Advanced Synthesis of Brominated Azetidinones for Scalable Beta-Lactam Production
Advanced Synthesis of Brominated Azetidinones for Scalable Beta-Lactam Production
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex beta-lactam scaffolds, which serve as the critical backbone for a vast array of life-saving antibiotics. Patent CN1164531A introduces a sophisticated methodology for preparing novel derivatives of 3-bromo-and 3,3-dibromo-4-oxo-1-azetidines, addressing long-standing challenges in stereoselectivity and functional group tolerance. This technology leverages the unique reactivity of 2-sulfinic acid derivatives, specifically 2-substituted sulfinamides, to access highly functionalized azetidinone cores that were previously difficult to synthesize with high fidelity. By shifting the synthetic focus to these specialized precursors, the process enables the precise installation of halogen and alkoxy groups at the C-2 position, creating versatile intermediates for the next generation of carbapenems and penems. 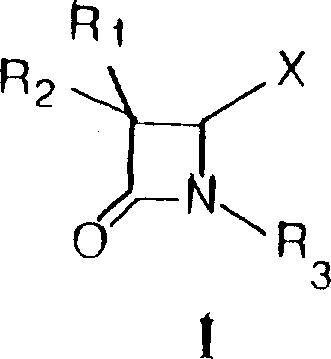 For R&D directors and process chemists, understanding the nuances of this transformation is vital for optimizing impurity profiles and ensuring regulatory compliance in API manufacturing. The ability to manipulate the oxidation state and substitution pattern of the azetidinone ring without compromising the integrity of the sensitive beta-lactam bond represents a significant leap forward in medicinal chemistry capabilities.
For R&D directors and process chemists, understanding the nuances of this transformation is vital for optimizing impurity profiles and ensuring regulatory compliance in API manufacturing. The ability to manipulate the oxidation state and substitution pattern of the azetidinone ring without compromising the integrity of the sensitive beta-lactam bond represents a significant leap forward in medicinal chemistry capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-halo azetidinones has relied heavily on the direct halogenation of penicillin sulfoxides or protected penicillanic acid derivatives, a approach fraught with significant operational and chemical drawbacks. Traditional methods often involve the use of aggressive halogenating agents like sulfuryl chloride or molecular chlorine under harsh conditions that can lead to unpredictable ring opening or epimerization at the C-5 and C-6 positions. Furthermore, the removal of excess halogenating agents and the resulting acidic byproducts typically requires extensive aqueous workups, generating substantial volumes of hazardous waste and complicating the isolation of the desired intermediate. These conventional routes frequently suffer from poor atom economy and limited substrate scope, making it difficult to introduce diverse functional groups at the C-2 position without resorting to multi-step protection and deprotection sequences. The reliance on unstable intermediates in older processes also poses risks for commercial scale-up, where reproducibility and safety are paramount concerns for supply chain stability.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes 2-sulfinamides of 4-oxo-azetidines as stable and versatile starting materials, offering a streamlined path to the target brominated derivatives. 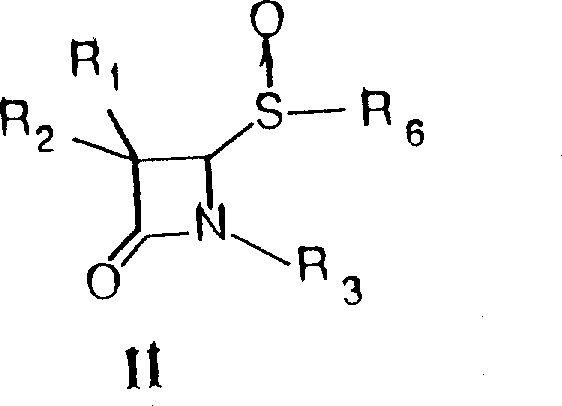 This innovative strategy involves the reaction of these sulfinamide precursors with various halogenating agents, including polymer-supported bromine, to generate 2-halo derivatives with exceptional control over the reaction outcome. The subsequent transformation of these halo-intermediates using silver tetrafluoroborate and alcohols allows for the mild and efficient introduction of alkoxy groups, bypassing the need for harsh acidic or basic conditions that could degrade the beta-lactam ring. This modular approach not only improves the overall yield and purity of the final products but also expands the chemical space accessible to medicinal chemists, enabling the rapid synthesis of libraries containing 2-alkoxy, 2-nitroxy, and 2-halo variants. By decoupling the halogenation step from the sensitive beta-lactam formation, this process provides a safer and more reliable foundation for the industrial production of complex antibiotic intermediates.
