Scalable Synthesis of 3,5-Bis(2,4-Dinitrophenoxy)Toluene for High-Performance Polyimide Manufacturing
The rapid advancement of the optoelectronics and aerospace sectors has placed unprecedented demand on high-performance polymers, specifically aromatic polyimides known for their exceptional thermal stability and mechanical strength. Patent CN101234979B introduces a groundbreaking methodology for synthesizing 3,5-bis(2,4-dinitrophenoxy)toluene, a pivotal intermediate in the production of highly branched polyimide resins. This compound acts as a precursor to aromatic polyvalent primary amines, which are indispensable for creating next-generation liquid crystal display (LCD) materials and advanced composite structures. The disclosed process addresses critical bottlenecks in traditional synthesis by optimizing reaction conditions to achieve superior purity and yield without compromising environmental standards. By leveraging a precise nucleophilic aromatic substitution strategy, this technology offers a reliable pathway for manufacturers seeking to secure a stable supply of high-purity electronic chemical intermediates. The implications for the supply chain are profound, as the method simplifies operations while ensuring the consistent quality required for sensitive microelectronic applications.
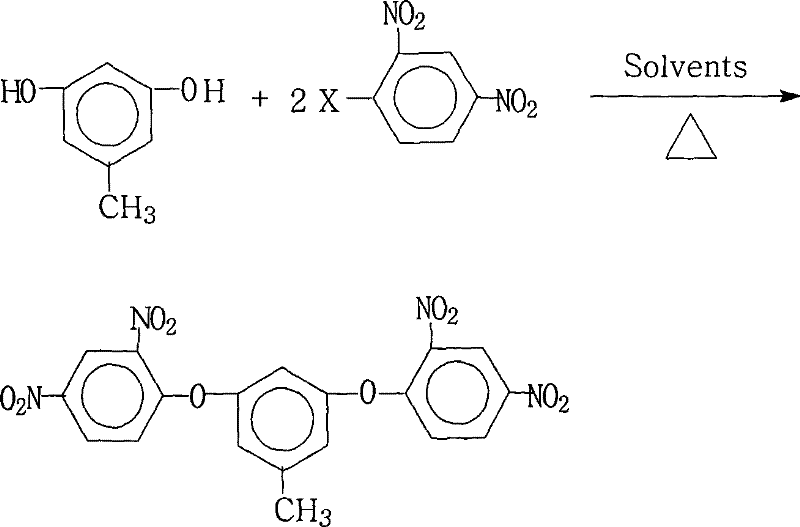
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex ether-linked aromatic compounds has been plagued by inefficient reaction kinetics and difficult purification protocols that escalate production costs. Traditional routes often suffer from incomplete conversion due to the poor solubility of phenolic substrates in standard organic media, leading to significant amounts of unreacted starting materials that contaminate the final product. Furthermore, many legacy processes rely on harsh reaction conditions or expensive phase-transfer catalysts that introduce metallic impurities, which are strictly prohibited in high-end electronic grade materials. The inability to effectively remove water generated during the etherification process often drives the equilibrium backward, resulting in suboptimal yields that necessitate energy-intensive recrystallization steps. These inefficiencies not only inflate the cost of goods sold but also create substantial waste streams that complicate regulatory compliance and environmental management. Consequently, procurement teams have long struggled to find suppliers capable of delivering this intermediate with the consistency required for mass production of polyimide films.
The Novel Approach
The methodology outlined in CN101234979B represents a paradigm shift by employing a sophisticated mixed solvent system combined with azeotropic water removal to drive the reaction to near-completion. By utilizing a specific molar ratio of 3,5-dihydroxytoluene to 2,4-dinitrohalobenzene between 1.0:2.0 and 1.0:2.2, the process ensures that the dihydroxy substrate is fully consumed, minimizing the formation of mono-substituted byproducts. The innovation lies in the dual-solvent strategy, which combines a water-insoluble organic solvent like toluene with a highly polar aprotic solvent such as N,N-dimethylformamide (DMF). This unique combination enhances the solubility of both the phenolic salt and the nitro-halide reactant, facilitating a homogeneous reaction environment that significantly accelerates the nucleophilic attack. Additionally, the continuous removal of water during the reflux period prevents hydrolysis of the sensitive nitro groups and pushes the equilibrium forward, resulting in isolated yields as high as 94.2% with purity levels exceeding 99.4%. This approach eliminates the need for complex chromatographic purification, making it ideally suited for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Nucleophilic Aromatic Substitution (SnAr)
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr), facilitated by the strong electron-withdrawing nature of the nitro groups positioned ortho and para to the leaving halide group. In this mechanism, the phenoxide anion, generated in situ by the deprotonation of 3,5-dihydroxytoluene using bases like potassium carbonate or sodium hydroxide, acts as a potent nucleophile. The presence of two nitro groups on the benzene ring significantly lowers the energy of the lowest unoccupied molecular orbital (LUMO), thereby stabilizing the negatively charged Meisenheimer complex intermediate formed upon nucleophilic attack. This stabilization is crucial for overcoming the high activation energy typically associated with displacing halides from aromatic rings under mild conditions. The choice of halide (fluorine, chlorine, bromine, or iodine) allows for fine-tuning of the reaction rate, with fluorine often providing superior reactivity in SnAr reactions due to its high electronegativity despite being a poorer leaving group in aliphatic systems. Understanding this electronic activation is key for R&D directors aiming to replicate or optimize the process for different scale-up scenarios.
