Scalable Synthesis of High-Purity 1,4-Bis(2,4-Dinitrophenoxy) Derivatives for Advanced Polyimide Applications
Scalable Synthesis of High-Purity 1,4-Bis(2,4-Dinitrophenoxy) Derivatives for Advanced Polyimide Applications
The rapid evolution of the electronic materials sector demands intermediates that offer not only exceptional thermal stability but also reproducible synthetic pathways suitable for mass production. Patent CN101215238A introduces a groundbreaking preparation method for 1,4-bis(2,4-dinitrophenoxy)-2,5-di-tert-butylbenzene, a pivotal precursor in the manufacture of advanced aromatic polyimides. This specific chemical architecture is indispensable for generating highly branched polyimide systems that meet the rigorous performance standards of liquid crystal displays (LCDs) and aerospace components. By leveraging a optimized nucleophilic aromatic substitution strategy, this technology addresses the historical lack of industrial protocols for this sterically hindered molecule. For R&D directors and procurement specialists seeking a reliable electronic chemical intermediate supplier, understanding the nuances of this patented route is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of this invention, the synthesis of 1,4-bis(2,4-dinitrophenoxy)-2,5-di-tert-butylbenzene lacked a standardized, industrially viable pathway, often forcing manufacturers to rely on theoretical etherification methods that were ill-suited for such bulky substrates. Conventional approaches to forming diaryl ethers typically struggle when faced with the significant steric hindrance presented by the tert-butyl groups at the 2 and 5 positions of the hydroquinone ring. Traditional conditions often necessitate excessive temperatures or aggressive catalysts that can degrade the sensitive nitro functionalities, leading to complex impurity profiles that are costly to remove. Furthermore, without a dedicated water-separation mechanism, equilibrium limitations in standard substitution reactions frequently result in incomplete conversion and suboptimal yields. These inefficiencies translate directly into higher production costs and inconsistent batch quality, creating significant bottlenecks for companies aiming for cost reduction in electronic chemical manufacturing.
The Novel Approach
The patented methodology overcomes these structural challenges through a meticulously engineered reaction environment that combines a specific molar excess of the halogenated reactant with a dual-solvent system designed for azeotropic water removal. By employing a molar ratio of 2,5-di-tert-butylhydroquinone to 2,4-dinitrohalobenzene ranging from 1.0:2.0 to 1.0:2.2, the process drives the equilibrium firmly toward the desired bis-ether product while minimizing mono-substituted byproducts. The innovation lies in the synergistic use of a water-immiscible solvent, such as toluene or xylene, paired with a polar aprotic solvent like DMF or NMP, which solubilizes the ionic intermediates effectively. This setup allows for heating under reflux with continuous water separation for 6 to 18 hours, ensuring that the reaction proceeds to completion without degrading the thermal sensitivity of the nitro groups. Consequently, this novel approach delivers a robust, scalable solution that transforms a previously elusive laboratory curiosity into a commercially viable commodity.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core of this synthesis relies on a classic yet finely tuned Nucleophilic Aromatic Substitution (SnAr) mechanism, where the electron-deficient nature of the 2,4-dinitrohalobenzene plays a critical role in facilitating the attack by the phenoxide anion. The presence of two strong electron-withdrawing nitro groups ortho and para to the leaving halogen atom significantly activates the aromatic ring, lowering the energy barrier for the formation of the Meisenheimer complex intermediate. In this specific system, the salt-forming agent, which can range from potassium carbonate to potassium hydroxide, deprotonates the hydroquinone hydroxyl groups to generate the active nucleophile in situ. The steric bulk of the tert-butyl groups requires careful management of reaction kinetics, which is achieved by maintaining temperatures between 80°C and 200°C depending on the specific solvent boiling point. This thermal energy ensures sufficient molecular collision frequency to overcome the steric repulsion without triggering unwanted decomposition pathways.
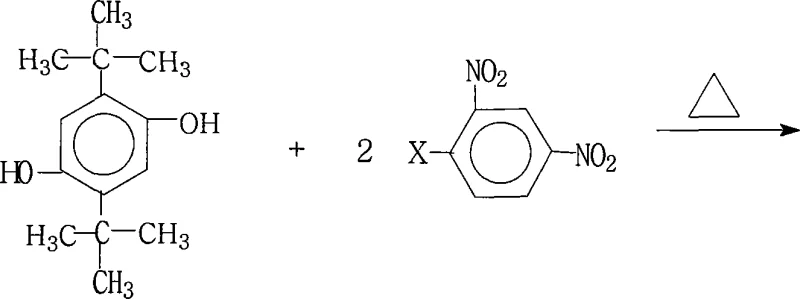
Impurity control is inherently built into the mechanistic design through the precise stoichiometry and the choice of the leaving group X, which can be fluorine, chlorine, bromine, or iodine. While fluorine is often the most reactive in SnAr reactions due to its high electronegativity, the patent demonstrates flexibility by allowing various halogens, providing procurement teams with options based on raw material availability and cost. The use of a base in slight excess relative to the hydroquinone ensures complete deprotonation, preventing the accumulation of unreacted starting material which could complicate downstream purification. Furthermore, the precipitation step, where water is added to the concentrated reaction mass, exploits the low solubility of the highly non-polar product in aqueous media, effectively crashing out the pure晶体 while leaving soluble inorganic salts and polar byproducts in the mother liquor. This elegant work-up procedure eliminates the need for column chromatography, making the process ideal for multi-kilogram scale-up.
