Advanced Synthesis of 2,3-Bis(2,4-Dinitrophenoxy)Naphthalene for High-Performance Polyimide Manufacturing
The rapid advancement of the optoelectronics and aerospace sectors has created an insatiable demand for high-performance aromatic polyimides, materials renowned for their exceptional thermal stability and mechanical strength. At the heart of synthesizing these advanced polymers lies the critical need for high-purity monomers, specifically 2,3-bis(2,4-dinitrophenoxy)naphthalene, which serves as a pivotal precursor for highly branched aromatic polyamine systems. Patent CN101284788B introduces a groundbreaking preparation method that addresses the historical lack of reported synthesis routes for this specific compound. By leveraging a sophisticated nucleophilic aromatic substitution strategy within a tailored mixed-solvent environment, this technology enables the production of the target molecule with unprecedented efficiency and purity. For R&D directors and procurement specialists alike, understanding this patented route is essential, as it represents a significant leap forward in cost reduction in electronic chemical manufacturing, offering a streamlined pathway from basic aromatic feedstocks to high-value functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diaryl ethers involving naphthalene derivatives has been plagued by significant technical hurdles that hindered widespread industrial adoption. Traditional approaches often struggled with the poor solubility of dihydroxynaphthalenes in standard organic media, leading to heterogeneous reaction conditions that drastically reduced reaction rates and overall yields. Furthermore, conventional methods frequently relied on harsh conditions or expensive phase-transfer catalysts that complicated the downstream purification process, introducing metallic impurities that are detrimental to electronic-grade applications. The inability to effectively remove the water byproduct generated during the etherification process often resulted in reversible reactions, limiting conversion and necessitating energy-intensive distillation steps. These inefficiencies not only inflated production costs but also generated substantial chemical waste, creating environmental compliance challenges for manufacturers striving to meet modern green chemistry standards.
The Novel Approach
The methodology disclosed in patent CN101284788B fundamentally reimagines this synthetic challenge by employing a dual-solvent system coupled with azeotropic water removal. By combining a water-insoluble organic solvent, such as toluene or xylene, with a highly polar aprotic solvent like N-methyl-2-pyrrolidone (NMP) or dimethylformamide (DMF), the process ensures optimal solubility for both the phenolic substrate and the nitro-halobenzene electrophile. This innovative solvent engineering allows the reaction to proceed homogeneously at elevated temperatures (80°C to 200°C) while continuously separating the generated water, thereby irreversibly driving the equilibrium toward the desired product. The result is a robust, scalable process that eliminates the need for exotic catalysts and simplifies the isolation of the final crystalline product through simple water precipitation. 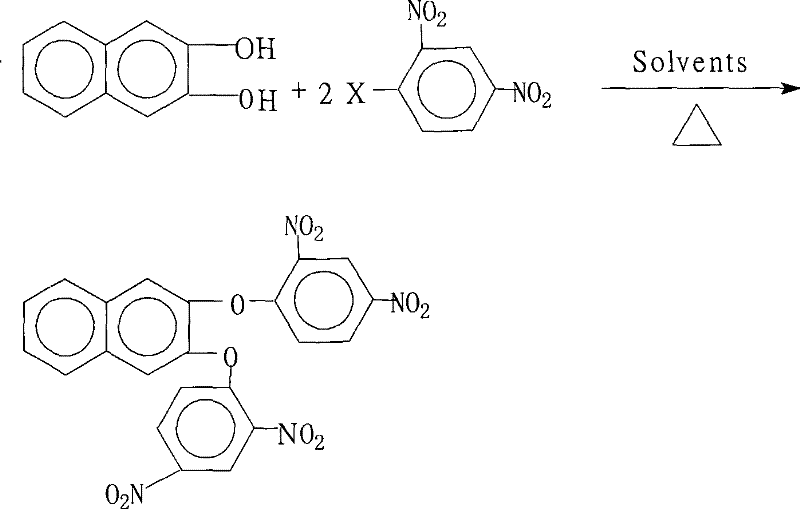
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution ($S_NAr$), facilitated by the strong electron-withdrawing nature of the nitro groups on the benzene ring. In this mechanism, the 2,3-dihydroxynaphthalene acts as the nucleophile, but it must first be activated by the salt-forming agent, typically an alkali metal carbonate or hydroxide. This base deprotonates the hydroxyl groups to form the corresponding phenoxide anions, which are significantly more nucleophilic than their neutral counterparts. These anions then attack the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene at the position ortho or para to the nitro groups, displacing the halide leaving group. The presence of two nitro groups greatly stabilizes the Meisenheimer complex intermediate, lowering the activation energy and allowing the reaction to proceed efficiently even with less reactive halides like chlorine or bromine.
From an impurity control perspective, the choice of reaction parameters is critical to minimizing side reactions such as over-alkylation or hydrolysis of the nitro groups. The patent specifies a precise molar ratio of 1.0:2.0 to 2.2 between the diol and the halobenzene, ensuring that there is a slight excess of the electrophile to drive the double substitution to completion without leaving unreacted mono-substituted intermediates. Furthermore, the controlled addition of the salt-forming agent prevents localized high pH environments that could degrade the sensitive nitro functionalities. The subsequent workup, involving concentration and water addition, exploits the differential solubility of the product versus the inorganic salts and unreacted starting materials, ensuring that the final isolated crystals possess the high purity (>99%) required for downstream polymerization into high-performance polyimides.
