Scalable Synthesis of 2,6-bis(2,4-dinitrophenoxy)naphthalene for Advanced Polyimide Applications
The landscape of advanced electronic materials is constantly evolving, driven by the demand for polyimides with superior thermal stability and mechanical properties for applications ranging from aerospace to liquid crystal displays (LCDs). A pivotal breakthrough in this domain is documented in patent CN101234981A, which discloses a highly efficient preparation method for 2,6-bis(2,4-dinitrophenoxy)naphthalene, a critical intermediate for synthesizing highly branched aromatic polyimides. Prior to this innovation, the specific synthetic route for this valuable compound remained largely unexplored in public literature, creating a bottleneck for manufacturers seeking to optimize the performance of next-generation optoelectronic materials. This patent introduces a robust protocol that leverages a specific binary solvent system and controlled stoichiometry to achieve exceptional yields and purity, addressing the longstanding challenges of scalability and environmental compliance in fine chemical synthesis. By enabling the production of high-quality intermediates through a streamlined process, this technology offers a strategic advantage for companies aiming to secure a reliable electronic chemical supplier for their advanced material supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex aromatic ethers, particularly those involving multiple nitro groups and naphthalene cores, has been fraught with significant technical hurdles that impede industrial adoption. Conventional approaches often suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate, thereby drastically reducing the overall process efficiency. Furthermore, many standard etherification protocols rely on harsh reaction conditions or expensive phase-transfer catalysts that leave behind metallic residues, necessitating rigorous and expensive purification steps to meet the stringent purity specifications required for electronic grade materials. The inability to effectively recover solvents in these traditional methods also results in substantial operational expenditures and a heavy environmental burden due to high volumes of organic waste. Additionally, the lack of a standardized, reproducible method for 2,6-bis(2,4-dinitrophenoxy)naphthalene previously forced R&D teams to rely on small-scale, non-optimized laboratory procedures that were ill-suited for commercial scale-up of complex polymer additives.
The Novel Approach
The methodology outlined in patent CN101234981A represents a paradigm shift by introducing a simplified yet highly effective reaction system that overcomes these historical limitations through precise engineering of the reaction environment. By employing a specific molar ratio of 2,6-naphthalenediol to 2,4-dinitrohalobenzene between 1.0:2.0 and 1.0:2.2, the process ensures complete conversion of the diol while minimizing side reactions, resulting in product yields that consistently exceed 87% and often reach nearly 98%. The innovation lies in the use of a mixed solvent system comprising water-insoluble organic solvents like toluene or xylene combined with polar aprotic solvents such as N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP), which facilitates excellent solubility of reactants and efficient water removal during reflux. This approach not only simplifies the post-reaction workup—where the product precipitates cleanly upon the addition of water—but also allows for the convenient recovery and recycling of the organic solvents, drastically reducing raw material consumption. Consequently, this novel approach provides a clear pathway for cost reduction in electronic chemical manufacturing by eliminating the need for complex chromatographic purifications and expensive catalyst removal steps.
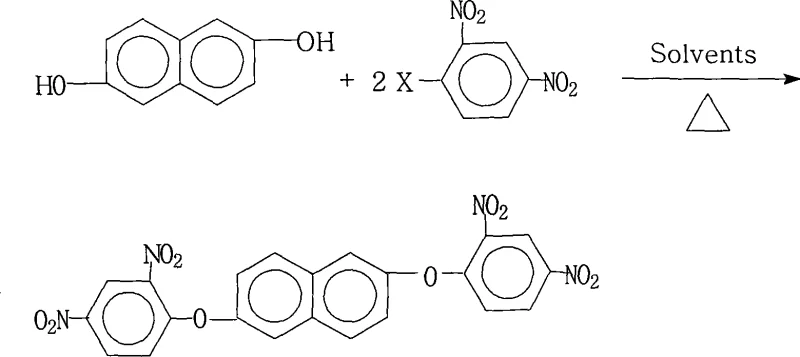
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr) reaction, where the phenoxide anion derived from 2,6-naphthalenediol attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene. The presence of two strongly electron-withdrawing nitro groups at the ortho and para positions relative to the halogen leaving group significantly activates the benzene ring, lowering the energy barrier for the nucleophilic attack and stabilizing the Meisenheimer complex intermediate. The choice of the salt-forming agent, such as potassium carbonate or sodium hydroxide, is critical in generating the reactive phenoxide species in situ without causing excessive degradation of the sensitive nitro groups under the elevated temperatures of 80°C to 200°C. The binary solvent system plays a dual mechanistic role: the polar aprotic component solvates the cation of the salt, increasing the nucleophilicity of the phenoxide anion, while the water-insoluble component facilitates the azeotropic removal of water generated during the salt formation, driving the equilibrium towards the product. This delicate balance of solvation and water management is what allows the reaction to proceed to high conversion rates within a reasonable timeframe of 3 to 18 hours, ensuring that the steric hindrance of the naphthalene core does not impede the double substitution.
From an impurity control perspective, the mechanism inherently favors the formation of the desired 2,6-substituted product due to the symmetry of the 2,6-naphthalenediol starting material and the specific activation pattern of the dinitrohalobenzene. The use of a slight excess of the halobenzene (up to 2.2 equivalents) ensures that the mono-substituted intermediate is rapidly consumed to form the final bis-ether, preventing the accumulation of partially reacted species that could act as chain terminators in subsequent polymerization steps. Furthermore, the precipitation workup strategy exploits the differential solubility of the product versus inorganic salts and unreacted starting materials in the aqueous-organic mixture, effectively purifying the crude product without the need for column chromatography. This mechanistic understanding underscores why the process achieves purity levels above 99%, as the reaction conditions are tuned to minimize the formation of byproducts such as hydrolysis products of the halobenzene or oxidative degradation of the naphthalene ring. For R&D directors, this implies a highly predictable impurity profile that simplifies the validation of the material for use in high-performance polyimide formulations.