How to Synthesize 1,4-Bis(2,4-Dinitrophenoxy) Efficiently
Executing this synthesis requires strict adherence to the defined parameters regarding solvent ratios and thermal profiles to maximize the yield, which has been demonstrated to reach as high as 96.7% in optimized embodiments. The process begins with the charging of the reactor with the hydroquinone derivative and the dinitrohalobenzene in a mixture of solvents, followed by the addition of the inorganic base. Operators must monitor the reflux and water separation closely, as the removal of the water byproduct is thermodynamic driver for the reaction completion. Once the reaction time of 6 to 18 hours is concluded, the solvent recovery phase becomes critical for economic viability, allowing the organic phase to be distilled and reused in subsequent batches.
- Charge reactor with 2,5-di-tert-butylhydroquinone and 2,4-dinitrohalobenzene at a molar ratio of 1.0: 2.0-2.2 in a mixed solvent system.
- Add salt-forming agent (e.g., potassium carbonate) and heat under reflux with water separation for 6-18 hours at 80-200°C.
- Concentrate reaction mixture, cool, precipitate product with water, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this patented process represents a significant opportunity to stabilize the sourcing of high-value polyimide precursors while simultaneously driving down operational expenditures. The ability to recover and recycle the organic solvents, such as toluene and DMF, drastically reduces the volume of hazardous waste generated per kilogram of product, aligning with increasingly stringent environmental regulations and reducing disposal costs. Moreover, the simplicity of the isolation procedure, which relies on straightforward filtration and drying rather than complex distillation or chromatography, minimizes the equipment footprint and energy consumption required for production. These factors combine to create a manufacturing profile that is not only cost-effective but also highly resilient to fluctuations in raw material pricing, offering a stable supply foundation for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reliance on commodity inorganic bases like potassium carbonate significantly lowers the direct material costs associated with production. By avoiding the need for specialized catalysts, the process also removes the requirement for costly metal scavenging steps, which are often necessary to meet the ultra-low metal specifications demanded by the electronics industry. The high atom economy of the reaction, coupled with the ability to recycle solvents multiple times without loss of efficacy, ensures that the overall cost per unit is minimized. This economic efficiency allows suppliers to offer competitive pricing structures without compromising on the stringent quality standards required for electronic grade materials.
- Enhanced Supply Chain Reliability: The raw materials utilized in this synthesis, specifically 2,5-di-tert-butylhydroquinone and various dinitrohalobenzenes, are commercially available from multiple global sources, mitigating the risk of single-supplier dependency. The robustness of the reaction conditions, which tolerate a range of halogens and bases, provides procurement managers with the flexibility to switch feedstock grades or suppliers based on market availability without revalidating the entire process. This adaptability is crucial for maintaining continuous production schedules in the face of global logistics disruptions or regional raw material shortages. Consequently, partners can rely on a consistent flow of high-purity intermediates to support their own downstream polymerization activities.
- Scalability and Environmental Compliance: The process is designed for easy scale-up from laboratory benchtop to industrial reactors, as evidenced by the successful demonstration of yields exceeding 96% in pilot-scale examples. The absence of corrosive reagents and the operation at atmospheric pressure reduce the engineering complexity and safety risks associated with high-pressure hydrogenation or acid-catalyzed reactions. Additionally, the minimal generation of three wastes (waste water, gas, and solids) due to solvent recycling and efficient precipitation aligns with green chemistry principles. This environmental compatibility simplifies the permitting process for new manufacturing facilities and ensures long-term operational sustainability in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate, derived directly from the patent specifications and industrial best practices. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing supply chains.
Q: What is the primary application of 1,4-bis(2,4-dinitrophenoxy)-2,5-di-tert-butylbenzene?
A: This compound serves as a critical precursor for synthesizing aromatic polyvalent primary amines, which are essential monomers for producing high-performance aromatic polyimides used in LCDs and aerospace materials.
Q: How does this process ensure high product purity?
A: The method utilizes a specific mixed solvent system and controlled reflux conditions that minimize side reactions, allowing for direct crystallization with purity levels exceeding 99% without complex chromatographic purification.
Q: Is the solvent system recyclable for cost efficiency?
A: Yes, the organic solvents employed, such as toluene and DMF, can be effectively recovered and recycled after concentration, significantly reducing raw material costs and environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bis(2,4-Dinitrophenoxy) Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced polyimides hinges on the availability of ultra-pure monomers produced via robust, scalable pathways. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the absence of critical impurities that could affect the thermal or electrical properties of the final polymer. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and commercial reliability, safeguarding your production timelines against potential disruptions.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and quality targets. Request a Customized Cost-Saving Analysis today to understand how switching to our supply model can optimize your bill of materials. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner in the electronic materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