How to Synthesize 2,3-Bis(2,4-Dinitrophenoxy)Naphthalene Efficiently
Implementing this synthesis on a pilot or production scale requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful charging of the reactor with the specified molar ratios of 2,3-naphthalenediol and 2,4-dinitrohalobenzene, followed by the addition of the mixed solvent system and the base. The reaction mixture is then heated to reflux, where the azeotropic removal of water becomes the rate-limiting step that dictates the overall reaction time, typically ranging from 3 to 18 hours depending on the specific solvent blend and scale. Detailed standardized operating procedures regarding temperature ramping, stirring speeds, and the precise timing of the water separation are critical for maximizing yield.
- Charge the reactor with 2,3-naphthalenediol and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0-2.2, along with a salt-forming agent and a mixed solvent system.
- Heat the mixture to reflux (80°C-200°C) with continuous water separation for 3 to 18 hours to drive the nucleophilic substitution reaction to completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash, and dry to obtain the final crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals like carbonates and common organic solvents drastically simplifies the raw material sourcing landscape. This shift reduces exposure to volatile pricing associated with specialty reagents and mitigates the risk of supply disruptions caused by the limited availability of niche catalysts. Moreover, the ability to recover and recycle the organic solvent system multiple times without significant loss of performance creates a closed-loop manufacturing process that substantially lowers the variable cost per kilogram of the final product.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its operational simplicity and the avoidance of costly purification steps. By utilizing a precipitation-based isolation method rather than complex chromatography or distillation, the process significantly reduces energy consumption and labor hours. The use of inexpensive halobenzenes (such as chloro- or bromo- derivatives) instead of more expensive fluoro- analogs, where feasible, further drives down the Bill of Materials (BOM) cost. Additionally, the high atom economy of the substitution reaction minimizes waste generation, reducing the financial burden associated with hazardous waste disposal and environmental compliance fees.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route directly translates to improved supply chain resilience for reliable polyimide intermediate supplier networks. Since the reaction operates under atmospheric pressure and does not involve corrosive reagents that degrade equipment, maintenance downtime is minimized, and reactor turnover rates are optimized. The starting materials, 2,3-dihydroxynaphthalene and dinitrohalobenzenes, are widely produced bulk chemicals with stable global supply chains, ensuring that production schedules are not held hostage by the scarcity of exotic precursors. This reliability allows for consistent lead times and the ability to scale production volumes rapidly in response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this process from laboratory grams to multi-tonne commercial batches is straightforward due to the absence of exothermic hazards or high-pressure requirements. The solvent system is designed for easy recovery, aligning with modern sustainability goals and reducing the facility's overall carbon footprint. The minimal generation of aqueous waste, primarily consisting of inorganic salts, simplifies wastewater treatment protocols. This environmental friendliness not only future-proofs the manufacturing site against tightening regulations but also enhances the brand value of the final electronic materials by associating them with green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear framework for evaluating the feasibility of adopting this route for commercial scale-up of complex polymer additives. Understanding these nuances is vital for technical teams assessing the integration of this intermediate into their existing production lines.
Q: What is the primary advantage of the solvent system used in this synthesis?
A: The patent utilizes a mixed solvent system comprising a water-insoluble organic solvent (like toluene) and a polar aprotic solvent (like NMP or DMF). This combination facilitates the dissolution of reactants while allowing for the efficient azeotropic removal of water generated during the reaction, significantly driving the equilibrium towards the product and enabling solvent recycling.
Q: Can this process be scaled for industrial production of polyimide monomers?
A: Yes, the process is explicitly designed for industrialization. It operates under atmospheric pressure without requiring specialized corrosion-resistant equipment, uses readily available raw materials, and features a simple workup procedure involving precipitation and filtration, making it highly suitable for commercial scale-up.
Q: What purity levels can be achieved with this preparation method?
A: According to the experimental data in patent CN101284788B, the method consistently yields products with purity exceeding 99%. Specific examples demonstrate purities of 99.1% to 99.5%, which is critical for downstream applications in high-performance electronic materials like LCDs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Bis(2,4-Dinitrophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation polyimides depends on the consistent availability of ultra-high-purity monomers like 2,3-bis(2,4-dinitrophenoxy)naphthalene. Our technical team has thoroughly analyzed the pathways described in CN101284788B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and state-of-the-art manufacturing facilities capable of meeting stringent purity specifications, ensuring that every batch delivered meets the exacting standards required for LCD and aerospace applications. Our commitment to quality assurance means that we can guarantee the structural integrity and low impurity profiles necessary for high-performance polymer synthesis.
We invite potential partners to engage with our technical procurement team to discuss how we can tailor our production capabilities to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency. Let us be your strategic partner in bridging the gap between innovative patent chemistry and industrial reality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
