Advanced Synthesis of High-Performance Polyimide Intermediates for Electronic Applications
Advanced Synthesis of High-Performance Polyimide Intermediates for Electronic Applications
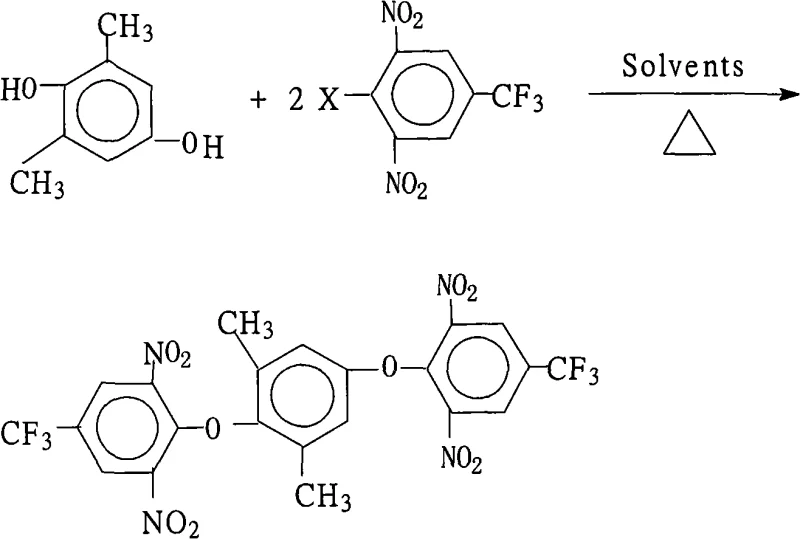
The rapid evolution of the electronic materials sector demands intermediates that offer exceptional thermal stability and mechanical strength, particularly for applications in liquid crystal displays (LCDs) and aerospace components. Patent CN101293839A introduces a groundbreaking preparation method for 1,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)-2,6-dimethylbenzene, a pivotal precursor in the synthesis of highly branched aromatic polyimides. This technology addresses the critical industry need for reliable electronic chemical supplier solutions by providing a route that ensures high purity and yield while maintaining environmental compliance. The strategic importance of this molecule lies in its ability to form polyimide systems with superior temperature resistance and comprehensive performance metrics, making it indispensable for next-generation functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic ethers required for high-performance polyimides has been plagued by significant technical and economic hurdles. Traditional pathways often necessitate harsh reaction conditions, including high-pressure environments or the use of expensive transition metal catalysts that are difficult to remove from the final product. These legacy methods frequently result in inconsistent impurity profiles, which can severely compromise the dielectric properties and thermal stability of the resulting polymer. Furthermore, conventional processes often generate substantial amounts of hazardous waste and require energy-intensive purification steps, such as multiple recrystallizations or column chromatography, which drastically inflate the cost of goods sold (COGS) and extend lead times for high-purity OLED material and related electronic chemical precursors.
The Novel Approach
The methodology disclosed in CN101293839A represents a paradigm shift by leveraging a highly efficient nucleophilic aromatic substitution strategy that operates under mild, atmospheric conditions. By utilizing a specific molar ratio of 2,6-dimethylhydroquinone to 2,6-dinitro-4-trifluoromethyl halobenzene (ranging from 1.0:2.0 to 2.2), the process maximizes conversion efficiency without requiring exotic catalysts. The innovation lies in the clever selection of a mixed solvent system comprising water-insoluble organic solvents and strong polar aprotic solvents, which facilitates the reaction kinetics while allowing for straightforward product isolation. This approach not only simplifies the operational workflow but also enables the direct precipitation of the product upon water addition, eliminating the need for complex distillation or extraction procedures typically associated with cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic yet optimized Nucleophilic Aromatic Substitution (SnAr) reaction, where the electron-deficient nature of the halobenzene ring is exploited to facilitate ether bond formation. The presence of two strongly electron-withdrawing nitro groups (-NO2) at the ortho positions and a trifluoromethyl group (-CF3) at the para position significantly activates the aromatic ring towards nucleophilic attack by stabilizing the Meisenheimer complex intermediate. This electronic activation allows the phenoxide anion, generated in situ from 2,6-dimethylhydroquinone by the salt-forming agent, to displace the halogen atom (F, Cl, Br, or I) with remarkable selectivity and speed. The reaction temperature range of 80°C to 200°C provides sufficient thermal energy to overcome the activation barrier while the reflux conditions ensure consistent kinetic progression over the 6 to 18-hour duration.