This innovative strategy involves the reaction of these sulfinamide precursors with various halogenating agents, including polymer-supported bromine, to generate 2-halo derivatives with exceptional control over the reaction outcome. The subsequent transformation of these halo-intermediates using silver tetrafluoroborate and alcohols allows for the mild and efficient introduction of alkoxy groups, bypassing the need for harsh acidic or basic conditions that could degrade the beta-lactam ring. This modular approach not only improves the overall yield and purity of the final products but also expands the chemical space accessible to medicinal chemists, enabling the rapid synthesis of libraries containing 2-alkoxy, 2-nitroxy, and 2-halo variants. By decoupling the halogenation step from the sensitive beta-lactam formation, this process provides a safer and more reliable foundation for the industrial production of complex antibiotic intermediates.
Mechanistic Insights into Silver-Mediated Substitution and Halogenation
The core of this synthetic breakthrough lies in the unique reactivity of the 2-sulfinamide moiety, which acts as a latent leaving group facilitating the introduction of halogens at the C-2 position. When the sulfinamide precursor is treated with a halogenating agent such as bromine or N-bromosuccinimide, the sulfur-nitrogen bond is cleaved, and a halogen atom is installed with high regioselectivity. This transformation likely proceeds through a transient sulfenyl halide intermediate that undergoes rearrangement or direct substitution to yield the 2-halo-3-bromo-azetidinone scaffold. The use of polymer-supported bromine further refines this mechanism by providing a controlled release of electrophilic bromine, minimizing over-halogenation and side reactions that often plague solution-phase halogenations. This level of control is critical for maintaining the stereochemical integrity of the azetidinone ring, ensuring that the resulting intermediates possess the correct configuration for downstream biological activity.
Following the initial halogenation, the conversion of the 2-halo derivative into 2-alkoxy or 2-nitroxy species is mediated by silver salts, a step that exemplifies the power of soft Lewis acid catalysis in organic synthesis. The silver cation coordinates with the halogen atom at the C-2 position, weakening the carbon-halogen bond and facilitating its departure as a silver halide precipitate. This generates a highly reactive carbocation or ion-pair intermediate that is immediately trapped by the nucleophilic solvent, such as methanol or isopropanol, to form the corresponding ether.  Alternatively, the use of silver nitrate introduces a nitroxy group, expanding the utility of the intermediate for further derivatization. The precipitation of silver halide drives the equilibrium towards the product, ensuring high conversion rates even with sterically hindered substrates. Finally, the removal of protecting groups, such as the benzyl ester, is achieved using aluminum chloride and anisole, a Lewis acid-mediated deprotection that is mild enough to preserve the sensitive functionality of the molecule while efficiently revealing the free carboxylic acid required for peptide coupling in antibiotic synthesis.
Alternatively, the use of silver nitrate introduces a nitroxy group, expanding the utility of the intermediate for further derivatization. The precipitation of silver halide drives the equilibrium towards the product, ensuring high conversion rates even with sterically hindered substrates. Finally, the removal of protecting groups, such as the benzyl ester, is achieved using aluminum chloride and anisole, a Lewis acid-mediated deprotection that is mild enough to preserve the sensitive functionality of the molecule while efficiently revealing the free carboxylic acid required for peptide coupling in antibiotic synthesis.
How to Synthesize 3-Bromo-4-Oxo-1-Azetidine Derivatives Efficiently
The practical execution of this synthesis requires careful attention to reaction conditions and reagent quality to maximize yield and minimize impurities. The process begins with the dissolution of the 2-sulfinamide precursor in a suitable organic solvent like chloroform or dichloromethane, followed by the controlled addition of the halogenating agent. Maintaining the reaction temperature within the specified range is crucial to prevent thermal degradation of the azetidinone ring. After the halogenation is complete, the reaction mixture is processed to isolate the 2-halo intermediate, which serves as the pivotal branch point for generating diverse derivatives. For those seeking to implement this chemistry in a GMP environment, the detailed standardized operating procedures regarding reagent stoichiometry, mixing times, and purification techniques are essential for consistent batch-to-batch performance.
