates without the need for excessive reagent loading. The operation is conducted under normal pressure, eliminating the need for specialized high-pressure equipment and reducing capital investment risks for production facilities. Additionally, the simplicity of the work-up procedure, which involves concentration, cooling, and water precipitation, streamlines the isolation of the solid product, thereby drastically simplifying the overall manufacturing workflow and enhancing operational efficiency for supply chain heads.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core of this synthesis relies on a classic yet optimized nucleophilic aromatic substitution reaction where the phenoxide anion attacks the electron-deficient aromatic ring of the dinitrohalobenzene. The presence of two strong electron-withdrawing nitro groups at the ortho and para positions relative to the halogen atom significantly activates the ring towards nucleophilic attack, allowing the reaction to proceed efficiently under the specified thermal conditions. The choice of the salt-forming agent, such as potassium carbonate or sodium hydroxide, is critical for generating the reactive phenoxide species in situ without causing side reactions like hydrolysis of the nitro groups. The reaction temperature, maintained between 80°C and 200°C, provides the necessary activation energy to overcome the kinetic barrier while ensuring the stability of the sensitive functional groups involved in the transformation.
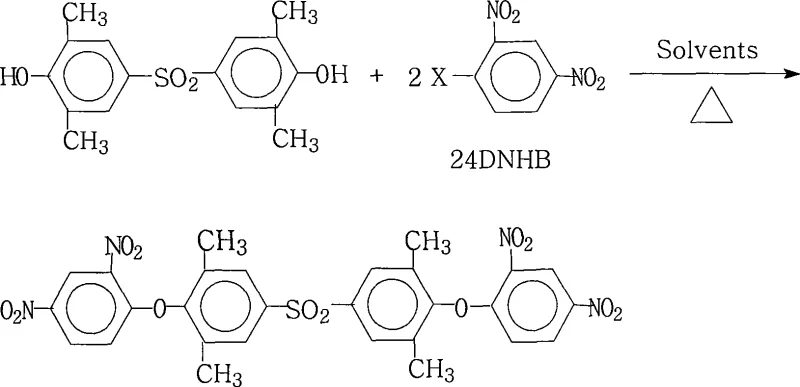
Impurity control is meticulously managed through the precise molar ratio of reactants, typically maintained at 1.0:2.0~2.2 for the dihydroxy sulfone to the dinitrohalobenzene. This slight excess of the halogenated reactant ensures that the dihydroxy starting material is fully consumed, preventing the formation of mono-substituted byproducts that could act as chain terminators in subsequent polymerization steps. The solvent system plays a dual role not only in dissolving the reactants but also in sequestering the inorganic salts formed during the reaction, preventing them from occluding within the crystal lattice of the product. The final crystallization step, induced by the addition of water to the concentrated reaction mass, leverages the differential solubility of the product versus the impurities, resulting in a crystalline solid with purity levels reaching 99.0% or higher as demonstrated in the experimental examples. This rigorous control over the reaction parameters ensures a clean impurity profile, which is vital for the performance of the final polyimide material.
How to Synthesize 4,4'-bis(2,4-dinitrophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone Efficiently
The synthesis of this high-value intermediate requires careful attention to the preparation of the reaction mixture and the management of the reflux process to ensure optimal yield and purity. The process begins with the precise weighing and charging of 4,4'-dihydroxy-3,3',5,5'-tetramethyldiphenylsulfone and the selected 2,4-dinitrohalobenzene into a reactor equipped with a water separator. A suitable salt-forming agent is added along with the binary solvent system, and the mixture is heated to reflux to initiate the reaction and remove water continuously. Detailed standardized synthesis steps regarding specific temperatures, stirring rates, and work-up procedures are provided in the guide below to assist technical teams in replicating this successful protocol.
- Prepare the reaction mixture by combining 4,4'-dihydroxy-3,3',5,5'-tetramethyldiphenylsulfone and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0~2.2 with a salt-forming agent.
- Heat the organic solvent system to reflux temperatures between 80°C and 200°C for 6 to 18 hours to facilitate water separation and reaction completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The process is designed with industrial scalability in mind, utilizing raw materials that are readily available in the global chemical market, which mitigates the risk of supply disruptions common with exotic reagents. The ability to recover and recycle the organic solvents repeatedly means that the consumption of consumables is significantly reduced, leading to a more sustainable and cost-effective operation over the long term. This efficiency translates directly into a more stable pricing structure for buyers, as the manufacturer is less exposed to volatility in raw material costs and waste disposal fees. Furthermore, the high yield and purity reduce the need for extensive downstream purification, shortening the overall production cycle time and enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the ability to recycle solvents contribute to a drastic simplification of the production process, which inherently lowers operational expenditures. By avoiding the use of expensive transition metal catalysts that require removal, the process reduces both material costs and the environmental burden associated with heavy metal waste treatment. The high atom economy of the reaction ensures that a maximum proportion of the starting materials is converted into the desired product, minimizing waste and maximizing resource utilization. These factors combine to create a manufacturing profile that supports significant cost savings without compromising on the quality or performance of the final chemical intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which operate under normal pressure and tolerate a range of temperatures, ensures consistent production output regardless of minor fluctuations in utility supplies. The use of common industrial solvents and reagents means that sourcing is straightforward and less prone to geopolitical or logistical bottlenecks that can plague specialized chemical supply chains. This reliability allows for better inventory planning and reduces the need for safety stock, freeing up working capital for other strategic investments. Ultimately, a stable and predictable manufacturing process fosters a stronger partnership between the supplier and the buyer, ensuring continuity of supply for critical electronic and aerospace applications.
- Scalability and Environmental Compliance: The process generates minimal three-waste emissions, aligning with increasingly stringent global environmental regulations and reducing the liability associated with waste disposal. The simplicity of the equipment requirements allows for easy scale-up from pilot plant to full commercial production without the need for major engineering redesigns or capital-intensive infrastructure upgrades. This scalability ensures that the supplier can rapidly respond to increases in demand, supporting the growth of customers in the fast-evolving fields of OLED materials and polymer additives. The environmentally friendly nature of the process also enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize sustainability in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized sulfone derivative. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific manufacturing workflows and product development pipelines.
Q: What are the primary applications of this sulfone derivative?
A: This compound serves as a critical monomer for synthesizing highly branched aromatic polyimides, which are essential for high-temperature resistant materials in aerospace and LCD display technologies.
Q: How does this method improve upon conventional synthesis routes?
A: The patented method utilizes a specific dual-solvent system and azeotropic water removal, resulting in significantly higher yields up to 96.8% and purity exceeding 99.0% compared to traditional methods.
Q: Is the process environmentally sustainable for large-scale production?
A: Yes, the process allows for convenient solvent recovery and recycling, minimizes waste generation, and operates under normal pressure without producing corrosive byproducts, ensuring environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-bis(2,4-dinitrophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation advanced materials. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory concept to industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that consistency is key for your R&D and production teams, and our state-of-the-art facilities are designed to deliver the reliability you need to maintain your competitive edge in the global market.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments that will demonstrate the tangible value of partnering with NINGBO INNO PHARMCHEM for your fine chemical intermediate needs.