From an impurity control perspective, the mechanism inherently favors the formation of the desired di-ether product due to the stoichiometric excess of the electrophilic halobenzene species. The use of a base such as potassium carbonate or sodium hydroxide ensures complete deprotonation of the hydroquinone hydroxyl groups, preventing side reactions associated with partial substitution. Furthermore, the choice of solvent plays a dual role: the polar aprotic component (e.g., DMF, NMP, DMSO) solvates the cation and enhances the nucleophilicity of the phenoxide, while the water-insoluble component (e.g., toluene, benzene) facilitates the azeotropic removal of water formed during the salt formation or present in the system. This precise control over the reaction environment minimizes the formation of oligomeric byproducts or hydrolysis derivatives, ensuring that the final crystalline product meets the stringent purity specifications required for commercial scale-up of complex polymer additives.
How to Synthesize 1,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)-2,6-dimethylbenzene Efficiently
Implementing this synthesis route requires careful attention to the mixing order and thermal management to ensure optimal yield and safety. The process begins with the charging of the hydroquinone derivative, the halobenzene electrophile, and the inorganic base into a reactor containing the pre-mixed solvent system. The mixture is then subjected to heating under reflux with a water separator to drive the equilibrium forward by removing generated water, a critical step for maximizing conversion. Following the reaction period, the workup procedure is notably streamlined, involving concentration of the liquor followed by the addition of water to induce crystallization, a technique that leverages the differential solubility of the product to achieve high purity without extensive downstream processing. For detailed standardized operating procedures and specific parameter optimization, please refer to the technical guide below.
- Mix 2,6-dimethylhydroquinone and 2,6-dinitro-4-trifluoromethyl halobenzene (molar ratio 1.0: 2.0-2.2) with a salt-forming agent in a mixed organic solvent system.
- Heat the mixture to reflux (80°C-200°C) with water separation for 6 to 18 hours to drive the etherification reaction to completion.
- Concentrate the reaction solution, cool, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of high-pressure reactors and corrosive reagents significantly lowers the capital expenditure (CAPEX) required for plant setup, as standard glass-lined or stainless steel vessels are sufficient for production. Moreover, the simplicity of the workup procedure—specifically the water precipitation step—drastically reduces energy consumption compared to traditional solvent-intensive purification methods, leading to substantial operational cost savings. The ability to recover and recycle the organic solvent system repeatedly further enhances the economic viability of the process, reducing the dependency on volatile raw material markets and minimizing waste disposal fees.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts and complex purification columns, relying instead on inexpensive inorganic bases and readily available solvents. The high atom economy and the ability to recycle solvents multiple times without loss of efficiency contribute to a leaner manufacturing cost structure, allowing for more competitive pricing in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as 2,6-dimethylhydroquinone and common halobenzenes, the supply chain is insulated from the bottlenecks often associated with exotic or proprietary reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent ratios, ensures consistent batch-to-batch quality, thereby reducing lead time for high-purity electronic chemicals and guaranteeing uninterrupted supply for downstream polymer manufacturers.
- Scalability and Environmental Compliance: The generation of minimal three-waste (wastewater, waste gas, waste residue) aligns perfectly with increasingly stringent global environmental regulations, reducing the regulatory burden on manufacturing sites. The straightforward scalability from laboratory to multi-ton production is facilitated by the absence of exothermic runaways or hazardous intermediates, making it an ideal candidate for rapid capacity expansion to meet growing demand in the electronics sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate, derived directly from the patent's experimental data and beneficial effects. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this monomer into new polyimide formulations or for procurement teams assessing supplier capabilities. The answers reflect the proven stability and efficiency of the described synthesis route.
Q: What are the primary advantages of this synthesis method for industrial scale-up?
A: The process operates at atmospheric pressure without corrosive byproducts, utilizes recyclable solvents, and achieves high purity (>99%) through simple water precipitation, significantly reducing equipment investment and waste treatment costs.
Q: Can the solvents used in this reaction be recovered and reused?
A: Yes, the patent explicitly highlights that the organic solvent system allows for convenient recovery and repeated cycling, which minimizes raw material consumption and environmental impact.
Q: What is the specific application of this chemical intermediate?
A: This compound serves as a critical precursor for synthesizing highly branched aromatic polyimide monomers, which are essential for manufacturing high-performance materials used in LCDs, aerospace, and microelectronics due to their thermal stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)-2,6-dimethylbenzene Supplier
As the demand for high-performance polyimides continues to surge in the semiconductor and display industries, securing a stable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 1,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)-2,6-dimethylbenzene meets the exacting standards required for advanced electronic applications. We combine technical expertise with a commitment to quality to deliver materials that empower your innovation.
We invite you to engage with our technical team to discuss how this optimized synthesis route can be tailored to your specific volume requirements. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments that demonstrate how we can become your strategic partner in the supply of critical electronic chemical intermediates.