How to Synthesize 2,6-bis(2,4-dinitrophenoxy)naphthalene Efficiently
Implementing this synthesis on an industrial scale requires careful attention to the specific parameters outlined in the patent to ensure reproducibility and safety. The process begins with the precise weighing of 2,6-naphthalenediol and the selected 2,4-dinitrohalobenzene, followed by their suspension in the optimized binary solvent mixture along with the chosen base. The reaction is then heated to reflux, typically utilizing a Dean-Stark trap or similar apparatus to continuously remove water, which is essential for driving the equilibrium forward and maintaining the activity of the base. Detailed standard operating procedures regarding temperature ramping, stirring rates, and the specific timing of water addition during the workup phase are critical to obtaining the crystalline product with the desired particle size and purity.
- Charge 2,6-naphthalenediol and 2,4-dinitrohalobenzene (molar ratio 1.0: 2.0-2.2) with a salt-forming agent into a reactor containing a mixture of water-insoluble and polar aprotic solvents.
- Heat the reaction mixture to reflux (80°C-200°C) with water separation for 3 to 18 hours to drive the etherification to completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented method translates into tangible strategic benefits that extend far beyond simple chemical synthesis. The ability to produce this key intermediate with high yield and purity using readily available raw materials like 2,6-naphthalenediol and common nitro-halobenzenes significantly de-risks the supply chain against volatility in specialty reagent markets. Moreover, the simplicity of the operation, which proceeds under normal atmospheric pressure without the need for specialized high-pressure reactors or corrosive reagents, lowers the barrier to entry for manufacturing partners and reduces capital expenditure requirements. The environmental profile of the process, characterized by minimal waste generation and the ability to recycle solvents repeatedly, aligns perfectly with modern sustainability mandates and reduces the long-term costs associated with waste disposal and regulatory compliance. Ultimately, this technology enables a more resilient and cost-effective supply chain for high-purity electronic chemical intermediates, ensuring consistent availability for downstream polymer production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of complex purification techniques like column chromatography lead to substantial cost savings in the overall production budget. By utilizing a solvent system that can be easily recovered and reused multiple times without significant loss of efficiency, the process drastically cuts down on one of the largest variable costs in fine chemical manufacturing. The high atom economy and yield mean that less raw material is wasted, further optimizing the cost per kilogram of the final product. Additionally, the reduced need for extensive wastewater treatment due to the low volume of aqueous waste contributes to lower operational overheads. These factors combine to create a highly competitive cost structure that allows for better margin management in the volatile electronic materials market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, xylene, and potassium carbonate ensures that the supply of raw materials is stable and not subject to the geopolitical risks often associated with exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and times without significant degradation of the product, provides flexibility in scheduling and batch planning, reducing the risk of production delays. The high purity of the crude product means that lead times for quality control testing and release can be shortened, allowing for faster turnover and more responsive delivery to customers. This reliability is crucial for maintaining the continuity of supply for critical applications in the LCD and aerospace sectors where material consistency is paramount.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, as evidenced by the use of standard reflux and filtration equipment that is common in most chemical plants. The absence of corrosive byproducts extends the lifespan of reactor vessels and piping, reducing maintenance downtime and replacement costs over the long term. From an environmental standpoint, the significant reduction in three wastes (waste water, waste gas, and solid waste) simplifies the permitting process and ensures compliance with increasingly stringent environmental regulations globally. This green chemistry approach not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,6-bis(2,4-dinitrophenoxy)naphthalene, based on the detailed specifications provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing polymer synthesis workflows. The answers provided reflect the specific advantages and operational parameters that distinguish this method from alternative synthetic routes.
Q: What are the critical reaction conditions for maximizing yield in this synthesis?
A: The patent specifies a molar ratio of 2,6-naphthalenediol to 2,4-dinitrohalobenzene between 1.0:2.0 and 1.0:2.2, utilizing a binary solvent system of water-insoluble organics (like toluene) and polar aprotic solvents (like DMF or NMP) at temperatures ranging from 80°C to 200°C.
Q: How does this method address environmental concerns in fine chemical production?
A: The process utilizes a solvent system that allows for convenient recovery and repeated recycling, significantly reducing waste generation. Additionally, the reaction operates under normal pressure without generating corrosive byproducts, minimizing equipment corrosion and hazardous waste disposal costs.
Q: What purity levels can be achieved with this preparation method?
A: Experimental data within the patent demonstrates that the method consistently yields products with purity exceeding 99%, with specific examples showing purity levels of 99.1% to 99.5%, making it suitable for high-performance polyimide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-bis(2,4-dinitrophenoxy)naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the development of next-generation electronic materials, and we are uniquely positioned to support your needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this synthesis, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that consistency is key in the electronics industry, and our dedicated process engineering team works tirelessly to optimize every step of the manufacturing process to deliver a product that exceeds expectations. By partnering with us, you gain access to a supply chain that is not only robust and reliable but also deeply committed to innovation and quality assurance.
We invite you to contact our technical procurement team to discuss how we can tailor our production capabilities to your specific volume requirements and timeline. Request a Customized Cost-Saving Analysis today to see how our optimized synthesis route can reduce your overall material costs without compromising on quality. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to being your trusted partner in the global electronic materials market.
