Advanced 3-Step Synthesis of 2-Bromo-6-Fluoronaphthalene for Commercial Scale-Up
Advanced 3-Step Synthesis of 2-Bromo-6-Fluoronaphthalene for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access complex halogenated aromatic intermediates, and patent CN101870636A presents a groundbreaking solution for the production of 2-bromo-6-fluoronaphthalene. This specific compound serves as a critical building block for synthesizing niacin receptor competitors and advanced liquid crystal materials, yet its historical manufacturing processes have been plagued by complexity and high costs. The disclosed innovation utilizes low-cost Tobias acid as the foundational starting material, streamlining the entire synthetic sequence into merely three robust steps involving bromination-debromination, diazotization, and thermal cracking. By shifting away from expensive, specialized precursors to a commodity dye intermediate, this method achieves high purity and stable quality while drastically simplifying the operational requirements for industrial manufacturers. The strategic replacement of costly reagents with widely available chemicals like fluoroboric acid further underscores the commercial viability of this approach for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-6-fluoronaphthalene has been hindered by cumbersome multi-step routes that impose severe constraints on both equipment and budget. Traditional methodologies, such as those described in prior art literature, often necessitate a five-step sequence that includes a high-pressure ammonolysis reaction to introduce the amino group, a process that demands specialized autoclaves and rigorous safety protocols. Furthermore, these conventional pathways frequently rely on hexafluorophosphoric acid for the generation of diazonium salts, a reagent that is not only prohibitively expensive but also difficult to source in bulk quantities compared to standard mineral acids. The combination of high-pressure conditions, complex purification requirements, and the use of premium-priced fluorinating agents results in a manufacturing process with a significantly elevated cost basis and limited scalability for general chemical producers.
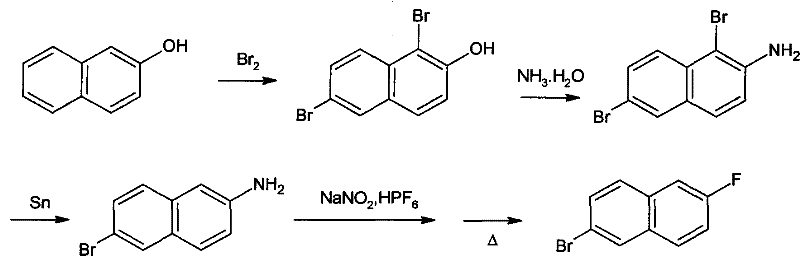
The Novel Approach
In stark contrast to the arduous traditional methods, the novel approach detailed in the patent leverages a clever bromination-debromination strategy starting from the abundant Tobias acid. This innovative route bypasses the need for high-pressure equipment entirely by utilizing a regioselective bromination in an acidic medium, followed by a selective reduction with metal powder to yield the key 6-bromo-2-naphthylamine intermediate. The subsequent conversion to the target molecule employs fluoroboric acid, a cheap and easily obtainable reagent, to form the diazonium salt which is then thermally decomposed to install the fluorine atom. This three-step sequence not only reduces the number of unit operations but also operates under mild atmospheric conditions, thereby lowering the barrier to entry for manufacturers and enabling a more agile response to market demand for this valuable intermediate.
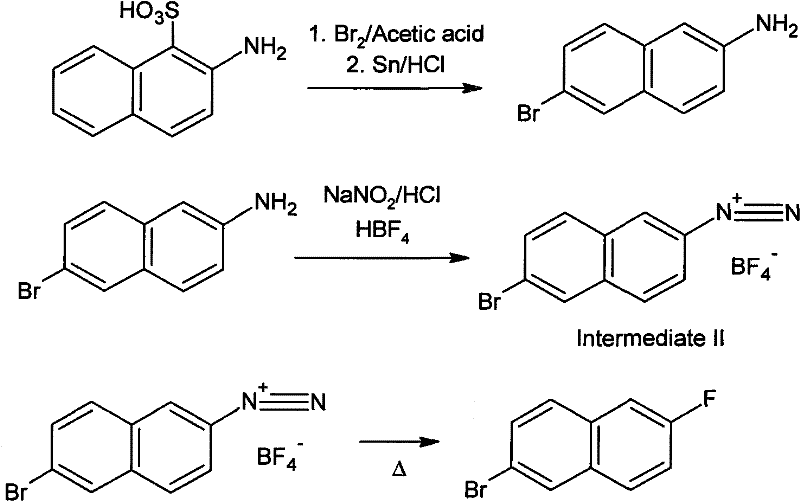
Mechanistic Insights into Bromination-Debromination and Diazotization
The core chemical ingenuity of this process lies in the initial transformation of Tobias acid, where the reaction conditions are meticulously tuned to achieve a specific substitution pattern on the naphthalene ring. When Tobias acid is treated with liquid bromine in an acetic acid medium, the sulfonic acid group at the 1-position is effectively replaced by a bromine atom, while a second bromination occurs selectively at the 6-position. This tandem substitution creates a 1,6-dibromo-2-naphthylamine intermediate in situ, which is then subjected to a controlled reduction using metal powders such as tin or iron. The reduction step is highly selective, removing the bromine atom at the 1-position while preserving the bromine at the 6-position, a critical selectivity that ensures the correct regiochemistry for the final product without requiring difficult chromatographic separations.