Impurity control in this system is meticulously managed through the stoichiometric excess of the nitro-halide reactant and the rigorous exclusion of moisture prior to the addition of the base. The formation of side products, such as mono-etherified species or hydrolyzed phenols, is minimized by maintaining the reaction temperature between 80°C and 200°C, which provides sufficient thermal energy for the second substitution step to occur rapidly. The use of a mixed solvent system also plays a critical role in impurity profiling; the polar component solvates the ionic transition states effectively, while the non-polar component aids in the azeotropic removal of water, preventing the reverse reaction. Post-reaction workup involves simple precipitation by adding water to the concentrated reaction mass, a technique that exploits the low solubility of the highly crystalline dinitro product in aqueous media. This physical separation method ensures that soluble inorganic salts and residual polar solvents remain in the mother liquor, yielding a crude product that requires minimal washing to achieve pharmaceutical or electronic grade purity specifications.
How to Synthesize 3,5-Bis(2,4-Dinitrophenoxy)Toluene Efficiently
Implementing this synthesis protocol requires careful attention to the order of addition and the efficiency of the water separation apparatus to maximize the thermodynamic drive of the reaction. The process begins by charging the reactor with the dihydroxytoluene, the nitro-halide, and the base within the pre-mixed solvent system, followed by heating to reflux with a Dean-Stark trap or similar device to continuously remove generated water. Detailed operational parameters, including specific solvent ratios and temperature ramps, are critical for reproducing the high yields reported in the patent examples. For a comprehensive guide on the exact step-by-step execution, including safety precautions and equipment specifications, please refer to the standardized synthesis protocol below.
- Charge 3,5-dihydroxytoluene and 2,4-dinitrohalobenzene (molar ratio 1: 2.0-2.2) with a base like potassium carbonate into a reactor containing a mixed solvent system.
- Heat the mixture to reflux (80°C-200°C) with continuous water separation for 3 to 18 hours to drive the nucleophilic substitution to completion.
- Concentrate the reaction solution, cool, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented method offers distinct advantages that directly address the pain points of cost volatility and supply continuity in the specialty chemicals market. The ability to operate at atmospheric pressure without the need for specialized high-pressure reactors significantly lowers capital expenditure requirements for manufacturing facilities, allowing for more flexible production scheduling. Furthermore, the simplicity of the workup procedure, which relies on precipitation and filtration rather than complex distillation or chromatography, drastically reduces the cycle time per batch. This efficiency translates into a higher throughput capacity, enabling suppliers to respond more agilely to fluctuating market demands for polyimide precursors. The robustness of the process also意味着 a lower risk of batch failure, ensuring a consistent flow of material to downstream polymerization units.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of expensive transition metal catalysts and the use of readily available commodity chemicals as starting materials. By avoiding precious metals, the process removes the need for costly metal scavenging steps and extensive analytical testing for residual heavy metals, which are mandatory for electronic grade materials. Additionally, the solvent system is designed for easy recovery; the organic solvents can be distilled and reused multiple times without significant loss of performance, leading to substantial savings in raw material consumption. The high yield achieved minimizes the waste of valuable nitro-aromatic starting materials, further optimizing the overall cost structure and enhancing the margin potential for large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on common industrial solvents like toluene and DMF, along with bulk chemicals like potassium carbonate, ensures that the supply chain is not vulnerable to the shortages often associated with exotic reagents. The process tolerance for various halides (chloro, bromo, fluoro) provides procurement managers with the flexibility to switch raw material sources based on availability and price fluctuations without altering the core process parameters. This adaptability is crucial for maintaining uninterrupted production schedules in a global market where logistics disruptions can occur unexpectedly. Moreover, the stability of the intermediate product allows for safer storage and transportation, reducing the risks associated with hazardous material handling and expanding the potential geographic reach of the supply network.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial tonnage is straightforward due to the absence of exothermic hazards that typically plague nitration or reduction chemistries. The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions that can be treated using standard wastewater management protocols, aligning with increasingly stringent environmental regulations. The ability to recycle solvents not only reduces the carbon footprint of the manufacturing process but also lowers the volume of volatile organic compound (VOC) emissions. This environmental friendliness makes the technology attractive for production in regions with strict ecological mandates, ensuring long-term operational sustainability and reducing the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this material into your specific polymer formulation or supply chain strategy.
Q: What is the primary application of 3,5-bis(2,4-dinitrophenoxy)toluene?
A: It serves as a critical intermediate for synthesizing highly branched aromatic polyimide monomers, which are essential for high-performance LCD materials and aerospace applications due to their thermal stability.
Q: How does this patent method improve yield compared to traditional routes?
A: By utilizing a specific mixed solvent system and controlled water separation during reflux, the method achieves yields up to 94.2% with purity exceeding 99%, significantly reducing downstream purification costs.
Q: Is the solvent system recyclable in this process?
A: Yes, the process utilizes organic solvents that are easily recovered and recycled after concentration, minimizing waste generation and enhancing the environmental profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Bis(2,4-Dinitrophenoxy)Toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final polyimide product depends entirely on the purity and consistency of the monomers used in its synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from R&D to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling via HPLC and NMR to meet the exacting standards of the electronic materials industry. We are committed to delivering high-purity 3,5-bis(2,4-dinitrophenoxy)toluene that empowers your team to develop superior LCD and aerospace materials with confidence.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing process can reduce your overall material costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency and technical excellence.
