Scalable Synthesis of 4,4'-Bis(2,4-Dinitrophenoxy)Diphenylmethane for Advanced Polyimide Applications
The development of advanced functional materials for the electronics and aerospace sectors relies heavily on the availability of high-performance polymer precursors. Patent CN101234990B introduces a highly efficient and industrially viable method for the preparation of 4,4'-bis(2,4-dinitrophenoxy)diphenylmethane, a critical intermediate in the synthesis of aromatic polyimides. This compound serves as a foundational building block for creating highly branched polyimide systems that exhibit superior thermal stability, mechanical strength, and electrical insulation properties required for next-generation LCD displays and microelectronic applications. The disclosed methodology addresses significant challenges in traditional synthetic routes by optimizing reaction conditions to achieve exceptional yields and purity without the need for complex catalytic systems or extreme pressures. By leveraging a straightforward nucleophilic aromatic substitution strategy, this process offers a robust pathway for manufacturers seeking to enhance their supply chain reliability for high-value electronic chemicals.
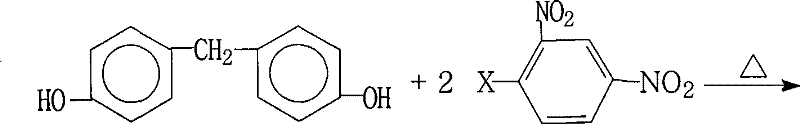
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing complex aromatic ethers often suffer from significant inefficiencies that hinder large-scale commercial adoption. Many conventional methods rely on the use of expensive phase-transfer catalysts or require stringent anhydrous conditions that are difficult to maintain in large reactor volumes, leading to inconsistent batch quality and increased operational costs. Furthermore, older processes frequently utilize single-solvent systems that do not facilitate the easy removal of reaction by-products, such as water generated during etherification, which can shift the chemical equilibrium backward and limit overall conversion rates. The reliance on corrosive reagents or the generation of substantial amounts of hazardous waste in these legacy methods also poses severe environmental compliance challenges and increases the burden on waste treatment facilities. Additionally, the difficulty in recovering and recycling solvents from these traditional one-pot reactions often results in higher raw material consumption and a larger carbon footprint for the manufacturing process.
The Novel Approach
The innovative approach detailed in the patent overcomes these historical barriers through a cleverly designed mixed-solvent system and optimized stoichiometry. By employing a combination of water-insoluble organic solvents, such as toluene or chlorobenzene, with strongly polar aprotic solvents like N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP), the process enables efficient azeotropic removal of water during the reflux stage. This continuous water separation drives the reaction equilibrium towards the product side, ensuring high conversion rates without the need for excessive reagent loading. The method operates under atmospheric pressure and moderate temperatures ranging from 80°C to 200°C, eliminating the need for specialized high-pressure equipment and reducing energy consumption significantly. Moreover, the simplicity of the workup procedure, which involves concentration, cooling, and precipitation with water, allows for the direct recovery of high-purity crystals while facilitating the recycling of the organic solvent phase for subsequent batches, thereby drastically reducing waste generation.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis is a classic nucleophilic aromatic substitution (SNAr) reaction, driven by the strong electron-withdrawing nature of the nitro groups on the benzene ring. In this mechanism, the phenoxide anion, generated in situ by the deprotonation of 4,4'-dihydroxydiphenylmethane by the salt-forming agent (such as potassium carbonate or sodium hydroxide), acts as a potent nucleophile. This nucleophile attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene at the position ortho or para to the nitro groups, where the electron density is lowest. The presence of two nitro groups significantly activates the ring towards nucleophilic attack, stabilizing the negatively charged Meisenheimer complex intermediate that forms during the rate-determining step. The subsequent elimination of the halide leaving group restores the aromaticity of the ring, resulting in the formation of the stable diphenyl ether linkage. The choice of halogen (fluorine, chlorine, bromine, or iodine) influences the reaction kinetics, with fluorine often providing faster reaction rates due to its high electronegativity enhancing the electrophilicity of the ring carbon, despite the stronger C-F bond.
Controlling impurities in this reaction is critical for downstream polyimide performance, and the patented process achieves this through precise control of reaction parameters and stoichiometry. By maintaining a molar ratio of 4,4'-dihydroxydiphenylmethane to 2,4-dinitrohalobenzene between 1.0:2.0 and 1.0:2.2, the process ensures that the diphenol is fully consumed, minimizing the presence of mono-substituted intermediates that could act as chain terminators in subsequent polymerization steps. The use of a mixed solvent system not only aids in water removal but also helps in solubilizing the ionic intermediates and the inorganic salts formed, preventing them from coating the reactants and inhibiting the reaction progress. Furthermore, the final purification step involving washing with hot water effectively removes residual inorganic salts and unreacted polar species, yielding a product with purity levels consistently above 99.0%. This high level of chemical purity is essential for ensuring the thermal and mechanical integrity of the final polyimide films used in sensitive electronic applications.