Following the formation of the amine intermediate, the process employs a classic Balz-Schiemann type reaction mechanism adapted for industrial efficiency using fluoroboric acid. The 6-bromo-2-naphthylamine undergoes diazotization in a strongly acidic medium at low temperatures to form the diazonium cation, which is immediately stabilized by the tetrafluoroborate anion. Upon heating, this stable salt undergoes thermal decomposition, releasing nitrogen gas and boron trifluoride while simultaneously forming the carbon-fluorine bond at the 2-position. The use of an inert high-boiling solvent during this thermal cracking step ensures smooth heat transfer and prevents localized overheating, which minimizes the formation of tar and other decomposition by-products, ultimately leading to a product with exceptional purity levels suitable for sensitive electronic or pharmaceutical applications.
How to Synthesize 2-Bromo-6-Fluoronaphthalene Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize yield and minimize waste generation throughout the three distinct stages. The initial bromination and reduction can be telescoped to some extent, allowing for the direct processing of the crude dibromo intermediate without isolation, which saves time and solvent usage. For the diazotization step, maintaining the reaction temperature below zero degrees Celsius is crucial to prevent premature decomposition of the diazonium species, while the final thermal cracking must be conducted within a narrow window of 130-150°C to ensure complete conversion without charring. Detailed standardized operating procedures regarding the specific addition rates of bromine and the precise pH control during salt formation are essential for reproducibility, and the full technical breakdown of these parameters is provided in the structured guide below for process engineers.
- Bromination of Tobias acid in acidic medium followed by selective reduction with metal powder to yield 6-bromo-2-naphthylamine.
- Diazotization of the amine intermediate using nitrous acid and fluoroboric acid to form the stable diazonium fluoroborate salt.
- Thermal decomposition of the diazonium salt in an inert medium at controlled temperatures to yield the final fluoro-product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this Tobias acid-based route represents a significant opportunity to optimize the cost structure and reliability of the supply chain for fluorinated naphthalene derivatives. By anchoring the synthesis on a commodity chemical that is mass-produced for the dye industry, the process decouples the supply of the intermediate from the volatile market of specialized fluorinated starting materials. This fundamental change in raw material sourcing mitigates the risk of supply disruptions and provides a stable pricing baseline that is less susceptible to the fluctuations often seen in the niche fine chemical sector. Additionally, the elimination of high-pressure processing steps reduces the capital expenditure required for manufacturing infrastructure, allowing for more flexible production scheduling and faster turnaround times for custom orders.
- Cost Reduction in Manufacturing: The substitution of expensive hexafluorophosphoric acid with widely available fluoroboric acid drives down the direct material costs significantly, as the latter is a basic chemical produced in vast quantities domestically. Although the yield of the thermal decomposition step with fluoroboric acid might be marginally lower than with phosphorus counterparts, the sheer price differential between the two acids results in a net positive impact on the overall cost of goods sold. Furthermore, the ability to telescope the bromination and reduction steps reduces solvent consumption and labor hours, contributing to a leaner and more cost-efficient manufacturing operation that enhances profit margins.
- Enhanced Supply Chain Reliability: Relying on Tobias acid as the primary feedstock ensures a robust supply chain because this material is produced by multiple manufacturers globally for the textile and dye sectors. This diversity of supply sources means that procurement teams are not dependent on a single vendor for critical starting materials, thereby reducing the risk of bottlenecks that could halt production lines. The simplified process flow also means that the manufacturing timeline is shorter, allowing for quicker replenishment of inventory and a more responsive capability to meet sudden spikes in demand from downstream pharmaceutical or electronic material clients.
- Scalability and Environmental Compliance: The absence of high-pressure reactions and the use of standard acidic media make this process inherently easier to scale from pilot plant to multi-ton commercial production without requiring exotic reactor designs. From an environmental perspective, the process generates fewer hazardous waste streams compared to routes involving heavy metal catalysts or aggressive fluorinating agents, simplifying wastewater treatment and disposal compliance. The use of inert solvents for the thermal cracking step allows for efficient solvent recovery and recycling, further minimizing the environmental footprint and aligning with modern green chemistry principles that are increasingly demanded by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational benefits and chemical rationale behind the chosen methodology for stakeholders evaluating the technology for potential adoption or partnership. Understanding these nuances is critical for assessing the feasibility of integrating this process into existing manufacturing portfolios.
Q: What are the key cost advantages of using Tobias acid as a starting material?
A: Tobias acid is a widely produced dye intermediate with established large-scale manufacturing, making it significantly cheaper and more readily available than specialized fluorinated naphthalene precursors used in traditional five-step routes.
Q: How does this method improve safety compared to prior art processes?
A: Unlike conventional methods that require high-pressure ammonolysis conditions, this novel route operates under atmospheric pressure and mild thermal conditions, eliminating the need for expensive high-pressure reactors and reducing operational hazards.
Q: Why is fluoroboric acid preferred over hexafluorophosphoric acid in this synthesis?
A: Fluoroboric acid is a basic chemical raw material with widespread domestic production and lower cost compared to the expensive and less accessible hexafluorophosphoric acid, providing a distinct economic advantage despite slightly lower yields in the scission step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-6-Fluoronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN101870636A for securing a competitive edge in the global market for pharmaceutical and electronic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and compliant with international standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-bromo-6-fluoronaphthalene meets the exacting requirements of our partners, whether they are developing next-generation liquid crystals or life-saving medications.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can be tailored to your specific volume and quality needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to delivering high-value chemical solutions.
