Advanced Synthesis of 2,4-Bis(2,4-Dinitrophenoxy)Toluene for High-Performance Polyimide Manufacturing
The global demand for high-performance polymeric materials in the electronics and aerospace sectors has necessitated the development of robust synthetic routes for specialized monomers. Patent CN101245013A introduces a groundbreaking preparation method for 2,4-bis(2,4-dinitrophenoxy)toluene, a pivotal intermediate in the production of highly branched aromatic polyimides. This compound is not merely a chemical building block but a cornerstone for next-generation liquid crystal displays (LCDs) and heat-resistant functional materials that require exceptional thermal stability and mechanical strength. The patented process distinguishes itself by optimizing the nucleophilic substitution reaction through a sophisticated dual-solvent system, achieving yields between 90.5% and 97.5% while maintaining purity levels as high as 99.9%. For R&D directors and procurement strategists, this represents a significant leap forward in securing a reliable supply chain for electronic chemical manufacturing, offering a pathway to reduce production costs without compromising the stringent quality standards required for optoelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for aromatic ether monomers often suffer from severe inefficiencies that hinder large-scale commercial viability. Conventional methods frequently rely on single-solvent systems that struggle to balance solubility and reaction kinetics, leading to incomplete conversions and the formation of difficult-to-remove impurities. Furthermore, many legacy processes require harsh reaction conditions, such as high pressures or the use of corrosive catalysts that degrade reactor integrity over time, necessitating frequent and costly equipment maintenance. The disposal of waste streams from these older methods is another critical bottleneck, as they often generate substantial volumes of hazardous byproducts that require complex treatment protocols. These factors collectively drive up the operational expenditure (OPEX) and extend lead times, making it challenging for manufacturers to respond agilely to market fluctuations in the fast-paced electronics industry.
The Novel Approach
The methodology outlined in CN101245013A fundamentally reengineers the synthesis landscape by employing a binary solvent system that synergistically enhances reaction efficiency. By combining a water-insoluble organic solvent, such as toluene or xylene, with a strong polar aprotic solvent like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), the process creates an ideal environment for the nucleophilic attack while facilitating the continuous removal of water via azeotropic distillation. This innovative approach allows the reaction to proceed smoothly at atmospheric pressure within a temperature range of 80°C to 200°C, eliminating the safety risks associated with high-pressure vessels. Moreover, the simplicity of the workup procedure—concentrating the reaction液,cooling, and precipitating with water—streamlines the isolation of the final product, ensuring that the 2,4-bis(2,4-dinitrophenoxy)toluene crystals are obtained with minimal downstream processing.
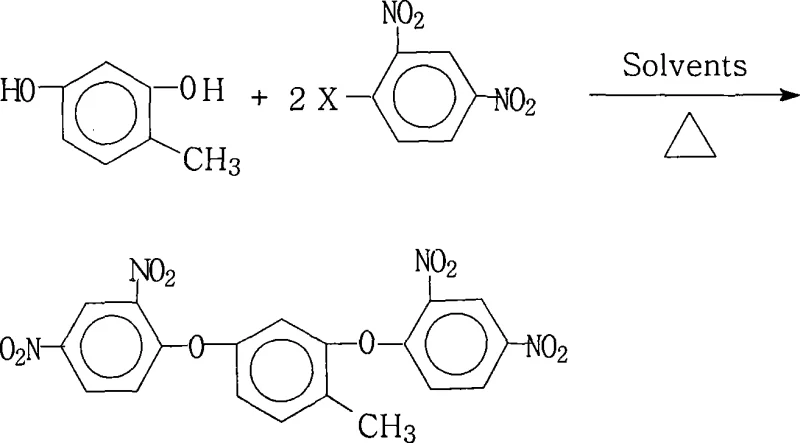
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution ($S_NAr$), facilitated by the strong electron-withdrawing nature of the nitro groups on the benzene ring. In this mechanism, the phenoxide anion, generated in situ by the deprotonation of 2,4-dihydroxytoluene by a base such as potassium carbonate or sodium hydroxide, acts as a potent nucleophile. The presence of two nitro groups at the ortho and para positions relative to the leaving halogen atom significantly activates the aromatic ring towards nucleophilic attack by stabilizing the negatively charged Meisenheimer complex intermediate. This electronic activation is crucial, as it lowers the activation energy barrier, allowing the reaction to proceed efficiently even with less reactive halogen leaving groups like chlorine or bromine, although fluorine is often preferred for its superior leaving group ability in $S_NAr$ contexts. The careful control of the molar ratio, typically maintaining a slight excess of the dinitrohalobenzene (1.0:2.0 to 2.2), ensures that both hydroxyl groups on the toluene core are fully substituted, minimizing the formation of mono-substituted byproducts that could compromise the linearity and thermal properties of the final polyimide.