How to Synthesize 4,4'-Bis(2,4-Dinitrophenoxy)Diphenylmethane Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory benchtop to industrial production scales. The process begins with the careful selection of reagents, specifically choosing the appropriate halogenated benzene derivative and base to match the desired reaction kinetics and cost profile. Operators must ensure that the solvent mixture is prepared with the correct volume ratios to optimize both solubility and azeotropic distillation efficiency. The reaction is then initiated by heating the mixture to reflux, with continuous monitoring of water separation to gauge reaction progress.
- Combine 4,4'-dihydroxydiphenylmethane and 2,4-dinitrohalobenzene (molar ratio 1.0: 2.0-2.2) with a salt-forming agent in a mixture of water-insoluble and polar aprotic solvents.
- Heat the reaction mixture to reflux (80°C-200°C) with continuous water separation for 3 to 18 hours to drive the nucleophilic substitution to completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The process is designed with industrial scalability in mind, utilizing readily available commodity chemicals that are sourced from stable global supply chains, thereby mitigating the risk of raw material shortages. The ability to operate under atmospheric pressure reduces the capital expenditure required for reactor infrastructure, as there is no need for expensive high-pressure vessels or complex safety systems associated with hazardous gas handling. Furthermore, the environmental profile of the process is significantly improved compared to traditional methods, aligning with increasingly stringent global regulations on industrial emissions and waste disposal. This alignment reduces the regulatory burden on manufacturing sites and minimizes the potential for production stoppages due to compliance issues.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven primarily by the efficiency of the solvent system and the simplicity of the workup. By enabling the recovery and repeated reuse of the organic solvent phase, the process drastically lowers the recurring cost of raw materials, which is often a major component of the total cost of goods sold in fine chemical manufacturing. The elimination of transition metal catalysts or exotic reagents further reduces material costs and removes the need for expensive metal scavenging steps that are typically required to meet purity specifications in electronic grade chemicals. Additionally, the high conversion rates achieved mean that less unreacted starting material needs to be separated and disposed of, reducing waste treatment costs and maximizing the utility of every kilogram of input material.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain stability by reducing the complexity of the manufacturing process. Since the reaction does not rely on moisture-sensitive catalysts or inert atmosphere conditions, it is less susceptible to variations in operational conditions that can lead to batch failures. The use of common solvents like toluene and DMF ensures that supply disruptions are unlikely, as these chemicals are produced in massive volumes globally. The short reaction times observed in certain embodiments, combined with the straightforward isolation procedure, allow for faster turnaround times between batches, enabling manufacturers to respond more agilely to fluctuations in market demand and reducing the lead time for delivering high-purity intermediates to customers.
- Scalability and Environmental Compliance: Scaling this process to multi-ton production is facilitated by the absence of exothermic hazards or gas evolution that typically complicate scale-up efforts. The reaction proceeds smoothly under reflux conditions, which are easily managed in large-scale reactors using standard heating and condensation equipment. From an environmental perspective, the significant reduction in three wastes (wastewater, waste gas, and solid waste) simplifies the permitting process for new production lines and lowers the ongoing operational costs associated with environmental management. The ability to recycle solvents not only conserves resources but also minimizes the volume of volatile organic compounds (VOCs) released into the atmosphere, supporting corporate sustainability goals and enhancing the company's reputation as a responsible manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-bis(2,4-dinitrophenoxy)diphenylmethane. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific polymer formulation or supply chain strategy.
Q: Which halogenated benzene derivatives are suitable for this synthesis?
A: The patent specifies that 2,4-dinitrohalobenzene where the halogen is fluorine, chlorine, bromine, or iodine can be effectively utilized, with 2,4-dinitrochlorobenzene and 2,4-dinitrofluorobenzene showing particularly high yields.
Q: How does the solvent system contribute to process efficiency?
A: The use of a mixed solvent system comprising water-insoluble solvents like toluene or chlorobenzene and polar aprotic solvents like DMF or NMP facilitates effective water removal via azeotropic distillation, driving the equilibrium forward and allowing for solvent recovery and recycling.
Q: What purity levels can be achieved with this method?
A: Experimental data within the patent demonstrates that this method consistently achieves product purity levels exceeding 99.0%, with specific examples reaching up to 99.6% purity after simple washing and drying procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(2,4-Dinitrophenoxy)Diphenylmethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of advanced electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4,4'-bis(2,4-dinitrophenoxy)diphenylmethane meets the exacting standards required for polyimide synthesis. Our facility is equipped to handle the specific solvent systems and reaction conditions described in the patent, allowing us to deliver a product that is optimized for performance and cost-efficiency.
We invite you to collaborate with us to optimize your supply chain for polyimide monomers. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific production needs, identifying opportunities to reduce costs without compromising on quality. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our solutions align perfectly with your project timelines and performance targets. Let us be your partner in driving innovation and efficiency in the electronic chemicals sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