- React 2-substituted sulfinamides of 4-oxo-azetidines with halogenating agents such as bromine or N-bromosuccinimide to form 2-halo derivatives.
- Treat the isolated 2-halo intermediates with silver tetrafluoroborate and alcohols to generate 2-alkoxy derivatives, or use silver nitrate for 2-nitroxy variants.
- Perform deprotection using aluminum chloride and anisole to obtain the final free carboxy group derivatives suitable for further coupling.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers compelling advantages that directly translate into operational efficiency and cost optimization. The shift towards using polymer-supported reagents for the halogenation step significantly simplifies the downstream processing workflow, as the spent reagent can be removed via simple filtration rather than complex aqueous extractions. This reduction in unit operations not only shortens the overall cycle time but also drastically lowers the consumption of solvents and water, aligning with modern sustainability goals and reducing the burden on waste treatment facilities. Furthermore, the enhanced stability of the sulfinamide starting materials compared to traditional penicillin sulfoxides improves inventory management and reduces the risk of raw material degradation during storage and transport.
- Cost Reduction in Manufacturing: The streamlined workflow inherent in this process eliminates several costly purification steps that are typically required to remove inorganic salts and acidic byproducts in conventional methods. By utilizing silver-mediated substitution, the reaction drives to completion through the precipitation of insoluble silver salts, which simplifies product isolation and reduces the need for extensive chromatographic purification. This efficiency gain leads to substantial cost savings in terms of labor, energy, and consumables, making the production of high-value beta-lactam intermediates more economically viable. Additionally, the higher purity of the crude product reduces the load on final crystallization steps, further enhancing the overall process economics without compromising quality standards.
- Enhanced Supply Chain Reliability: The versatility of this synthetic platform allows for the use of widely available and commercially stable raw materials, mitigating the risk of supply disruptions associated with specialized or unstable reagents. The ability to produce a wide range of derivatives from a common intermediate creates a flexible manufacturing asset that can be rapidly repurposed to meet fluctuating market demands for different antibiotic classes. This adaptability ensures a continuous and reliable supply of critical intermediates, safeguarding the production schedules of downstream API manufacturers against raw material shortages. Moreover, the robustness of the reaction conditions minimizes the likelihood of batch failures, contributing to a more predictable and dependable supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that can be easily translated from laboratory benchtop to multi-ton commercial production without significant re-engineering. The reduced generation of hazardous waste, particularly through the avoidance of liquid bromine and the minimization of aqueous effluents, simplifies regulatory compliance and lowers the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the operation against tightening environmental regulations but also enhances the corporate social responsibility profile of the supply chain partners involved. The safe handling of reagents and the simplified workup procedures contribute to a safer working environment, reducing occupational health risks and associated liabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing manufacturing portfolios.
Q: What are the primary advantages of using sulfinamide precursors in azetidinone synthesis?
A: Using 2-sulfinamide precursors allows for a more controlled halogenation process compared to direct penicillin modification. This route minimizes side reactions and facilitates the introduction of diverse functional groups at the C-2 position, leading to higher purity intermediates essential for sensitive beta-lactam antibiotic production.
Q: How does the polymer-supported bromine method impact process safety?
A: The utilization of polymer-supported bromine significantly enhances process safety by reducing the handling of free liquid bromine. This solid-phase reagent simplifies the workup procedure through simple filtration, thereby lowering the risk of exposure and reducing the complexity of waste treatment protocols in large-scale manufacturing environments.
Q: Can this synthetic route be adapted for different C-2 substituents?
A: Yes, the methodology is highly versatile. By varying the nucleophile in the silver-mediated step, manufacturers can access 2-alkoxy, 2-nitroxy, or 2-halo derivatives. This flexibility supports the rapid development of diverse beta-lactam analogues, including penems and monobactams, without requiring entirely new synthetic pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-4-Oxo-1-Azetidine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of next-generation antibiotics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate chemistry described in patent CN1164531A can be executed with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch of 3-bromo-and 3,3-dibromo-4-oxo-1-azetidine derivatives we supply adheres to the highest industry standards, providing our partners with the confidence they need to advance their drug development programs.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage these advanced synthetic capabilities for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how our optimized manufacturing processes can enhance your supply chain resilience and reduce your overall cost of goods sold. Let us be your strategic partner in navigating the complexities of beta-lactam intermediate synthesis.