Impurity control is intrinsically linked to the solvent dynamics and the water removal strategy employed in this patent. The use of a Dean-Stark apparatus or similar water-separation technique during the reflux period is critical for shifting the equilibrium towards product formation by continuously removing the water generated during the salt-formation step. If water accumulates in the reaction medium, it can hydrolyze the sensitive dinitrohalobenzene starting material or reverse the deprotonation of the phenol, leading to stalled reaction kinetics and lower yields. Furthermore, the specific choice of base and its molar ratio (0.10 to 8.00 relative to the dihydroxytoluene) plays a pivotal role in scavenging the acid byproduct (HX) without promoting side reactions such as oxidation of the phenolic moiety. The result is a clean reaction profile where the primary impurity is unreacted starting material, which is easily removed during the aqueous wash step, yielding a product with purity specifications that meet the rigorous demands of high-end electronic material applications.
How to Synthesize 2,4-Bis(2,4-Dinitrophenoxy)Toluene Efficiently
Executing this synthesis requires precise attention to the solvent ratios and thermal profiles to maximize the benefits of the patented process. The procedure begins with the charging of the reactor with the phenolic substrate and the halogenated aromatic compound in the specified molar proportions, followed by the addition of the base and the dual-solvent mixture. The reaction mixture is then heated to reflux, where the azeotropic removal of water drives the reaction to completion over a period ranging from 3 to 18 hours, depending on the specific reactivity of the halogen substituent used. Detailed standardized operating procedures regarding specific flow rates, agitation speeds, and crystallization parameters are essential for reproducibility at scale.
- Charge the reactor with 2,4-dihydroxytoluene and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0-2.2, along with a salt-forming agent like potassium carbonate.
- Add a mixed solvent system comprising a water-insoluble solvent (e.g., toluene) and a polar aprotic solvent (e.g., DMF), then heat to reflux (80°C-200°C) for 3-18 hours with water separation.
- Concentrate the reaction mixture, cool, add water to precipitate the solid, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic advantages that extend beyond mere chemical yield. The ability to operate at atmospheric pressure significantly reduces capital expenditure (CAPEX) requirements, as it eliminates the need for specialized high-pressure autoclaves and the associated safety infrastructure. This simplification of equipment requirements translates directly into lower barriers to entry for production and reduced maintenance overheads, allowing for more flexible manufacturing scheduling. Additionally, the robustness of the solvent system means that the process is less susceptible to minor fluctuations in raw material quality, providing a buffer against supply chain variability that is common in the global chemical market.
- Cost Reduction in Manufacturing: The economic viability of this process is heavily bolstered by the efficient recovery and recycling of the organic solvents. Since the solvent system comprises common industrial chemicals like toluene and DMF, established distillation technologies can be employed to recover over ninety percent of the solvent volume for reuse in subsequent batches. This closed-loop solvent management drastically reduces the recurring cost of raw materials and minimizes the expenses associated with hazardous waste disposal. Furthermore, the elimination of expensive transition metal catalysts, which are often required in cross-coupling alternatives, removes the need for costly metal scavenging steps, further streamlining the production cost structure and enhancing the overall margin potential for the final polyimide product.
- Enhanced Supply Chain Reliability: The raw materials utilized in this synthesis, specifically 2,4-dihydroxytoluene and various dinitrohalobenzenes, are commodity chemicals with well-established global supply chains. Unlike exotic reagents that may be subject to geopolitical restrictions or limited production capacity, these precursors are readily available from multiple vendors, mitigating the risk of supply disruptions. The flexibility of the process to accept different halogen variants (fluoro, chloro, bromo) provides procurement teams with the agility to switch suppliers based on price and availability without needing to requalify the entire manufacturing process. This adaptability ensures a consistent flow of high-purity intermediates, safeguarding the production schedules of downstream polymer manufacturers against market volatility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles. The absence of corrosive reagents and the operation under ambient pressure create a safer working environment, reducing insurance premiums and regulatory compliance burdens. The significant reduction in three wastes (waste water, waste gas, and solid waste) simplifies the permitting process for new production facilities and ensures long-term operational sustainability. As regulatory frameworks regarding industrial emissions tighten globally, adopting a process that inherently generates less waste positions the manufacturer as a responsible partner, future-proofing the supply chain against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chain. The following questions address common concerns regarding scalability, purity, and application suitability, drawing directly from the experimental data and technical specifications provided in the patent literature. These insights are designed to clarify the operational feasibility and commercial potential of producing 2,4-bis(2,4-dinitrophenoxy)toluene using this advanced methodology.
Q: What is the primary application of 2,4-bis(2,4-dinitrophenoxy)toluene?
A: It serves as a critical intermediate for synthesizing highly branched aromatic polyimides, which are essential for high-performance LCDs and aerospace materials due to their thermal stability.
Q: How does this patent improve upon traditional synthesis methods?
A: The method utilizes a recyclable dual-solvent system and operates at atmospheric pressure, eliminating the need for expensive high-pressure equipment and reducing hazardous waste generation significantly.
Q: What purity levels can be achieved with this process?
A: The described process consistently yields products with purity exceeding 99.0%, often reaching up to 99.9%, which is crucial for electronic grade applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Bis(2,4-Dinitrophenoxy)Toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN101245013A can be realized in a full-scale plant setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2,4-bis(2,4-dinitrophenoxy)toluene meets the exacting standards required for high-performance polyimide applications. Our commitment to quality assurance ensures that the thermal and mechanical properties of your final electronic materials remain consistent and reliable.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving market for advanced electronic chemicals.
